Restor Dent Endod.
2023 May;48(2):e20. 10.5395/rde.2023.48.e20.
Stem cell-derived exosomes for dentin-pulp complex regeneration: a mini-review
- Affiliations
-
- 1Department of Oral Biology, Faculty of Dentistry, Mansoura University, Mansoura, Egypt
- 2Department of Oral Biology, Faculty of Dentistry, Menoufia University, Shibin el Kom, Egypt
- 3Department of Oral Biology, Faculty of Oral and Dental Medicine, Delta University for Science and Technology, Dakahlia, Egypt
- KMID: 2548223
- DOI: http://doi.org/10.5395/rde.2023.48.e20
Abstract
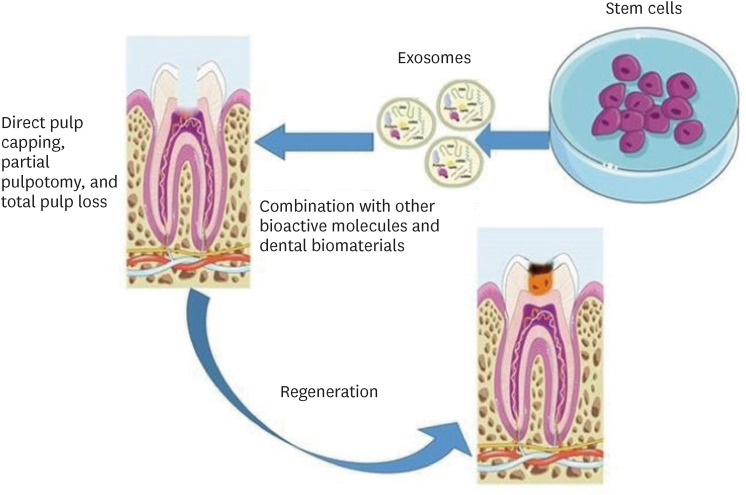
- This mini-review was conducted to present an overview of the use of exosomes in regenerating the dentin-pulp complex (DPC). The PubMed and Scopus databases were searched for relevant articles published between January 1, 2013 and January 1, 2023. The findings of basic in vitro studies indicated that exosomes enhance the proliferation and migration of mesenchymal cells, as human dental pulp stem cells, via mitogenactivated protein kinases and Wingless-Int signaling pathways. In addition, they possess proangiogenic potential and contribute to neovascularization and capillary tube formation by promoting endothelial cell proliferation and migration of human umbilical vein endothelial cells. Likewise, they regulate the migration and differentiation of Schwann cells, facilitate the conversion of M1 pro-inflammatory macrophages to M2 anti-inflammatory phenotypes, and mediate immune suppression as they promote regulatory T cell conversion. Basic in vivo studies have indicated that exosomes triggered the regeneration of dentin-pulp–like tissue, and exosomes isolated under odontogenic circumstances are particularly strong inducers of tissue regeneration and stem cell differentiation. Exosomes are a promising regenerative tool for DPC in cases of small pulp exposure or for whole-pulp tissue regeneration.
Figure
Reference
-
1. Yu C, Abbott PV. An overview of the dental pulp: its functions and responses to injury. Aust Dent J. 2007; 52(Supplement):S4–SS16.
Article2. Goldberg M, Kulkarni AB, Young M, Boskey A. Dentin: structure, composition and mineralization. Front Biosci (Elite Ed). 2011; 3:711–735. PMID: 21196346.
Article3. Cordeiro MM, Dong Z, Kaneko T, Zhang Z, Miyazawa M, Shi S, Smith AJ, Nör JE. Dental pulp tissue engineering with stem cells from exfoliated deciduous teeth. J Endod. 2008; 34:962–969. PMID: 18634928.
Article4. Duncan HF, Yamauchi Y. Pulp exposure management. Clin Dent Rev. 2019; 3:4.
Article5. Swanson WB, Gong T, Zhang Z, Eberle M, Niemann D, Dong R, Rambhia KJ, Ma PX. Controlled release of odontogenic exosomes from a biodegradable vehicle mediates dentinogenesis as a novel biomimetic pulp capping therapy. J Control Release. 2020; 324:679–694. PMID: 32534011.
Article6. Huang CC, Narayanan R, Alapati S, Ravindran S. Exosomes as biomimetic tools for stem cell differentiation: applications in dental pulp tissue regeneration. Biomaterials. 2016; 111:103–115. PMID: 27728810.
Article7. Li Z, Liu L, Wang L, Song D. The effects and potential applications of concentrated growth factor in dentin-pulp complex regeneration. Stem Cell Res Ther. 2021; 12:357. PMID: 34147130.
Article8. Simon SR, Berdal A, Cooper PR, Lumley PJ, Tomson PL, Smith AJ. Dentin-pulp complex regeneration: from lab to clinic. Adv Dent Res. 2011; 23:340–345. PMID: 21677089.9. Asgary S, Parirokh M, Eghbal MJ, Ghoddusi J. SEM evaluation of pulp reaction to different pulp capping materials in dog’s teeth. Iran Endod J. 2006; 1:117–123. PMID: 24379876.10. Aljandan B, AlHassan H, Saghah A, Rasheed M, Ali AA. The effectiveness of using different pulp-capping agents on the healing response of the pulp. Indian J Dent Res. 2012; 23:633–637. PMID: 23422610.
Article11. Boland GM, Perkins G, Hall DJ, Tuan RS. Wnt 3a promotes proliferation and suppresses osteogenic differentiation of adult human mesenchymal stem cells. J Cell Biochem. 2004; 93:1210–1230. PMID: 15486964.
Article12. Giuroiu CL, Căruntu ID, Lozneanu L, Melian A, Vataman M, Andrian S. Dental pulp: correspondences and contradictions between clinical and histological diagnosis. BioMed Res Int. 2015; 2015:960321. PMID: 26078972.
Article13. Imura N, Pinheiro ET, Gomes BP, Zaia AA, Ferraz CC, Souza-Filho FJ. The outcome of endodontic treatment: a retrospective study of 2000 cases performed by a specialist. J Endod. 2007; 33:1278–1282. PMID: 17963946.
Article14. Cvek M. Prognosis of luxated non-vital maxillary incisors treated with calcium hydroxide and filled with gutta-percha. A retrospective clinical study. Endod Dent Traumatol. 1992; 8:45–55. PMID: 1521505.
Article15. Kim JY, Xin X, Moioli EK, Chung J, Lee CH, Chen M, Fu SY, Koch PD, Mao JJ. Regeneration of dental-pulp-like tissue by chemotaxis-induced cell homing. Tissue Eng Part A. 2010; 16:3023–3031. PMID: 20486799.
Article16. Garcia-Godoy F, Murray PE. Recommendations for using regenerative endodontic procedures in permanent immature traumatized teeth. Dent Traumatol. 2012; 28:33–41. PMID: 21794081.
Article17. Palma PJ, Ramos JC, Martins JB, Diogenes A, Figueiredo MH, Ferreira P, Viegas C, Santos JM. Histologic evaluation of regenerative endodontic procedures with the use of chitosan scaffolds in immature dog teeth with apical periodontitis. J Endod. 2017; 43:1279–1287. PMID: 28577961.
Article18. Palma PJ, Martins J, Diogo P, Sequeira D, Ramos JC, Diogenes A, Sequeira D, Ramos JC, Diogenes A, Santos JM. Does apical papilla survive and develop in apical periodontitis presence after regenerative endodontic procedures? Appl Sci. 2019; 9:3942–3955.
Article19. Chrepa V, Pitcher B, Henry MA, Diogenes A. Survival of the apical papilla and its resident stem cells in a case of advanced pulpal necrosis and apical periodontitis. J Endod. 2017; 43:561–567. PMID: 28190588.
Article20. Smith AJ, Cassidy N, Perry H, Bègue-Kirn C, Ruch JV, Lesot H. Reactionary dentinogenesis. Int J Dev Biol. 1995; 39:273–280. PMID: 7626417.21. Simon S, Smith AJ, Berdal A, Lumley PJ, Cooper PR. The MAP kinase pathway is involved in odontoblast stimulation via p38 phosphorylation. J Endod. 2010; 36:256–259. PMID: 20113784.
Article22. Bai Y, Cheng X, Liu X, Guo Q, Wang Z, Fu Y, He W, Yu Q. Transforming growth factor-β1 promotes early odontoblastic differentiation of dental pulp stem cells via activating AKT, Erk1/2 and p38 MAPK pathways. J Dent Sci. 2023; 18:87–94. PMID: 36643229.
Article23. Guo W, Fan Z, Wang S, Du J. ALK5 is essential for tooth germ differentiation during tooth development. Biotech Histochem. 2019; 94:481–490. PMID: 31144525.
Article24. Nakashima M, Iohara K. Mobilized dental pulp stem cells for pulp regeneration: initiation of clinical trial. J Endod. 2014; 40(Supplement):S26–S32. PMID: 24698690.
Article25. Nakashima M, Iohara K, Murakami M, Nakamura H, Sato Y, Ariji Y, Matsushita K. Pulp regeneration by transplantation of dental pulp stem cells in pulpitis: a pilot clinical study. Stem Cell Res Ther. 2017; 8:61. PMID: 28279187.
Article26. Mitsiadis TA, Orsini G, Jimenez-Rojo L. Stem cell-based approaches in dentistry. Eur Cell Mater. 2015; 30:248–257. PMID: 26562631.
Article27. Nassar W, El-Ansary M, Sabry D, Mostafa MA, Fayad T, Kotb E, Temraz M, Saad AN, Essa W, Adel H. Umbilical cord mesenchymal stem cells derived extracellular vesicles can safely ameliorate the progression of chronic kidney diseases. Biomater Res. 2016; 20:21. PMID: 27499886.
Article28. Ivica A, Ghayor C, Zehnder M, Valdec S, Weber FE. Pulp-derived exosomes in a fibrin-based regenerative root filling material. J Clin Med. 2020; 9:491. PMID: 32054086.
Article29. Cooper LF, Ravindran S, Huang CC, Kang M. A role for exosomes in craniofacial tissue engineering and regeneration. Front Physiol. 2020; 10:1569. PMID: 32009978.
Article30. Xian X, Gong Q, Li C, Guo B, Jiang H. Exosomes with highly angiogenic potential for possible use in pulp regeneration. J Endod. 2018; 44:751–758. PMID: 29426641.
Article31. Hu X, Zhong Y, Kong Y, Chen Y, Feng J, Zheng J. Lineage-specific exosomes promote the odontogenic differentiation of human dental pulp stem cells (DPSCs) through TGFβ1/smads signaling pathway via transfer of microRNAs. Stem Cell Res Ther. 2019; 10:170. PMID: 31196201.
Article32. Zhang S, Thiebes AL, Kreimendahl F, Ruetten S, Buhl EM, Wolf M, Jockenhoevel S, Apel C. Extracellular vesicles-loaded fibrin gel supports rapid neovascularization for dental pulp regeneration. Int J Mol Sci. 2020; 21:4226. PMID: 32545804.
Article33. Zhang S, Yang Y, Jia S, Chen H, Duan Y, Li X, Wang S, Wang T, Lyu Y, Chen G, Tian W. Exosome-like vesicles derived from Hertwig’s epithelial root sheath cells promote the regeneration of dentin-pulp tissue. Theranostics. 2020; 10:5914–5931. PMID: 32483427.
Article34. Zhuang X, Ji L, Jiang H, Liu Y, Liu X, Bi J, Zhao W, Ding Z, Chen X. Exosomes derived from stem cells from the apical papilla promote dentine-pulp complex regeneration by inducing specific dentinogenesis. Stem Cells Int. 2020; 2020:5816723. PMID: 32565828.
Article35. Huang X, Qiu W, Pan Y, Li J, Chen Z, Zhang K, Luo Y, Wu B, Xu W. Exosomes from LPS-stimulated hDPSCs activated the angiogenic potential of HUVECs in vitro. Stem Cells Int. 2021; 2021:6685307. PMID: 33936213.36. Chen WJ, Xie J, Lin X, Ou MH, Zhou J, Wei XL, Chen WX. The role of small extracellular vesicles derived from lipopolysaccharide-preconditioned human dental pulp stem cells in dental pulp regeneration. J Endod. 2021; 47:961–969. PMID: 33775732.
Article37. Wu M, Liu X, Li Z, Huang X, Guo H, Guo X, Yang X, Li B, Xuan K, Jin Y. SHED aggregate exosomes shuttled miR-26a promote angiogenesis in pulp regeneration via TGF-β/SMAD2/3 signalling. Cell Prolif. 2021; 54:e13074. PMID: 34101281.
Article38. Li J, Ju Y, Liu S, Fu Y, Zhao S. Exosomes derived from lipopolysaccharide-preconditioned human dental pulp stem cells regulate Schwann cell migration and differentiation. Connect Tissue Res. 2021; 62:277–286. PMID: 31769319.
Article39. Zeng J, He K, Mai R, Lin T, Wei R, Nong J, Wu Y. Exosomes from human umbilical cord mesenchymal stem cells and human dental pulp stem cells ameliorate lipopolysaccharide-induced inflammation in human dental pulp stem cells. Arch Oral Biol. 2022; 138:105411. PMID: 35338830.
Article40. Yu S, Chen X, Liu Y, Zhuang XY, Wang AC, Liu XM, Zhu S. Exosomes derived from stem cells from the apical papilla alleviate inflammation in rat pulpitis by upregulating regulatory T cells. Int Endod J. 2022; 55:517–530. PMID: 35274316.
Article41. Stanko P, Altanerova U, Jakubechova J, Repiska V, Altaner C. Dental mesenchymal stem/stromal cells and their exosomes. Stem Cells Int. 2018; 2018:8973613. PMID: 29760738.
Article42. Théry C, Zitvogel L, Amigorena S. Exosomes: composition, biogenesis and function. Nat Rev Immunol. 2002; 2:569–579. PMID: 12154376.
Article43. Février B, Raposo G. Exosomes: endosomal-derived vesicles shipping extracellular messages. Curr Opin Cell Biol. 2004; 16:415–421. PMID: 15261674.
Article44. Qian X, An N, Ren Y, Yang C, Zhang X, Li L. Immunosuppressive effects of mesenchymal stem cells-derived exosomes. Stem Cell Rev Rep. 2021; 17:411–427. PMID: 32935222.
Article45. Zarà M, Amadio P, Campodonico J, Sandrini L, Barbieri SS. Exosomes in cardiovascular diseases. Diagnostics (Basel). 2020; 10:943. PMID: 33198302.
Article46. Guo SC, Tao SC, Yin WJ, Qi X, Yuan T, Zhang CQ. Exosomes derived from platelet-rich plasma promote the re-epithelization of chronic cutaneous wounds via activation of YAP in a diabetic rat model. Theranostics. 2017; 7:81–96. PMID: 28042318.
Article47. Brunello G, Zanotti F, Trentini M, Zanolla I, Pishavar E, Favero V, Favero R, Favero L, Bressan E, Bonora M, Sivolella S, Zavan B. Exosomes derived from dental pulp stem cells show different angiogenic and osteogenic properties in relation to the age of the donor. Pharmaceutics. 2022; 14:908. PMID: 35631496.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dental Pulp Stem Cells and Current in vivo Approaches to Study Dental Pulp Stem Cells in Pulp Injury and Regeneration
- Advances in Research on Stem Cell-Based Pulp Regeneration
- Mesenchymal Stem Cell-Derived Exosomes: A Promising Therapeutic Ace Card to Address Autoimmune Diseases
- Development of a mouse model for pulp-dentin complex regeneration research: a preliminary study
- Induced Pluripotent Stem (iPS) Cells in Dentistry: A Review