J Stroke.
2024 Jan;26(1):13-25. 10.5853/jos.2023.01578.
Update of Anticoagulation Use in Cardioembolic Stroke With a Special Reference to Endovascular Treatment
- Affiliations
-
- 1Stroke Unit, Metropolitan Hospital, Piraeus, Greece
- 2Second Department of Neurology, “Attikon” University Hospital, School of Medicine, National and Kapodistrian University of Athens, Athens, Greece
- 3Department of Neurointerventions, National Institute of Mental Health, Neurology and Neurosurgery, Semmelweis University, Department of Neurosurgery, Budapest, Hungary
- 4Interventional Neuroradiology Unit, Metropolitan Hospital, Piraeus, Greece
- 5Second Department of Radiology, Interventional Radiology Unit, “Attikon” University Hospital, Athens, Greece
- 6Second Department of Neurology, AHEPA University Hospital, Aristotle University of Thessaloniki, School of Medicine, Thessaloniki, Greece
- 7Department of Neurology, University Hospitals Cleveland Medical Center, Cleveland, OH, USA
- 8Department of Neurology and Radiology, Boston Medical Center, Boston University Chobanian and Avedisian School of Medicine, Boston, MA, USA
- 9Department of Neurology, Rhode Island Hospital, Alpert Medical School of Brown University, Providence, RI, USA
- 10Department of Neurology, Saarland University Medical Centre, Homburg, Germany
- 11Department of Applied Clinical Sciences and Biotechnology, University of L’Aquila, L’Aquila, Italy
- 12Department of Neurology, GHU Paris Psychiatrie et Neurosciences, Paris, France
- 13INSERM U1266, Paris, France
- 14FHU Neurovasc, Paris, France
- 15Université Paris Cité, Paris, France
- 16Department of Neurology, University of Tennessee Health Science Center, Memphis, TN, USA
- KMID: 2551344
- DOI: http://doi.org/10.5853/jos.2023.01578
Abstract
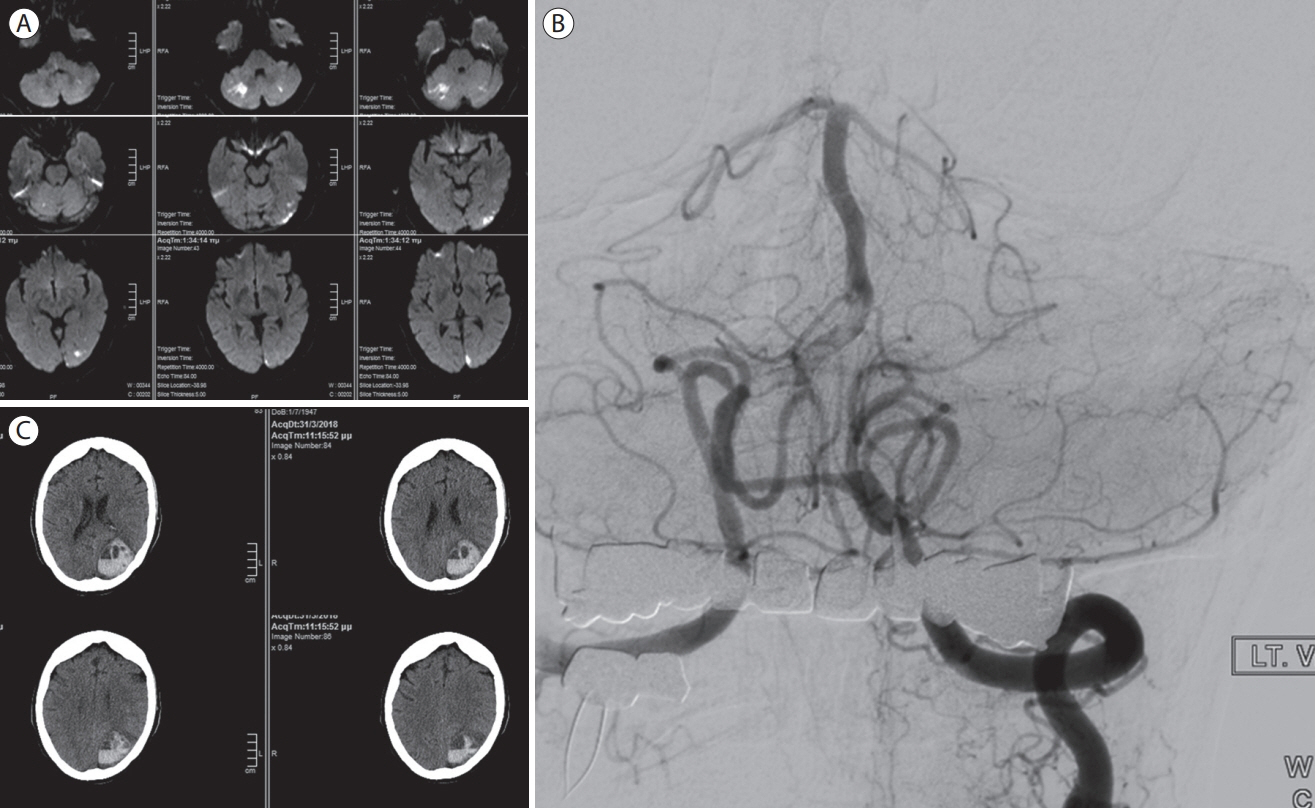
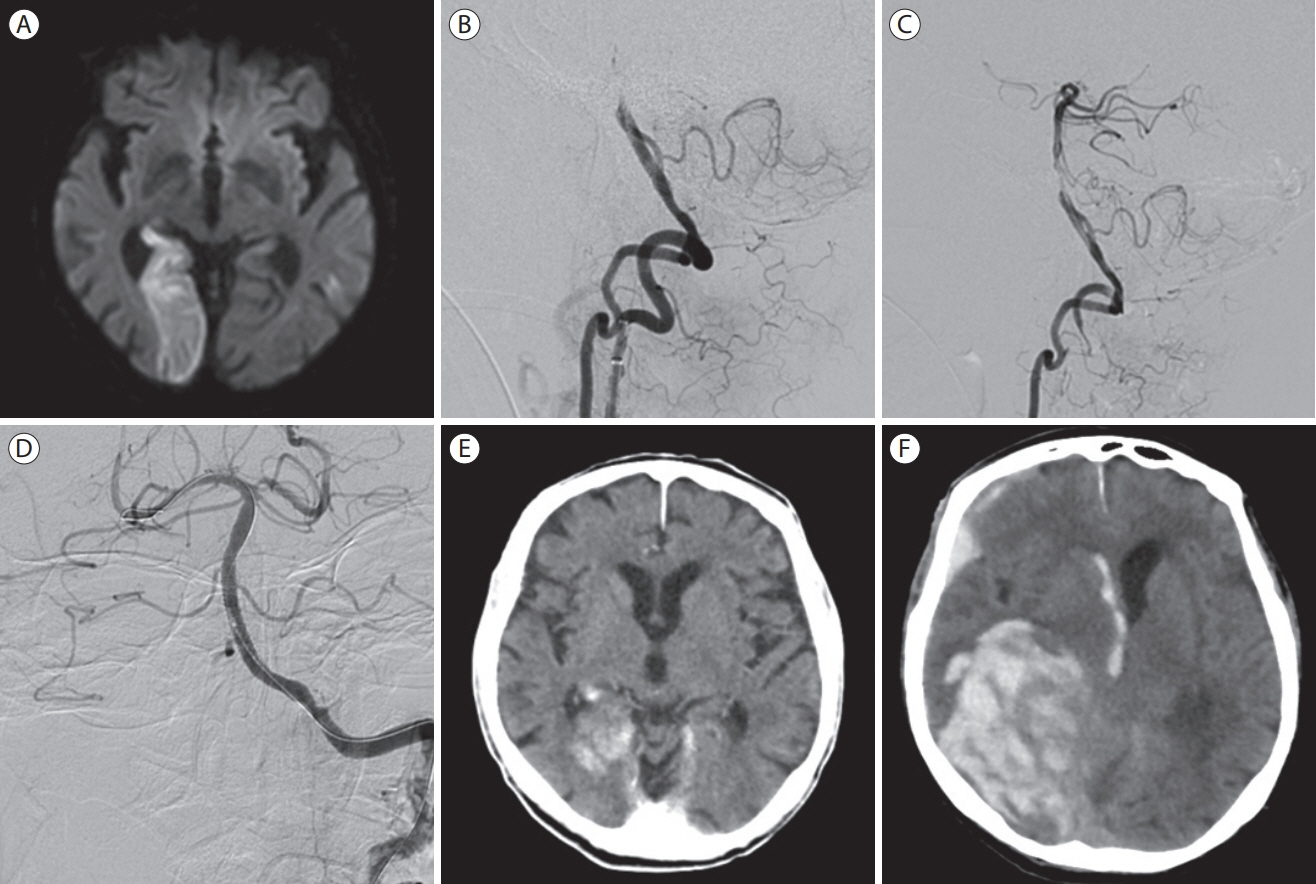
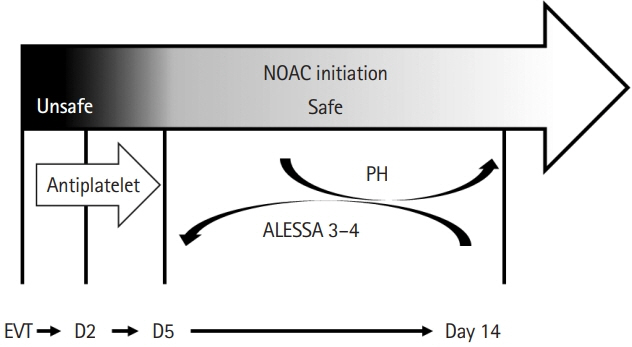
- Cardioembolic stroke is a major cause of morbidity, with a high risk of recurrence, and anticoagulation represents the mainstay of secondary stroke prevention in most patients. The implementation of endovascular treatment in routine clinical practice complicates the decision to initiate anticoagulation, especially in patients with early hemorrhagic transformation who are considered at higher risk of hematoma expansion. Late hemorrhagic transformation in the days and weeks following stroke remains a potentially serious complication for which we still do not have any established clinical or radiological prediction tools. The optimal time to initiate therapy is challenging to define since delaying effective secondary prevention treatment exposes patients to the risk of recurrent embolism. Consequently, there is clinical equipoise to define and individualize the optimal timepoint to initiate anticoagulation combining the lowest risk of hemorrhagic transformation and ischemic recurrence in cardioembolic stroke patients. In this narrative review, we will highlight and critically outline recent observational and randomized relevant evidence in different subtypes of cardioembolic stroke with a special focus on anticoagulation initiation following endovascular treatment. We will refer mainly to the commonest cause of cardioembolism, non-valvular atrial fibrillation, and examine the possible risk and benefit of anticoagulation before, during, and shortly after the acute phase of stroke. Other indications of anticoagulation after ischemic stroke will be briefly discussed. We provide a synthesis of available data to help clinicians individualize the timing of initiation of oral anticoagulation based on the presence and extent of hemorrhagic transformation as well as stroke severity.
Keyword
Figure
Reference
-
References
1. Aguilar MI, Hart R, Pearce LA. Oral anticoagulants versus antiplatelet therapy for preventing stroke in patients with non-valvular atrial fibrillation and no history of stroke or transient ischemic attacks. Cochrane Database Syst Rev. 2007; 3:CD006186.2. Liu C, Shi F, Chen Z, Yan S, Ding X, Lou M. Severe blood-brain barrier disruption in cardioembolic stroke. Front Neurol. 2018; 9:55.3. Kobeissi H, Ghozy S, Seymour T, Gupta R, Bilgin C, Kadirvel R, et al. Outcomes of patients with atrial fibrillation following thrombectomy for stroke: a systematic review and meta-analysis. JAMA Netw Open. 2023; 6:e2249993.4. Berge E, Abdelnoor M, Nakstad PH, Sandset PM. Low molecular-weight heparin versus aspirin in patients with acute ischaemic stroke and atrial fibrillation: a double-blind randomised study. Lancet. 2000; 355:1205–1210.5. Saxena R, Lewis S, Berge E, Sandercock PA, Koudstaal PJ. Risk of early death and recurrent stroke and effect of heparin in 3169 patients with acute ischemic stroke and atrial fibrillation in the international stroke trial. Stroke. 2001; 32:2333–2337.6. Fang MC, Go AS, Chang Y, Borowsky LH, Pomernacki NK, Udaltsova N, et al. Thirty-day mortality after ischemic stroke and intracranial hemorrhage in patients with atrial fibrillation on and off anticoagulants. Stroke. 2012; 43:1795–1799.7. Paciaroni M, Agnelli G, Corea F, Ageno W, Alberti A, Lanari A, et al. Early hemorrhagic transformation of brain infarction: rate, predictive factors, and influence on clinical outcome: results of a prospective multicenter study. Stroke. 2008; 39:2249–2256.8. van der Steen W, van der Ende NA, van Kranendonk KR, Chalos V, Brouwer J, van Oostenbrugge RJ, et al. Timing of symptomatic intracranial hemorrhage after endovascular stroke treatment. Eur Stroke J. 2022; 7:393–401.9. Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, et al. 2016 ESC guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur J Cardiothorac Surg. 2016; 50:e1–e88.10. Ahmed N, Steiner T, Caso V, Wahlgren N; ESO-KSU Session Participants. Recommendations from the ESO-Karolinska Stroke Update Conference, Stockholm 13-15 November 2016. Eur Stroke J. 2017; 2:95–102.11. Powers WJ, Rabinstein AA, Ackerson T, Adeoye OM, Bambakidis NC, Becker K, et al. Guidelines for the early management of patients with acute ischemic stroke: 2019 update to the 2018 guidelines for the early management of acute ischemic stroke: a guideline for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2019; 50:e344–e418.12. Nogueira RG, Gupta R, Jovin TG, Levy EI, Liebeskind DS, Zaidat OO, et al. Predictors and clinical relevance of hemorrhagic transformation after endovascular therapy for anterior circulation large vessel occlusion strokes: a multicenter retrospective analysis of 1122 patients. J Neurointerv Surg. 2015; 7:16–21.13. Hacke W, Kaste M, Fieschi C, von Kummer R, Davalos A, Meier D, et al. Randomised double-blind placebo-controlled trial of thrombolytic therapy with intravenous alteplase in acute ischaemic stroke (ECASS II). Lancet. 1998; 352:1245–1251.14. Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, Guidetti D, et al. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008; 359:1317–1329.15. Wahlgren N, Ahmed N, Dávalos A, Ford GA, Grond M, Hacke W, et al. Thrombolysis with alteplase for acute ischaemic stroke in the Safe Implementation of Thrombolysis in Stroke-Monitoring Study (SITS-MOST): an observational study. Lancet. 2007; 369:275–282.16. von Kummer R, Broderick JP, Campbell BC, Demchuk A, Goyal M, Hill MD, et al. The Heidelberg bleeding classification: classification of bleeding events after ischemic stroke and reperfusion therapy. Stroke. 2015; 46:2981–2986.17. Paciaroni M, Bandini F, Agnelli G, Tsivgoulis G, Yaghi S, Furie KL, et al. Hemorrhagic transformation in patients with acute ischemic stroke and atrial fibrillation: time to initiation of oral anticoagulant therapy and outcomes. J Am Heart Assoc. 2018; 7:e010133.18. Tsivgoulis G, Katsanos AH, Seiffge DJ, Paciaroni M, Wilson D, Koga M, et al. Fatal intracranial haemorrhage occurring after oral anticoagulant treatment initiation for secondary stroke prevention in patients with atrial fibrillation. Eur J Neurol. 2020; 27:1612–1617.19. Seiffge DJ, Paciaroni M, Wilson D, Koga M, Macha K, Cappellari M, et al. Direct oral anticoagulants versus vitamin K antagonists after recent ischemic stroke in patients with atrial fibrillation. Ann Neurol. 2019; 85:823–834.20. Yaghi S, Mistry E, Liberman AL, Giles J, Asad SD, Liu A, et al. Anticoagulation type and early recurrence in cardioembolic stroke: the IAC study. Stroke. 2020; 51:2724–2732.21. Hong KS, Kwon SU, Lee SH, Lee JS, Kim YJ, Song TJ, et al. Rivaroxaban vs warfarin sodium in the ultra-early period after atrial fibrillation–related mild ischemic stroke: a randomized clinical trial. JAMA Neurol. 2017; 74:1206–1215.22. Palaiodimou L, Stefanou MI, Katsanos AH, Paciaroni M, Sacco S, De Marchis GM, et al. Early anticoagulation in patients with acute ischemic stroke due to atrial fibrillation: a systematic review and meta-analysis. J Clin Med. 2022; 11:4981.23. Polymeris AA, Zietz A, Schaub F, Meya L, Traenka C, Thilemann S, et al. Once versus twice daily direct oral anticoagulants in patients with recent stroke and atrial fibrillation. Eur Stroke J. 2022; 7:221–229.24. Klijn CJ, Paciaroni M, Berge E, Korompoki E, Kõrv J, Lal A, et al. Antithrombotic treatment for secondary prevention of stroke and other thromboembolic events in patients with stroke or transient ischemic attack and non-valvular atrial fibrillation: a European Stroke Organisation guideline. Eur Stroke J. 2019; 4:198–223.25. Seiffge DJ, Werring DJ, Paciaroni M, Dawson J, Warach S, Milling TJ, et al. Timing of anticoagulation after recent ischaemic stroke in patients with atrial fibrillation. Lancet Neurol. 2019; 18:117–126.26. Malhotra K, Khunger M, Liebeskind DS. Non-vitamin K oral anticoagulants in stroke patients: practical issues. J Stroke. 2017; 19:104–106.27. Paciaroni M, Agnelli G, Caso V, Tsivgoulis G, Furie KL, Tadi P, et al. Prediction of early recurrent thromboembolic event and major bleeding in patients with acute stroke and atrial fibrillation by a risk stratification schema: the ALESSA score study. Stroke. 2017; 48:726–732.28. Yaghi S, Henninger N, Giles JA, Leon Guerrero C, Mistry E, Liberman AL, et al. Ischaemic stroke on anticoagulation therapy and early recurrence in acute cardioembolic stroke: the IAC study. J Neurol Neurosurg Psychiatry. 2021; 92:1062–1067.29. Yaghi S. Diagnosis and management of cardioembolic stroke. Continuum (Minneap Minn). 2023; 29:462–485.30. Kimura S, Toyoda K, Yoshimura S, Minematsu K, Yasaka M, Paciaroni M, et al. Practical “1-2-3-4-day” rule for starting direct oral anticoagulants after ischemic stroke with atrial fibrillation: combined hospital-based cohort study. Stroke. 2022; 53:1540–1549.31. Oldgren J, Åsberg S, Hijazi Z, Wester P, Bertilsson M, Norrving B; National TIMING Collaborators. Early versus delayed non–vitamin K antagonist oral anticoagulant therapy after acute ischemic stroke in atrial fibrillation (TIMING): a registry-based randomized controlled noninferiority study. Circulation. 2022; 146:1056–1066.32. Steffel J, Collins R, Antz M, Cornu P, Desteghe L, Haeusler KG, et al. 2021 European Heart Rhythm Association practical guide on the use of non-vitamin K antagonist oral anticoagulants in patients with atrial fibrillation. Europace. 2021; 23:1612–1676.33. Connolly SJ, Karthikeyan G, Ntsekhe M, Haileamlak A, El Sayed A, El Ghamrawy A, et al. Rivaroxaban in rheumatic heart disease-associated atrial fibrillation. N Engl J Med. 2022; 387:978–988.34. Eikelboom JW, Connolly SJ, Brueckmann M, Granger CB, Kappetein AP, Mack MJ, et al. Dabigatran versus warfarin in patients with mechanical heart valves. N Engl J Med. 2013; 369:1206–1214.35. Fanaroff AC, Vora AN, Lopes RD. Non-vitamin K antagonist oral anticoagulants in patients with valvular heart disease. Eur Heart J Suppl. 2022; 24(Suppl A):A19–A31.36. Jawitz OK, Wang TY, Lopes RD, Chavez A, Boyer B, Kim H, et al. Rationale and design of PROACT Xa: a randomized, multicenter, open-label, clinical trial to evaluate the efficacy and safety of apixaban versus warfarin in patients with a mechanical On-X aortic heart valve. Am Heart J. 2020; 227:91–99.37. Neale T. Another DOAC fails in the setting of mechanical heart valves [Internet]. New York: TCTMD; 2022 September 28 [accessed May 6, 2023]. Available from: https://www.tctmd.com/news/another-doac-fails-setting-mechanical-heart-valves.38. Otto CM, Nishimura RA, Bonow RO, Carabello BA, Erwin JP 3rd, Gentile F, et al. 2020 ACC/AHA guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Joint Committee on clinical practice guidelines. Circulation. 2021; 143:e72–e227.39. Kuramatsu JB, Sembill JA, Gerner ST, Sprügel MI, Hagen M, Roeder SS, et al. Management of therapeutic anticoagulation in patients with intracerebral haemorrhage and mechanical heart valves. Eur Heart J. 2018; 39:1709–1723.40. Kleindorfer DO, Towfighi A, Chaturvedi S, Cockroft KM, Gutierrez J, Lombardi-Hill D, et al. 2021 guideline for the prevention of stroke in patients with stroke and transient ischemic attack: a guideline from the American Heart Association/American Stroke Association. Stroke. 2021; 52:e364–e467.41. Robinson AA, Trankle CR, Eubanks G, Schumann C, Thompson P, Wallace RL, et al. Off-label use of direct oral anticoagulants compared with warfarin for left ventricular thrombi. JAMA Cardiol. 2020; 5:685–692.42. Huang L, Tan Y, Pan Y. Systematic review of efficacy of direct oral anticoagulants and vitamin K antagonists in left ventricular thrombus. ESC Heart Fail. 2022; 9:3519–3532.43. Kelly J, Rudd A, Lewis R, Hunt BJ. Venous thromboembolism after acute stroke. Stroke. 2001; 32:262–267.44. Aboul-Nour H, Maraey A, Jumah A, Khalil M, Elzanaty AM, Elsharnoby H, et al. Mechanical thrombectomy for acute ischemic stroke in metastatic cancer patients: a nationwide cross-sectional analysis. J Stroke. 2023; 25:119–125.45. Gómez-Outes A, Lecumberri R, Suárez-Gea ML, Terleira-Fernández AI, Monreal M, Vargas-Castrillón E. Case fatality rates of recurrent thromboembolism and bleeding in patients receiving direct oral anticoagulants for the initial and extended treatment of venous thromboembolism: a systematic review. J Cardiovasc Pharmacol Ther. 2015; 20:490–500.46. Psychogios K, Kargiotis O, Safouris A, Repousis P, Tsivgoulis G. Floating carotid thrombus in a patient with essential thrombocytemia due to calreticulin mutation. Stroke. 2023; 54:e207–e208.47. Singh RJ, Chakraborty D, Dey S, Ganesh A, Al Sultan AS, Eesa M, et al. Intraluminal thrombi in the cervico-cephalic arteries. Stroke. 2019; 50:357–364.48. Das S, Goldstein ED, de Havenon A, Abbasi M, Nguyen TN, Aguiar de Sousa D, et al. Composition, treatment, and outcomes by radiologically defined thrombus characteristics in acute ischemic stroke. Stroke. 2023; 54:1685–1694.49. Kim JS, Lee KB, Chang DI, Cha JK, Lee JS. Early administration of edoxaban after acute ischemic stroke in patients with non-valvular atrial fibrillation: a pilot randomized trial. J Stroke. 2023; 25:311–314.50. Fischer U, Koga M, Strbian D, Branca M, Abend S, Trelle S, et al. Early versus later anticoagulation for stroke with atrial fibrillation. N Engl J Med. 2023; 388:2411–2421.51. Best JG, Arram L, Ahmed N, Balogun M, Bennett K, Bordea E, et al. Optimal timing of anticoagulation after acute ischemic stroke with atrial fibrillation (OPTIMAS): protocol for a randomized controlled trial. Int J Stroke. 2022; 17:583–589.52. King BT, Lawrence PD, Milling TJ, Warach SJ. Optimal delay time to initiate anticoagulation after ischemic stroke in atrial fibrillation (START): methodology of a pragmatic, response-adaptive, prospective randomized clinical trial. Int J Stroke. 2019; 14:977–982.53. Alrohimi A, Jickling G, Jeerakathil T, Shuaib A, Khan K, Kate M, et al. Protocol for LASER: a randomized evaluation and an associated registry of early anticoagulation with edoxaban after ischemic stroke in patients with atrial fibrillation. Front Neurol. 2021; 12:645822.54. Georgi B, Mielke J, Chaffin M, Khera AV, Gelis L, Mundl H, et al. Leveraging human genetics to estimate clinical risk reductions achievable by inhibiting factor XI. Stroke. 2019; 50:3004–3012.55. Shoamanesh A, Mundl H, Smith EE, Masjuan J, Milanov I, Hirano T, et al. Factor XIa inhibition with asundexian after acute non-cardioembolic ischaemic stroke (PACIFIC-stroke): an international, randomised, double-blind, placebo-controlled, phase 2b trial. Lancet. 2022; 400:997–1007.56. Sharma M, Molina CA, Toyoda K, Bereczki D, Bangdiwala SI, Kasner SE, et al. Safety and efficacy of factor XIa inhibition with milvexian for secondary stroke prevention (AXIOMATIC-SSP): a phase 2, international, randomised, double-blind, placebo-controlled, dose-finding trial. Lancet Neurol. 2024; 23:46–59.57. Bayer. OCEANIC-AF study stopped early due to lack of efficacy [Internet]. Leverkusen: Bayer; 2023 November 19 [accessed November 29, 2023]. Available from: https://www.bayer.com/media/en-us/oceanic-af-study-stopped-early-due-to-lack-of-efficacy/.58. Meinel TR, Kniepert JU, Seiffge DJ, Gralla J, Jung S, Auer E, et al. Endovascular stroke treatment and risk of intracranial hemorrhage in anticoagulated patients. Stroke. 2020; 51:892–898.59. Ramos-Araque ME, Chavarría-Miranda A, Gómez-Vicente B, López-Cancio Martínez E, Castañón Apilánez M, Castellanos M, et al. Oral anticoagulation and risk of symptomatic hemorrhagic transformation in stroke patients treated with mechanical thrombectomy: data from the nordictus registry. Front Neurol. 2020; 11:594251.60. Küpper C, Feil K, Wollenweber FA, Tiedt S, Herzberg M, Dorn F, et al. Endovascular stroke treatment in orally anticoagulated patients: an analysis from the German stroke registry-endovascular treatment. J Neurol. 2021; 268:1762–1769.61. Turc G, Bhogal P, Fischer U, Khatri P, Lobotesis K, Mazighi M, et al. European Stroke Organisation (ESO) - European Society for Minimally Invasive Neurological Therapy (ESMINT) guidelines on mechanical thrombectomy in acute ischaemic strokeendorsed by Stroke Alliance for Europe (SAFE). Eur Stroke J. 2019; 4:6–12.62. van de Graaf RA, Chalos V, van Es ACGM, Emmer BJ, Lycklama À Nijeholt GJ, van der Worp HB, et al. Periprocedural intravenous heparin during endovascular treatment for ischemic stroke: results from the MR CLEAN registry. Stroke. 2019; 50:2147–2155.63. van de Graaf RA, Chalos V, Del Zoppo GJ, van der Lugt A, Dippel DWJ, Roozenbeek B. Periprocedural antithrombotic treatment during acute mechanical thrombectomy for ischemic stroke: a systematic review. Front Neurol. 2018; 9:238.64. Paciaroni M, Agnelli G, Micheli S, Caso V. Efficacy and safety of anticoagulant treatment in acute cardioembolic stroke: a meta-analysis of randomized controlled trials. Stroke. 2007; 38:423–430.65. van der Steen W, van de Graaf RA, Chalos V, Lingsma HF, van Doormaal PJ, Coutinho JM, et al. Safety and efficacy of aspirin, unfractionated heparin, both, or neither during endovascular stroke treatment (MR CLEAN-MED): an open-label, multicentre, randomised controlled trial. Lancet. 2022; 399:1059–1069.66. Altavilla R, Caso V, Bandini F, Agnelli G, Tsivgoulis G, Yaghi S, et al. Anticoagulation after stroke in patients with atrial fibrillation. Stroke. 2019; 50:2093–2100.67. Douketis JD, Spyropoulos AC, Kaatz S, Becker RC, Caprini JA, Dunn AS, et al. Perioperative bridging anticoagulation in patients with atrial fibrillation. N Engl J Med. 2015; 373:823–833.68. Kovacs MJ, Wells PS, Anderson DR, Lazo-Langner A, Kearon C, Bates SM, et al. Postoperative low molecular weight heparin bridging treatment for patients at high risk of arterial thromboembolism (PERIOP2): double blind randomised controlled trial. BMJ. 2021; 373:n1205.69. Yang M, Huo X, Gao F, Wang A, Ma N, Liebeskind DS, et al. Safety and efficacy of heparinization during mechanical thrombectomy in acute ischemic stroke. Front Neurol. 2019; 10:299.70. Lee JS, Lee SJ, Hong JM, Alverne FJAM, Lima FO, Nogueira RG. Endovascular treatment of large vessel occlusion strokes due to intracranial atherosclerotic disease. J Stroke. 2022; 24:3–20.71. Liu X, Dai Q, Ye R, Zi W, Liu Y, Wang H, et al. Endovascular treatment versus standard medical treatment for vertebrobasilar artery occlusion (BEST): an open-label, randomised controlled trial. Lancet Neurol. 2020; 19:115–122.72. Zhu X, Cao G. Safety of glycoprotein IIb-IIIa inhibitors used in stroke-related treatment: a systematic review and meta-analysis. Clin Appl Thromb Hemost. 2020; 26:1076029620942594.73. Xu Y, Liu C, Li W, Nie X, Huang S, Li X, et al. Efficacy and safety of early anticoagulation after endovascular treatment in patients with atrial fibrillation. Stroke Vasc Neurol. 2023; 8:405–412.74. Goyal M, Menon BK, van Zwam WH, Dippel DW, Mitchell PJ, Demchuk AM, et al. Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. Lancet. 2016; 387:1723–1731.75. Olthuis SGH, Pirson FAV, Pinckaers FME, Hinsenveld WH, Nieboer D, Ceulemans A, et al. Endovascular treatment versus no endovascular treatment after 6-24 h in patients with ischaemic stroke and collateral flow on CT angiography (MR CLEAN-LATE) in the Netherlands: a multicentre, open-label, blinded-endpoint, randomised, controlled, phase 3 trial. Lancet. 2023; 401:1371–1380.76. Huo X, Ma G, Tong X, Zhang X, Pan Y, Nguyen TN, et al. Trial of endovascular therapy for acute ischemic stroke with large infarct. N Engl J Med. 2023; 388:1272–1283.77. Yoshimura S, Sakai N, Yamagami H, Uchida K, Beppu M, Toyoda K, et al. Endovascular therapy for acute stroke with a large ischemic region. N Engl J Med. 2022; 386:1303–1313.78. Palaiodimou L, Eleftheriou A, Katsanos AH, Safouris A, Magoufis G, Spiliopoulos S, et al. Endovascular treatment for acute basilar artery occlusion: a fragility index meta-analysis. J Clin Med. 2023; 12:2617.79. Sabben C, Charbonneau F, Delvoye F, Strambo D, Heldner MR, Ong E, et al. Endovascular therapy or medical management alone for isolated posterior cerebral artery occlusion: a multicenter study. Stroke. 2023; 54:928–937.80. Berberich A, Finitsis S, Strambo D, Michel P, Herweh C, Meyer L, et al. Endovascular therapy versus no endovascular therapy in patients receiving best medical management for acute isolated occlusion of the posterior cerebral artery: a systematic review and meta-analysis. Eur J Neurol. 2022; 29:2664–2673.81. Nishimoto T, Oka F, Okazaki K, Ishihara H. Timing of direct oral anticoagulants for hemorrhagic transformation after endovascular treatment in acute ischemic stroke. J Stroke Cerebrovasc Dis. 2022; 31:106507.82. Ma H, Che R, Zhang Q, Yu W, Wu L, Zhao W, et al. The optimum anticoagulation time after endovascular thrombectomy for atrial fibrillation-related large vessel occlusion stroke: a real-world study. J Neurol. 2023; 270:2084–2095.83. Hallevi H, Albright KC, Martin-Schild S, Barreto AD, Savitz SI, Escobar MA, et al. Anticoagulation after cardioembolic stroke: to bridge or not to bridge? Arch Neurol. 2008; 65:1169–1173.84. Strbian D, Durukan A, Pitkonen M, Marinkovic I, Tatlisumak E, Pedrono E, et al. The blood-brain barrier is continuously open for several weeks following transient focal cerebral ischemia. Neuroscience. 2008; 153:175–181.85. Jickling GC, Liu D, Stamova B, Ander BP, Zhan X, Lu A, et al. Hemorrhagic transformation after ischemic stroke in animals and humans. J Cereb Blood Flow Metab. 2014; 34:185–199.86. Liu HS, Chung HW, Chou MC, Liou M, Wang CY, Kao HW, et al. Effects of microvascular permeability changes on contrast-enhanced T1 and pharmacokinetic MR imagings after ischemia. Stroke. 2013; 44:1872–1877.87. Elsaid N, Mustafa W, Saied A. Radiological predictors of hemorrhagic transformation after acute ischemic stroke: an evidence-based analysis. Neuroradiol J. 2020; 33:118–133.88. Wilson D, Ambler G, Shakeshaft C, Brown MM, Charidimou A, Al-Shahi Salman R, et al. Cerebral microbleeds and intracranial haemorrhage risk in patients anticoagulated for atrial fibrillation after acute ischaemic stroke or transient ischaemic attack (CROMIS-2): a multicentre observational cohort study. Lancet Neurol. 2018; 17:539–547.89. Martí-Fàbregas J, Medrano-Martorell S, Merino E, Prats-Sánchez L, Marín R, Delgado-Mederos R, et al. MRI predicts intracranial hemorrhage in patients who receive long-term oral anticoagulation. Neurology. 2019; 92:e2432–e2443.90. Seidel G, Cangür H, Albers T, Burgemeister A, Meyer-Wiethe K. Sonographic evaluation of hemorrhagic transformation and arterial recanalization in acute hemispheric ischemic stroke. Stroke. 2009; 40:119–123.91. Bernardo-Castro S, Sousa JA, Brás A, Cecília C, Rodrigues B, Almendra L, et al. Pathophysiology of blood-brain barrier permeability throughout the different stages of ischemic stroke and its implication on hemorrhagic transformation and recovery. Front Neurol. 2020; 11:594672.92. Rust R. Insights into the dual role of angiogenesis following stroke. J Cereb Blood Flow Metab. 2020; 40:1167–1171.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- How well Do We Prevent Cardioembolic Stroke?
- Cardioembolic Stroke in Atrial Fibrillation-Rationale for Preventive Closure of the Left Atrial Appendage
- Etiology and Status of Preventive Therapy of Cardioembolic Stroke: Hospital-Based Retrospective Analysis
- Immediate Anticoagulation for Acute Cardioembolic Stroke is Still Popular in Selective Cases in Korea
- Transient Increase in Intrathoracic Pressure as a Contributing Factor to Cardioembolic Stroke