Nutr Res Pract.
2023 Dec;17(6):1099-1112. 10.4162/nrp.2023.17.6.1099.
Purple perilla frutescens extracts containing α-asarone inhibit inflammatory atheroma formation and promote hepatic HDL cholesterol uptake in dyslipidemic apoE-deficient mice
- Affiliations
-
- 1Department of Food Science and Nutrition and Korean Institute of Nutrition, Hallym University, Chuncheon 24252, Korea
- 2Department of Food and Nutrition, Andong National University, Andong 36729, Korea
- KMID: 2548867
- DOI: http://doi.org/10.4162/nrp.2023.17.6.1099
Abstract
- BACKGROUND/OBJECTIVES
Dyslipidemia causes metabolic disorders such as atherosclerosis and fatty liver syndrome due to abnormally high blood lipids. Purple perilla frutescens extract (PPE) possesses various bioactive compounds such as α-asarone, chlorogenic acid and rosmarinic acid. This study examined whether PPE and α-asarone improved dyslipidemia-associated inflammation and inhibited atheroma formation in apolipoprotein E (apoE)-deficient mice, an experimental animal model of atherosclerosis.
MATERIALS/METHODS
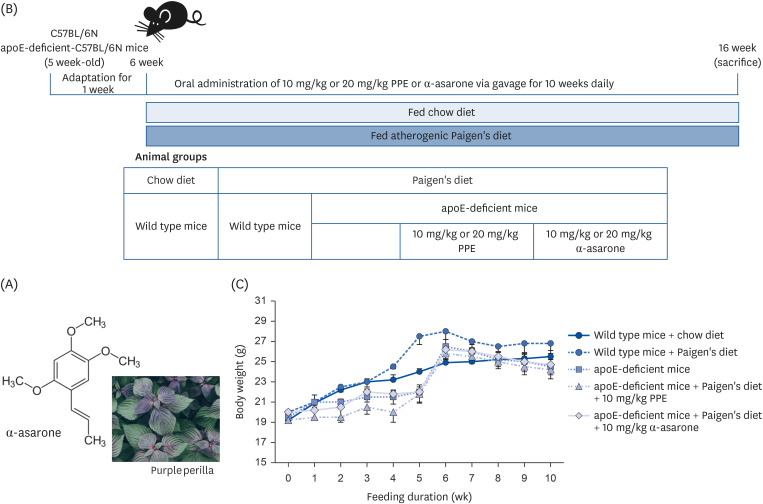
ApoE-deficient mice were fed on high cholesterol-diet (Paigen’s diet) and orally administrated with 10–20 mg/kg PPE and α-asarone for 10 wk.
RESULTS
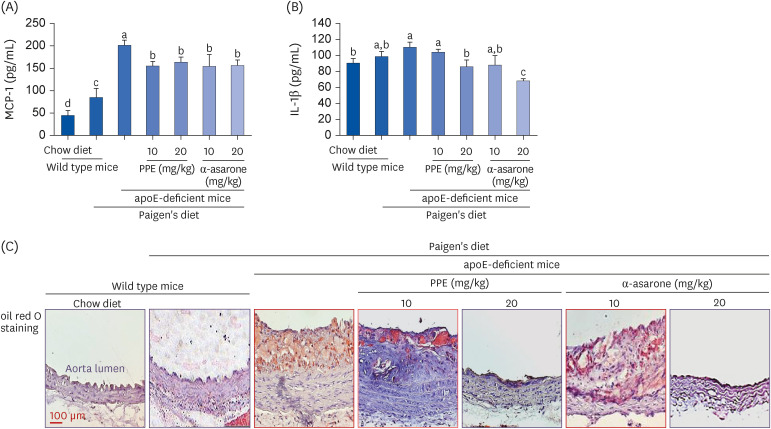
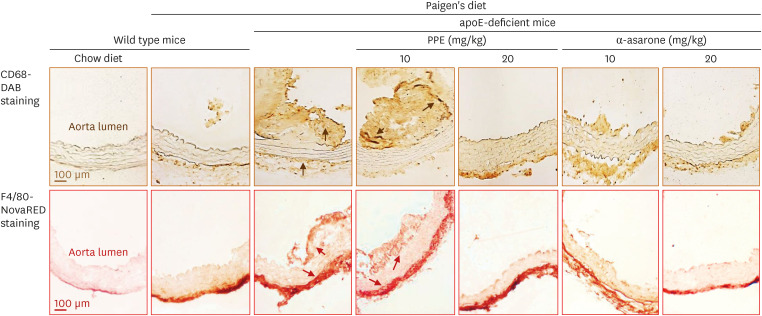
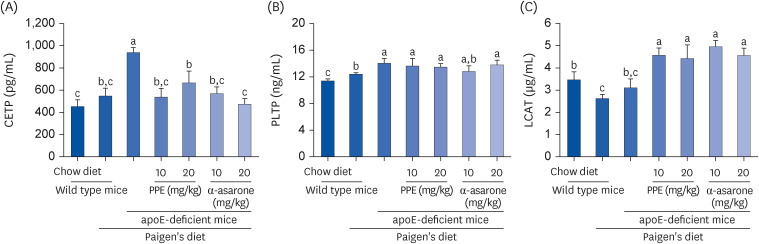
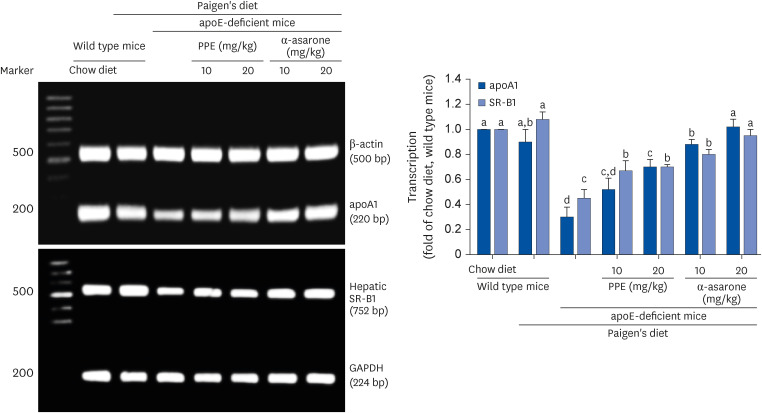
The Paigen’s diet reduced body weight gain in apoE-deficient mice, which was not restored by PPE or α-asarone. PPE or α-asarone improved the plasma lipid profiles in Paigen’s diet-fed apoE-deficient mice, and despite a small increase in high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein (LDL)-cholesterol, and very LDL were significantly reduced. Paigen’s diet-induced systemic inflammation was reduced in PPE or α-asarone-treated apoE-deficient mice. Supplying PPE or α-asarone to mice lacking apoE suppressed aorta atherogenesis induced by atherogenic diet. PPE or α-asarone diminished aorta accumulation of CD68- and/or F4/80-positive macrophages induced by atherogenic diet in apoE-deficient mice. Treatment of apoE-deficient mice with PPE and α-asarone resulted in a significant decrease in plasma cholesteryl ester transfer protein level and an increase in lecithin:cholesterol acyltransferase reduced by supply of Paigen’s diet. Supplementation of PPE and α-asarone enhanced the transcription of hepatic apoA1 and SR-B1 reduced by Paigen’s diet in apoE-deficient mice.
CONCLUSIONS
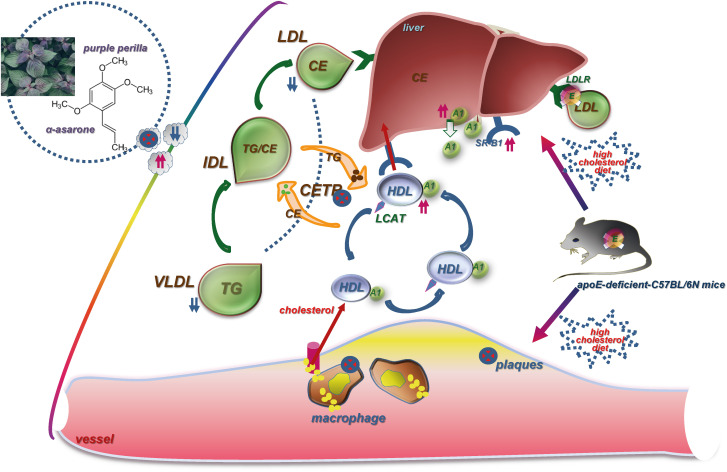
α-Asarone in PPE inhibited inflammation-associated atheroma formation and promoted hepatic HDL-C trafficking in dyslipidemic mice.
Keyword
Figure
Reference
-
1. Musunuru K. Atherogenic dyslipidemia: cardiovascular risk and dietary intervention. Lipids. 2010; 45:907–914. PMID: 20524075.2. Hedayatnia M, Asadi Z, Zare-Feyzabadi R, Yaghooti-Khorasani M, Ghazizadeh H, Ghaffarian-Zirak R, Nosrati-Tirkani A, Mohammadi-Bajgiran M, Rohban M, Sadabadi F, et al. Dyslipidemia and cardiovascular disease risk among the MASHAD study population. Lipids Health Dis. 2020; 19:42. PMID: 32178672.3. Chiu S, Williams PT, Krauss RM. Effects of a very high saturated fat diet on LDL particles in adults with atherogenic dyslipidemia: a randomized controlled trial. PLoS One. 2017; 12:e0170664. PMID: 28166253.4. Pedro-Botet J, Ascaso JF, Blasco M, Brea Á, Díaz Á, Hernández-Mijares A, Pintó X, Millán J. Grupo de trabajo sobre Dislipidemia Aterogénica, Sociedad Española de Arteriosclerosis. Triglycerides, HDL cholesterol and atherogenic dyslipidaemia in the 2019 European guidelines for the management of dyslipidaemias. Clin Investig Arterioscler. 2020; 32:209–218.5. Kones R, Rumana U. Current treatment of dyslipidemia: evolving roles of non-statin and newer drugs. Drugs. 2015; 75:1201–1228. PMID: 26169307.6. Sahebkar A, Watts GF. New LDL-cholesterol lowering therapies: pharmacology, clinical trials, and relevance to acute coronary syndromes. Clin Ther. 2013; 35:1082–1098. PMID: 23932550.7. Mahley RW, Rall SC Jr. Apolipoprotein E: far more than a lipid transport protein. Annu Rev Genomics Hum Genet. 2000; 1:507–537. PMID: 11701639.8. Phillips MC. Apolipoprotein E isoforms and lipoprotein metabolism. IUBMB Life. 2014; 66:616–623. PMID: 25328986.9. Getz GS, Reardon CA. Apoprotein E as a lipid transport and signaling protein in the blood, liver, and artery wall. J Lipid Res. 2009; 50(Suppl):S156–S161. PMID: 19018038.10. Moghadasian MH, McManus BM, Nguyen LB, Shefer S, Nadji M, Godin DV, Green TJ, Hill J, Yang Y, Scudamore CH, et al. Pathophysiology of apolipoprotein E deficiency in mice: relevance to apo E-related disorders in humans. FASEB J. 2001; 15:2623–2630. PMID: 11726538.11. Rosenson RS, Brewer HB Jr, Davidson WS, Fayad ZA, Fuster V, Goldstein J, Hellerstein M, Jiang XC, Phillips MC, Rader DJ, et al. Cholesterol efflux and atheroprotection: advancing the concept of reverse cholesterol transport. Circulation. 2012; 125:1905–1919. PMID: 22508840.12. Rochlani Y, Pothineni NV, Kovelamudi S, Mehta JL. Metabolic syndrome: pathophysiology, management, and modulation by natural compounds. Ther Adv Cardiovasc Dis. 2017; 11:215–225. PMID: 28639538.13. Ji X, Shi S, Liu B, Shan M, Tang D, Zhang W, Zhang Y, Zhang L, Zhang H, Lu C, et al. Bioactive compounds from herbal medicines to manage dyslipidemia. Biomed Pharmacother. 2019; 118:109338. PMID: 31545238.14. Hu Y, Chen X, Hu M, Zhang D, Yuan S, Li P, Feng L. Medicinal and edible plants in the treatment of dyslipidemia: advances and prospects. Chin Med. 2022; 17:113. PMID: 36175900.15. Yu H, Qiu JF, Ma LJ, Hu YJ, Li P, Wan JB. Phytochemical and phytopharmacological review of Perilla frutescens L. (Labiatae), a traditional edible-medicinal herb in China. Food Chem Toxicol. 2017; 108:375–391. PMID: 27890564.16. Liu S, Jin X, Shang Y, Wang L, Du K, Chen S, Li J, He J, Fang S, Chang Y. A comprehensive review of the botany, ethnopharmacology, phytochemistry, pharmacology, toxicity and quality control of Perillae Fructus. J Ethnopharmacol. 2023; 304:116022. PMID: 36481246.17. Park SH, Paek JH, Shin D, Lee JY, Lim SS, Kang YH. Purple perilla extracts with α-asarone enhance cholesterol efflux from oxidized LDL-exposed macrophages. Int J Mol Med. 2015; 35:957–965. PMID: 25673178.18. Park SH, Shin D, Lim SS, Lee JY, Kang YH. Purple perilla extracts allay ER stress in lipid-laden macrophages. PLoS One. 2014; 9:e110581. PMID: 25333946.19. Paek JH, Shin KH, Kang YH, Lee JY, Lim SS. Rapid identification of aldose reductase inhibitory compounds from Perilla frutescens. BioMed Res Int. 2013; 2013:679463. PMID: 24308003.20. Wang Z, Kwon SH, Hwang SH, Kang YH, Lee JY, Lim SS. Competitive binding experiments can reduce the false positive results of affinity-based ultrafiltration-HPLC: a case study for identification of potent xanthine oxidase inhibitors from Perilla frutescens extract. J Chromatogr B Analyt Technol Biomed Life Sci. 2017; 1048:30–37.21. Rodríguez-Páez L, Juárez-Sanchez M, Antúnez-Solís J, Baeza I, Wong C. Alpha-asarone inhibits HMG-CoA reductase, lowers serum LDL-cholesterol levels and reduces biliary CSI in hypercholesterolemic rats. Phytomedicine. 2003; 10:397–404. PMID: 12834005.22. Hwang SH, Kwon SH, Kang YH, Lee JY, Lim SS. Rapid high performance liquid chromatography determination and optimization of extraction parameters of the α-asarone isolated from Perilla frutescens L. Molecules. 2017; 22:270. PMID: 28208640.23. Nakashima Y, Plump AS, Raines EW, Breslow JL, Ross R. ApoE-deficient mice develop lesions of all phases of atherosclerosis throughout the arterial tree. Arterioscler Thromb. 1994; 14:133–140. PMID: 8274468.24. Lloyd CM, Phillips ARJ, Cooper GJS, Dunbar PR. Three-colour fluorescence immunohistochemistry reveals the diversity of cells staining for macrophage markers in murine spleen and liver. J Immunol Methods. 2008; 334:70–81. PMID: 18367204.25. Lo Sasso G, Schlage WK, Boué S, Veljkovic E, Peitsch MC, Hoeng J. The Apoe(-/-) mouse model: a suitable model to study cardiovascular and respiratory diseases in the context of cigarette smoke exposure and harm reduction. J Transl Med. 2016; 14:146. PMID: 27207171.26. De Giorgi M, Li A, Hurley A, Barzi M, Doerfler AM, Cherayil NA, Smith HE, Brown JD, Lin CY, Bissig KD, et al. Targeting the Apoa1 locus for liver-directed gene therapy. Mol Ther Methods Clin Dev. 2021; 21:656–669. PMID: 34141821.27. Ganesan LP, Mates JM, Cheplowitz AM, Avila CL, Zimmerer JM, Yao Z, Maiseyeu A, Rajaram MV, Robinson JM, Anderson CL. Scavenger receptor B1, the HDL receptor, is expressed abundantly in liver sinusoidal endothelial cells. Sci Rep. 2016; 6:20646. PMID: 26865459.28. Leiva A, Verdejo H, Benítez ML, Martínez A, Busso D, Rigotti A. Mechanisms regulating hepatic SR-BI expression and their impact on HDL metabolism. Atherosclerosis. 2011; 217:299–307. PMID: 21741044.29. Mahley RW. Apolipoprotein E: from cardiovascular disease to neurodegenerative disorders. J Mol Med (Berl). 2016; 94:739–746. PMID: 27277824.30. Chowaniec Z, Skoczyńska A. Plasma lipid transfer proteins: the role of PLTP and CETP in atherogenesis. Adv Clin Exp Med. 2018; 27:429–436. PMID: 29558025.31. Manthei KA, Patra D, Wilson CJ, Fawaz MV, Piersimoni L, Shenkar JC, Yuan W, Andrews PC, Engen JR, Schwendeman A, et al. Structural analysis of lecithin:cholesterol acyltransferase bound to high density lipoprotein particles. Commun Biol. 2020; 3:28. PMID: 31942029.32. Rong S, Zhao S, Kai X, Zhang L, Zhao Y, Xiao X, Bao W, Liu L. Procyanidins extracted from the litchi pericarp attenuate atherosclerosis and hyperlipidemia associated with consumption of a high fat diet in apolipoprotein-E knockout mice. Biomed Pharmacother. 2018; 97:1639–1644. PMID: 29793326.33. Li J, Zhong Z, Yuan J, Chen X, Huang Z, Wu Z. Resveratrol improves endothelial dysfunction and attenuates atherogenesis in apolipoprotein E-deficient mice. J Nutr Biochem. 2019; 67:63–71. PMID: 30856465.34. Wu C, Luan H, Zhang X, Wang S, Zhang X, Sun X, Guo P. Chlorogenic acid protects against atherosclerosis in ApoE-/- mice and promotes cholesterol efflux from RAW264.7 macrophages. PLoS One. 2014; 9:e95452. PMID: 25187964.35. Park SH, Kang MK, Choi YJ, Kim YH, Antika LD, Kim DY, Lee EJ, Lim SS, Kang YH. α-Asarone blocks 7β-hydroxycholesterol-exposed macrophage injury through blocking elF2α phosphorylation and prompting beclin-1-dependent autophagy. Oncotarget. 2017; 8:7370–7383. PMID: 28088783.36. Park SH, Kang MK, Choi YJ, Kim YH, Antika LD, Lim SS, Kang YH. Dietary compound α-asarone alleviates ER stress-mediated apoptosis in 7β-hydroxycholesterol-challenged macrophages. Mol Nutr Food Res. 2016; 60:1033–1047. PMID: 26893256.37. Farrell N, Norris G, Lee SG, Chun OK, Blesso CN. Anthocyanin-rich black elderberry extract improves markers of HDL function and reduces aortic cholesterol in hyperlipidemic mice. Food Funct. 2015; 6:1278–1287. PMID: 25758596.38. Xu M, Lv C, Wang H, Lu Q, Ye M, Zhu X, Liu R. Peanut skin extract ameliorates high-fat diet-induced atherosclerosis by regulating lipid metabolism, inflammation reaction and gut microbiota in ApoE-/- mice. Food Res Int. 2022; 154:111014. PMID: 35337573.39. Olszanecki R, Jawień J, Gajda M, Mateuszuk L, Gebska A, Korabiowska M, Chłopicki S, Korbut R. Effect of curcumin on atherosclerosis in apoE/LDLR-double knockout mice. J Physiol Pharmacol. 2005; 56:627–635. PMID: 16391419.40. Huang W, Zeng Z, Lang Y, Xiang X, Qi G, Lu G, Yang X. Cannabis seed oil alleviates experimental atherosclerosis by ameliorating vascular inflammation in apolipoprotein-E-deficient mice. J Agric Food Chem. 2021; 69:9102–9110. PMID: 34037390.41. Peng Y, Xu J, Zeng Y, Chen L, Xu XL. Polydatin attenuates atherosclerosis in apolipoprotein E-deficient mice: role of reverse cholesterol transport. Phytomedicine. 2019; 62:152935. PMID: 31085374.42. Millar CL, Duclos Q, Blesso CN. Effects of dietary flavonoids on reverse cholesterol transport, HDL metabolism, and HDL function. Adv Nutr. 2017; 8:226–239. PMID: 28298268.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Anti-Helicobacter pylori activity of acomplex mixture of Lactobacillus paracasei HP7 including the extract of Perilla frutescens var. acuta and Glycyrrhiza glabra
- Inhibition of Proinflammatory Cytokine Generation in Lung Inflammation by the Leaves of Perilla frutescens and Its Constituents
- Dietary ellagic acid blocks inflammation-associated atherosclerotic plaque formation in cholesterol-fed apoE-deficient mice
- Ellagic acid, a functional food component, ameliorates functionality of reverse cholesterol transport in murine model of atherosclerosis
- Synergistic effect of probiotic HP7 and the extracts of Perilla frutescens and Glycyrrhiza glabra to alleviate Helicobacter pylori-induced damages