Restor Dent Endod.
2023 Feb;48(1):e2. 10.5395/rde.2023.48.e2.
Antimicrobial and cytotoxic properties of calcium-enriched mixture cement, Iranian propolis, and propolis with herbal extracts in primary dental pulp stem cells
- Affiliations
-
- 1Department of Pediatrics, Faculty of Dentistry, Shahid Beheshti University of Medical Sciences, Tehran, Iran
- 2Medicinal Plants and Natural Products Research Center, Department of Pharmacognosy and Pharmaceutical Biotechnology, School of Pharmacy, Hamadan University of Medical Sciences, Hamadan, Iran
- 3Department of Pediatrics, Faculty of Dentistry, Hamadan University of Medical Sciences, Hamadan, Iran
- 4Department of Microbiology, School of Medicine, Hamadan University of Medical Sciences, Hamadan, Iran
- 5Endometrium and Endometriosis Research Center, Department of Anatomy, School of Medicine, Hamadan University of Medical Sciences, Hamadan, Iran
- 6Department of Biostatistics, School of Public Health and Research Center for Health Sciences, Hamadan University of Medical Sciences, Hamadan, Iran
- KMID: 2548170
- DOI: http://doi.org/10.5395/rde.2023.48.e2
Abstract
Objectives
In this study, natural substances were introduced as primary dental pulp caps for use in pulp therapy, and the antimicrobial and cytotoxic properties of these substances were investigated.
Materials and Methods
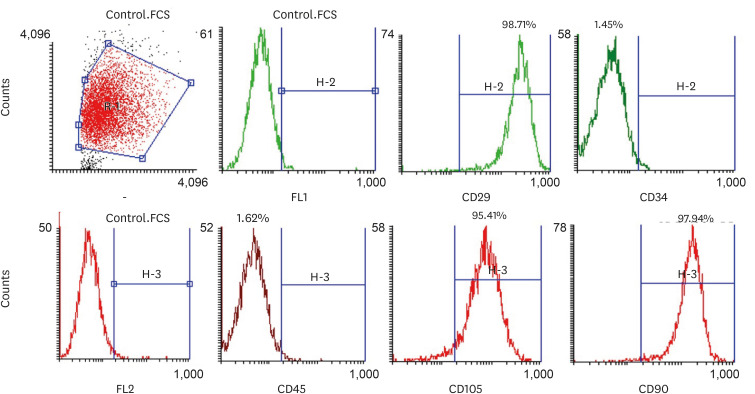
In this in vitro study, the antimicrobial properties of calciumenriched mixture (CEM) cement, propolis, and propolis individually combined with the extracts of several medicinal plants were investigated against Enterococcus faecalis, Escherichia coli, Pseudomonas aeruginosa, and Staphylococcus aureus. Then, the cytotoxicity of each substance or mixture against pulp stem cells extracted from 30 primary healthy teeth was evaluated at 4 concentrations. Data were gathered via observation, and optical density values were obtained using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) test and recorded. SPSS software version 23 was used to analyze the data. Data were evaluated using 2-way analysis of variance and the Tukey test.
Results
Regarding antimicrobial properties, thyme alone and thyme + propolis had the lowest minimum inhibitory concentrations (MICs) against the growth of S. aureus, E. coli, and P. aeruginosa bacteria. For E. faecalis, thyme + propolis had the lowest MIC, followed by thyme alone. At 24 and 72 hours, thyme + propolis, CEM cement, and propolis had the greatest bioviability in the primary dental pulp stem cells, and lavender + propolis had the lowest bioviability.
Conclusions
Of the studied materials, thyme + propolis showed the best results in the measures of practical performance as a dental pulp cap.
Keyword
Figure
Reference
-
1. McTigue DJ, Fields HW, Pinkham JR, Casamassimo PS. Pediatric dentistry: infancy through adolescence. New Delhi: Elsevier India;2013.2. de Albuquerque DS, Gominho LF, Dos Santos RA. Albuquerque DSd. Histologic evaluation of pulpotomy performed with ethyl-cyanoacrylate and calcium hydroxide. Braz Oral Res. 2006; 20:226–230. PMID: 17119705.
Article3. Cengiz SB, Batirbaygil Y, Onur MA, Atilla P, Asan E, Altay N, Cehreli ZC. Histological comparison of alendronate, calcium hydroxide and formocresol in amputated rat molar. Dent Traumatol. 2005; 21:281–288. PMID: 16149924.
Article4. Ozório JE, Carvalho LF, de Oliveira DA, de Sousa-Neto MD, Perez DE. Standardized propolis extract and calcium hydroxide as pulpotomy agents in primary pig teeth. J Dent Child (Chic). 2012; 79:53–58. PMID: 22828758.5. Shayegan A, Petein M, Abbeele AV. Beta-tricalcium phosphate, white mineral trioxide aggregate, white Portland cement, ferric sulfate, and formocresol used as pulpotomy agents in primary pig teeth. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008; 105:536–542. PMID: 18329589.
Article6. Sonmez D, Sari S, Cetinbaş T. A comparison of four pulpotomy techniques in primary molars: a long-term follow-up. J Endod. 2008; 34:950–955. PMID: 18634926.
Article7. Malekafzali B, Shekarchi F, Asgary S. Treatment outcomes of pulpotomy in primary molars using two endodontic biomaterials. A 2-year randomised clinical trial. Eur J Paediatr Dent. 2011; 12:189–193. PMID: 22077689.8. Mohammadi Z, Dummer PM. Properties and applications of calcium hydroxide in endodontics and dental traumatology. Int Endod J. 2011; 44:697–730. PMID: 21535021.
Article9. Ahangari Z, Naseri M, Jalili M, Mansouri Y, Mashhadiabbas F, Torkaman A. Effect of propolis on dentin regeneration and the potential role of dental pulp stem cell in Guinea pigs. Cell J. 2012; 13:223–228. PMID: 23508294.10. Noorollahian H, Ebrahimi M, Javidi Dashtbiaz M, Mir F. Comparison of Iranian propolis and formocresol in pulpotomized primary molars: a preliminary study. J Mashhad Dent Sch. 2014; 38:267–274.11. Park YK, Ikegaki M. Preparation of water and ethanolic extracts of propolis and evaluation of the preparations. Biosci Biotechnol Biochem. 1998; 62:2230–2232. PMID: 27393593.
Article12. Nadia Z, Rachid M. Antioxidant and antibacterial activities of Thymus vulgaris L. Med Aromat Plant Res J. 2013; 1:5–11.13. Sarikurkcu C, Zengin G, Oskay M, Uysal S, Ceylan R, Aktumsek A. Composition, antioxidant, antimicrobial and enzyme inhibition activities of two Origanum vulgare subspecies (subsp. vulgare and subsp. hirtum) essential oils. Ind Crops Prod. 2015; 70:178–184.
Article14. Amanlou M, Dadkhah F, Salehnia A, Farsam H, Dehpour AR. An anti-inflammatory and anti-nociceptive effects of hydroalcoholic extract of Satureja khuzistanica Jamzad extract. J Pharm Pharm Sci. 2005; 8:102–106. PMID: 15946603.15. Kuo CL, Chi CW, Liu TY. The anti-inflammatory potential of berberine in vitro and in vivo . Cancer Lett. 2004; 203:127–137. PMID: 14732220.16. Liu CS, Zheng YR, Zhang YF, Long XY. Research progress on berberine with a special focus on its oral bioavailability. Fitoterapia. 2016; 109:274–282. PMID: 26851175.
Article17. Singh M, Srivastava S, Rawat AK. Antimicrobial activities of Indian Berberis species. Fitoterapia. 2007; 78:574–576. PMID: 17583443.
Article18. Ait Said L, Zahlane K, Ghalbane I, El Messoussi S, Romane A, Cavaleiro C, Salgueiro L. Chemical composition and antibacterial activity of Lavandula coronopifolia essential oil against antibiotic-resistant bacteria. Nat Prod Res. 2015; 29:582–585. PMID: 25174508.
Article19. Peana AT, D’Aquila PS, Chessa ML, Moretti MD, Serra G, Pippia P. (-)-Linalool produces antinociception in two experimental models of pain. Eur J Pharmacol. 2003; 460:37–41. PMID: 12535857.
Article20. Dahl JE, Frangou-Polyzois MJ, Polyzois GL. In vitro biocompatibility of denture relining materials. Gerodontology. 2006; 23:17–22. PMID: 16433637.
Article21. Sforcin JM, Bankova V. Propolis: is there a potential for the development of new drugs? J Ethnopharmacol. 2011; 133:253–260. PMID: 20970490.
Article22. Seibert JB, Bautista-Silva JP, Amparo TR, Petit A, Pervier P, Dos Santos Almeida JC, Azevedo MC, Silveira BM, Brandão GC, de Souza GH, de Medeiros Teixeira LF, Dos Santos OD. Development of propolis nanoemulsion with antioxidant and antimicrobial activity for use as a potential natural preservative. Food Chem. 2019; 287:61–67. PMID: 30857719.
Article23. Stuart CH, Schwartz SA, Beeson TJ, Owatz CB. Enterococcus faecalis: its role in root canal treatment failure and current concepts in retreatment. J Endod. 2006; 32:93–98. PMID: 16427453.
Article24. Zare Jahromi M, Toubayani H, Rezaei M. Propolis: a new alternative for root canal disinfection. Iran Endod J. 2012; 7:127–133. PMID: 23056131.25. Madhubala MM, Srinivasan N, Ahamed S. Comparative evaluation of propolis and triantibiotic mixture as an intracanal medicament against Enterococcus faecalis . J Endod. 2011; 37:1287–1289. PMID: 21846550.
Article26. do Nascimento TG, Silva AS, Lessa Constant PB, da Silva SA, Fidelis de Moura MA, de Almeida CP, Silva VC, Wanderley AB, Basílio Júnior ID, Escodro PB. Phytochemical screening, antioxidant and antibacterial activities of some commercial extract of propolis. J Apic Res. 2018; 57:246–254.
Article27. Sandoval LVH. In vitro antimicrobial activity of ethanolic propolis extracts from santander against Enterococcus faecalis . EC Dent Sci. 2019; 18:1077–1083.28. Machado MEL, Di Spagna Souza A, de Rezende EC, dos Santos EB. Evaluación in vitro de la actividad antibacteriana del paramonoclorofenol alcanforado, clorhexidina al 2% y extracto de propolis al 50% sobre tres bacterias encontradas en el interior del canal radicular. Rev Soc Chil Endod. 2007; 15:16–19.29. Valera MC, da Rosa JA, Maekawa LE, de Oliveira LD, Carvalho CA, Koga-Ito CY, Jorge AO. Action of propolis and medications against Escherichia coli and endotoxin in root canals. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010; 110:e70–e74. PMID: 20868987.30. Libério SA, Pereira AL, Araújo MJ, Dutra RP, Nascimento FR, Monteiro-Neto V, Ribeiro MN, Gonçalves AG, Guerra RN. The potential use of propolis as a cariostatic agent and its actions on mutans group streptococci. J Ethnopharmacol. 2009; 125:1–9. PMID: 19422903.
Article31. Mohammadzadeh S, Shariatpanahi M, Hamedi M, Ahmadkhaniha R, Samadi N, Ostad SN. Chemical composition, oral toxicity and antimicrobial activity of Iranian propolis. Food Chem. 2007; 103:1097–1103.
Article32. de Rezende GPSR, da Costa LRRS, Pimenta FC, Baroni DA. In vitro antimicrobial activity of endodontic pastes with propolis extracts and calcium hydroxide: a preliminary study. Braz Dent J. 2008; 19:301–305. PMID: 19180318.33. Bretz WA, Chiego DJ Jr, Marcucci MC, Cunha I, Custódio A, Schneider LG. Preliminary report on the effects of propolis on wound healing in the dental pulp. Z Natforsch C J Biosci. 1998; 53:1045–1048.
Article34. Ghilotti J, Sanz JL, López-García S, Guerrero-Gironés J, Pecci-Lloret MP, Lozano A, Llena C, Rodríguez-Lozano FJ, Forner L, Spagnuolo G. Comparative surface morphology, chemical composition, and cytocompatibility of Bio-C Repair, Biodentine, and ProRoot MTA on hDPCs. Materials (Basel). 2020; 13:2189. PMID: 32397585.
Article35. Sanz JL, Rodríguez-Lozano FJ, Lopez-Gines C, Monleon D, Llena C, Forner L. Dental stem cell signaling pathway activation in response to hydraulic calcium silicate-based endodontic cements: a systematic review of in vitro studies. Dent Mater. 2021; 37:e256–e268. PMID: 33573840.36. Mendonça ICG, Medeiros MLBB, Penteado RAPM, Parolia A, Porto ICCM. An overview of the toxic effects and allergic reactions caused by propolis. Pharmacologyonline. 2013; 2:96–105.37. Erdemli HK, Akyol S, Armutcu F, Akyol O. Antiviral properties of caffeic acid phenethyl ester and its potential application. J Intercult Ethnopharmacol. 2015; 4:344–347. PMID: 26649239.
Article38. Zare Jahromi M, Ranjbarian P, Shiravi S. Cytotoxicity evaluation of Iranian propolis and calcium hydroxide on dental pulp fibroblasts. J Dent Res Dent Clin Dent Prospect. 2014; 8:130–133.39. Parolia A, Kundabala M, Rao NN, Acharya SR, Agrawal P, Mohan M, Thomas M. A comparative histological analysis of human pulp following direct pulp capping with propolis, mineral trioxide aggregate and Dycal. Aust Dent J. 2010; 55:59–64. PMID: 20415913.
Article40. Dorman HJ, Deans SG. Antimicrobial agents from plants: antibacterial activity of plant volatile oils. J Appl Microbiol. 2000; 88:308–316. PMID: 10736000.
Article41. Odabaş ME, Cinar C, Tulunoğlu O, Işik B. A new haemostatic agent’s effect on the success of calcium hydroxide pulpotomy in primary molars. Pediatr Dent. 2011; 33:529–534. PMID: 22353416.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Systemic Contact Dermatitis from Propolis Ingestion
- Antimicrobial Activity of Korean Propolis Extracts on Oral Pathogenic Microorganisms
- Antimicrobial effect of Australia propolis on cariogenic and periodontopathic bacteria
- In vitro Evaluation of the Antifungal Activity of Propolis Extract on Cryptococcus neoformans and Candida albicans
- A Case of Allergic Contact Dermatitis to Propolis on the Lips and Oral Mucosa