Diabetes Metab J.
2023 Nov;47(6):826-836. 10.4093/dmj.2022.0273.
Clinical and Lifestyle Determinants of Continuous Glucose Monitoring Metrics in Insulin-Treated Patients with Type 2 Diabetes Mellitus
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Korea University College of Medicine, Seoul,
- 2Department of Convergence IT Engineering, Pohang University of Science and Technology, Pohang, Korea
- 3Department of Electrical Engineering, Pohang University of Science and Technology, Pohang, Korea
- 4BK21 FOUR R&E Center for Learning Health Systems, Korea University, Seoul, Korea
- KMID: 2548159
- DOI: http://doi.org/10.4093/dmj.2022.0273
Abstract
- Background
There was limited evidence to evaluate the association between lifestyle habits and continuous glucose monitoring (CGM) metrics. Thus, we aimed to depict the behavioral and metabolic determinants of CGM metrics in insulin-treated patients with type 2 diabetes mellitus (T2DM).
Methods
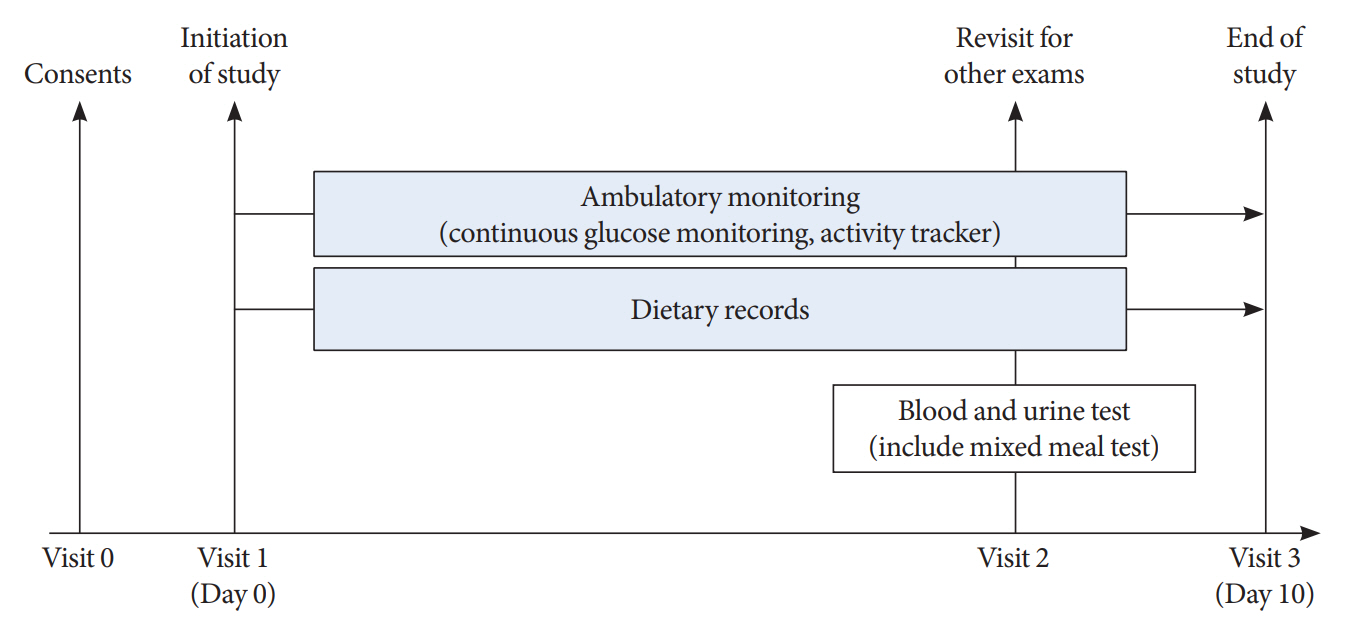
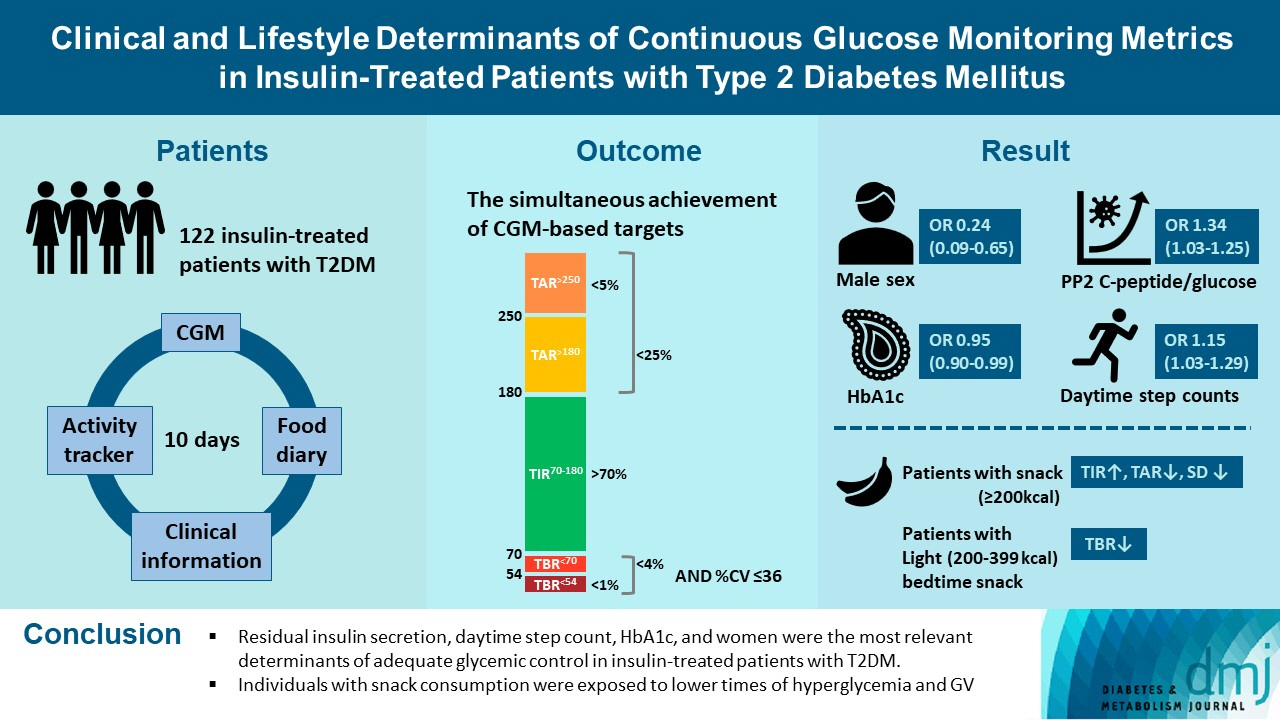
This is a prospective observational study. We analyzed data from 122 insulin-treated patients with T2DM. Participants wore Dexcom G6 and Fitbit, and diet information was identified for 10 days. Multivariate-adjusted logistic regression analysis was performed for the simultaneous achievement of CGM-based targets, defined by the percentage of time in terms of hyper, hypoglycemia and glycemic variability (GV). Intake of macronutrients and fiber, step counts, sleep, postprandial C-peptide-to-glucose ratio (PCGR), information about glucose lowering medications and metabolic factors were added to the analyses. Additionally, we evaluated the impact of the distribution of energy and macronutrient during a day, and snack consumption on CGM metrics.
Results
Logistic regression analysis revealed that female, participants with high PCGR, low glycosylated hemoglobin (HbA1c) and daytime step count had a higher probability of achieving all targets based on CGM (odds ratios [95% confidence intervals] which were 0.24 [0.09 to 0.65], 1.34 [1.03 to 1.25], 0.95 [0.9 to 0.99], and 1.15 [1.03 to 1.29], respectively). And participants who ate snacks showed a shorter period of hyperglycemia and less GV compared to those without.
Conclusion
We confirmed that residual insulin secretion, daytime step count, HbA1c, and women were the most relevant determinants of adequate glycemic control in insulin-treated patients with T2DM. In addition, individuals with snack consumption were exposed to lower times of hyperglycemia and GV.
Figure
Reference
-
1. Wakasugi S, Mita T, Katakami N, Okada Y, Yoshii H, Osonoi T, et al. Associations between continuous glucose monitoring-derived metrics and arterial stiffness in Japanese patients with type 2 diabetes. Cardiovasc Diabetol. 2021; 20:15.
Article2. Mori H, Okada Y, Kurozumi A, Narisawa M, Tanaka Y. Factors influencing inter-day glycemic variability in diabetic outpatients receiving insulin therapy. J Diabetes Investig. 2017; 8:69–74.
Article3. Siegelaar SE, Holleman F, Hoekstra JB, DeVries JH. Glucose variability: does it matter? Endocr Rev. 2010; 31:171–82.
Article4. Smith-Palmer J, Brandle M, Trevisan R, Orsini Federici M, Liabat S, Valentine W. Assessment of the association between glycemic variability and diabetes-related complications in type 1 and type 2 diabetes. Diabetes Res Clin Pract. 2014; 105:273–84.
Article5. ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, et al. 6. Glycemic targets: standards of care in diabetes-2023. Diabetes Care. 2023; 46(Suppl 1):S97–110.6. Bergenstal RM, Ahmann AJ, Bailey T, Beck RW, Bissen J, Buckingham B, et al. Recommendations for standardizing glucose reporting and analysis to optimize clinical decision making in diabetes: the Ambulatory Glucose Profile (AGP). Diabetes Technol Ther. 2013; 15:198–211.
Article7. Danne T, Nimri R, Battelino T, Bergenstal RM, Close KL, DeVries JH, et al. International consensus on use of continuous glucose monitoring. Diabetes Care. 2017; 40:1631–40.
Article8. Ohara M, Hiromura M, Nagaike H, Kohata Y, Fujikawa T, Goto S, et al. Relationship between glucose variability evaluated by continuous glucose monitoring and clinical factors, including glucagon-stimulated insulin secretion in patients with type 2 diabetes. Diabetes Res Clin Pract. 2019; 158:107904.
Article9. Gomez AM, Henao-Carillo DC, Taboada L, Fuentes O, Lucero O, Sanko A, et al. Clinical factors associated with high glycemic variability defined by coefficient of variation in patients with type 2 diabetes. Med Devices (Auckl). 2021; 14:97–103.10. Abbie E, Francois ME, Chang CR, Barry JC, Little JP. A low-carbohydrate protein-rich bedtime snack to control fasting and nocturnal glucose in type 2 diabetes: a randomized trial. Clin Nutr. 2020; 39:3601–6.
Article11. Kishimoto I, Ohashi A. Impact of lifestyle behaviors on postprandial hyperglycemia during continuous glucose monitoring in adult males with overweight/obesity but without diabetes. Nutrients. 2021; 13:3092.
Article12. Park SH, Yao J, Chua XH, Chandran SR, Gardner DS, Khoo CM, et al. Diet and physical activity as determinants of continuously measured glucose levels in persons at high risk of type 2 diabetes. Nutrients. 2022; 14:366.
Article13. Cho CH, Lee T, Kim MG, In HP, Kim L, Lee HJ. Mood prediction of patients with mood disorders by machine learning using passive digital phenotypes based on the circadian rhythm: prospective observational cohort study. J Med Internet Res. 2019; 21:e11029.
Article14. The Ministry of Food and Drug Safety. Integrated data collection of nutritional ingredients for eating out (2012-2017). Available from: https://various.foodsafetykorea.go.kr/nutrient/ (cited 2023 May 24).15. Kohri T, Kaba N. Study of the effects of snack-centered dietary education on first-grade elementary students and duration of these effects. Am J Public Health Res. 2015; 3:1–7.16. Suh Y, Lee EK, Chung YJ. Comparison of nutritional status by energy level of night snack in Korean adults: using the data from 2005 Korean National Health and Nutrition Examination Survey. Korean J Nutr. 2012; 45:479–88.
Article17. Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009; 150:604–12.
Article18. Lee EY, Hwang S, Lee SH, Lee YH, Choi AR, Lee Y, et al. Postprandial C-peptide to glucose ratio as a predictor of b-cell function and its usefulness for staged management of type 2 diabetes. J Diabetes Investig. 2014; 5:517–24.19. Beck RW, Connor CG, Mullen DM, Wesley DM, Bergenstal RM. The fallacy of average: how using HbA1c alone to assess glycemic control can be misleading. Diabetes Care. 2017; 40:994–9.
Article20. Carpenter C, Yang CH, West D. A comparison of sedentary behavior as measured by the Fitbit and ActivPAL in college students. Int J Environ Res Public Health. 2021; 18:3914.
Article21. Ohara M, Fukui T, Ouchi M, Watanabe K, Suzuki T, Yamamoto S, et al. Relationship between daily and day-to-day glycemic variability and increased oxidative stress in type 2 diabetes. Diabetes Res Clin Pract. 2016; 122:62–70.
Article22. Jin SM, Kim TH, Bae JC, Hur KY, Lee MS, Lee MK, et al. Clinical factors associated with absolute and relative measures of glycemic variability determined by continuous glucose monitoring: an analysis of 480 subjects. Diabetes Res Clin Pract. 2014; 104:266–72.
Article23. Saisho Y. Postprandial C-peptide to glucose ratio as a marker of b cell function: implication for the management of type 2 diabetes. Int J Mol Sci. 2016; 17:744.
Article24. Ranjan A, Schmidt S, Damm-Frydenberg C, Holst JJ, Madsbad S, Norgaard K. Short-term effects of a low carbohydrate diet on glycaemic variables and cardiovascular risk markers in patients with type 1 diabetes: a randomized open-label crossover trial. Diabetes Obes Metab. 2017; 19:1479–84.25. Tay J, Thompson CH, Luscombe-Marsh ND, Wycherley TP, Noakes M, Buckley JD, et al. Effects of an energy-restricted low-carbohydrate, high unsaturated fat/low saturated fat diet versus a high-carbohydrate, low-fat diet in type 2 diabetes: a 2-year randomized clinical trial. Diabetes Obes Metab. 2018; 20:858–71.
Article26. Jakubowicz D, Barnea M, Wainstein J, Froy O. High caloric intake at breakfast vs. dinner differentially influences weight loss of overweight and obese women. Obesity (Silver Spring). 2013; 21:2504–12.
Article27. Jakubowicz D, Wainstein J, Tsameret S, Landau Z. Role of high energy breakfast “big breakfast diet” in clock gene regulation of postprandial hyperglycemia and weight loss in type 2 diabetes. Nutrients. 2021; 13:1558.
Article28. Partula V, Deschasaux M, Druesne-Pecollo N, Latino-Martel P, Desmetz E, Chazelas E, et al. Associations between consumption of dietary fibers and the risk of cardiovascular diseases, cancers, type 2 diabetes, and mortality in the prospective NutriNet-Sante cohort. Am J Clin Nutr. 2020; 112:195–207.
Article29. Timmer R, Bogaardt L, Brummelhuis WJ, van Oostrom CT, van Kerkhof LW, Wong A, et al. A randomized crossover trial assessing time of day snack consumption and resulting postprandial glycemic response in a real-life setting among healthy adults. Chronobiol Int. 2022; 39:1329–39.
Article30. Manders RJ, Praet SF, Meex RC, Koopman R, de Roos AL, Wagenmakers AJ, et al. Protein hydrolysate/leucine co-ingestion reduces the prevalence of hyperglycemia in type 2 diabetic patients. Diabetes Care. 2006; 29:2721–2.
Article31. Miya A, Nakamura A, Handa T, Nomoto H, Kameda H, Cho KY, et al. Log-linear relationship between endogenous insulin secretion and glycemic variability in patients with type 2 diabetes on continuous glucose monitoring. Sci Rep. 2021; 11:9057.
Article32. Zhou Z, Sun B, Huang S, Zhu C, Bian M. Glycemic variability: adverse clinical outcomes and how to improve it? Cardiovasc Diabetol. 2020; 19:102.
Article33. Yoo JH, Kim G, Lee HJ, Sim KH, Jin SM, Kim JH. Effect of structured individualized education on continuous glucose monitoring use in poorly controlled patients with type 1 diabetes: a randomized controlled trial. Diabetes Res Clin Pract. 2022; 184:109209.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Glucose Management Using Continuous Glucose Monitors
- Understanding of type 1 diabetes mellitus: what we know and where we go
- Management of Adults with Type 1 Diabetes: Current Status and Suggestions
- 2023 Clinical Practice Guidelines for Diabetes: Continuous Glucose Monitoring and Insulin Pumps
- Patient Education in Type 2 Diabetes Mellitus through Continuous Glucose Monitoring Reports