J Korean Med Sci.
2023 Aug;38(34):e267. 10.3346/jkms.2023.38.e267.
Associations of Serum Uric Acid Level With Liver Enzymes, Nonalcoholic Fatty Liver Disease, and Liver Fibrosis in Korean Men and Women: A CrossSectional Study Using Nationally Representative Data
- Affiliations
-
- 1Department of Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 2Department of Preventive Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 3Department of Internal Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 4Regeneration Medicine Research Center, Yonsei University Wonju College of Medicine, Wonju, Korea
- 5Cell Therapy and Tissue Engineering Center, Yonsei University Wonju College of Medicine, Wonju, Korea
- 6Institute of Occupational and Environmental Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- KMID: 2545544
- DOI: http://doi.org/10.3346/jkms.2023.38.e267
Abstract
- Background
This study aimed to determine whether serum uric acid (SUA) levels are associated with various indices of liver damage in the adult Korean population.
Methods
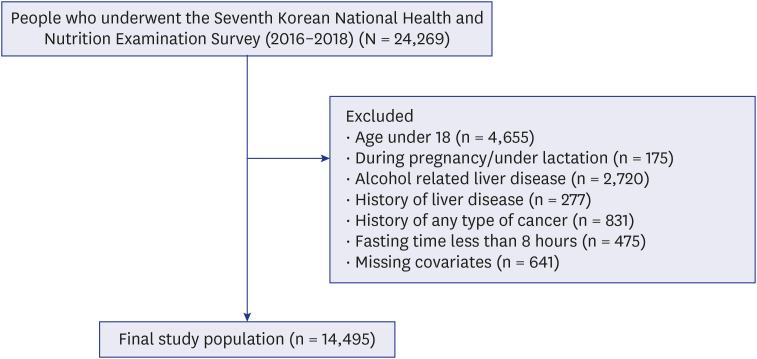
We used the Seventh (VII) Korean National Health and Nutritional Examination Surveys. Our study population comprised 6,007 men and 8,488 women. Levels of SUA were divided into four groups (≤ 5.3, 5.3–6.0, 6.0–7.0, and > 7.0 mg/dL for men and ≤ 4.0, 4.0–4.8, 4.8–6.0, and > 6.0 mg/dL for women). Elevated liver enzyme levels were defined as > 35 (men) and > 31 (women) IU/L for aspartate aminotransferase (AST), > 45 (men) and > 34 (women) IU/L for alanine aminotransferase (ALT). Hepatic steatosis index and fibrosis (FIB)-4 index was used to determine nonalcoholic fatty liver disease (NAFLD) and liver FIB, respectively. Adjusted odds ratios (aORs) were calculated by logistic regression analysis for liver enzymes, NAFLD, and liver FIB, according to the SUA level.
Results
Among women, the 4.8–6.0 and > 6.0 mg/dL SUA groups showed higher ORs of elevated AST (aOR, 1.78 and 2.03; 95% confidence interval [CI], 1.37–2.32 and 1.40–2.96, respectively; P < 0.001) and the 4.0–4.8, 4.8–6.0, and > 6.0 mg/dL SUA groups showed a higher ORs of ALT elevation (aOR, 1.35, 2.26, and 2.37; 95% CI, 1.02–1.79, 1.72–2.97, and 1.60–3.50, respectively; P < 0.001) compared to the lowest level SUA group. Among women with normal ALT, > 6.0 mg/dL SUA group showed higher OR of NAFLD status (aOR, 1.52; 95% CI, 1.06–2.19). Among men and women with NAFLD, hyperuricemia showed higher ORs of liver FIB (aOR, 2.25 and 1.89; 95% CI, 1.21–4.19 and 1.09–3.27, respectively) than the lowest level SUA group.
Conclusion
High SUA levels may be associated with elevated liver enzymes and NAFLD, mainly in women. Even in women with normal ALT levels, SUA levels may predict the NAFLD status. Hyperuricemia may predict advanced liver FIB in both men and women with NAFLD. Further studies investigating the causal effects of SUA on liver damage are required.
Figure
Reference
-
1. Duckworth WC, Hamel FG, Peavy DE. Hepatic metabolism of insulin. Am J Med. 1988; 85(5A):71–76. PMID: 3057896.
Article2. Lala V, Goyal A, Minter DA. Liver Function Tests. Treasure Island, FL, USA: StatPearls Publishing;2022.3. Giannini EG, Testa R, Savarino V. Liver enzyme alteration: a guide for clinicians. CMAJ. 2005; 172(3):367–379. PMID: 15684121.
Article4. Eslam M, Newsome PN, Sarin SK, Anstee QM, Targher G, Romero-Gomez M, et al. A new definition for metabolic dysfunction-associated fatty liver disease: an international expert consensus statement. J Hepatol. 2020; 73(1):202–209. PMID: 32278004.
Article5. Zhou WC, Zhang QB, Qiao L. Pathogenesis of liver cirrhosis. World J Gastroenterol. 2014; 20(23):7312–7324. PMID: 24966602.
Article6. Hediger MA, Johnson RJ, Miyazaki H, Endou H. Molecular physiology of urate transport. Physiology (Bethesda). 2005; 20(2):125–133. PMID: 15772301.
Article7. Facchini F, Chen YD, Hollenbeck CB, Reaven GM. Relationship between resistance to insulin-mediated glucose uptake, urinary uric acid clearance, and plasma uric acid concentration. JAMA. 1991; 266(21):3008–3011. PMID: 1820474.
Article8. Sautin YY, Johnson RJ. Uric acid: the oxidant-antioxidant paradox. Nucleosides Nucleotides Nucleic Acids. 2008; 27(6):608–619. PMID: 18600514.
Article9. Li L, Zhang Y, Zeng C. Update on the epidemiology, genetics, and therapeutic options of hyperuricemia. Am J Transl Res. 2020; 12(7):3167–3181. PMID: 32774692.10. Ryu S, Chang Y, Zhang Y, Kim SG, Cho J, Son HJ, et al. A cohort study of hyperuricemia in middle-aged South Korean men. Am J Epidemiol. 2012; 175(2):133–143. PMID: 22156041.
Article11. Dalbeth N, Choi HK, Joosten LA, Khanna PP, Matsuo H, Perez-Ruiz F, et al. Gout. Nat Rev Dis Primers. 2019; 5(1):69. PMID: 31558729.
Article12. Liebman SE, Taylor JG, Bushinsky DA. Uric acid nephrolithiasis. Curr Rheumatol Rep. 2007; 9(3):251–257. PMID: 17531180.
Article13. Yoo TW, Sung KC, Shin HS, Kim BJ, Kim BS, Kang JH, et al. Relationship between serum uric acid concentration and insulin resistance and metabolic syndrome. Circ J. 2005; 69(8):928–933. PMID: 16041161.
Article14. Sui X, Church TS, Meriwether RA, Lobelo F, Blair SN. Uric acid and the development of metabolic syndrome in women and men. Metabolism. 2008; 57(6):845–852. PMID: 18502269.
Article15. Selby JV, Friedman GD, Quesenberry CP Jr. Precursors of essential hypertension: pulmonary function, heart rate, uric acid, serum cholesterol, and other serum chemistries. Am J Epidemiol. 1990; 131(6):1017–1027. PMID: 2343854.
Article16. Fang J, Alderman MH. Serum uric acid and cardiovascular mortality the NHANES I epidemiologic follow-up study, 1971-1992. National Health and Nutrition Examination Survey. JAMA. 2000; 283(18):2404–2410. PMID: 10815083.17. Bos MJ, Koudstaal PJ, Hofman A, Witteman JC, Breteler MM. Uric acid is a risk factor for myocardial infarction and stroke: the Rotterdam study. Stroke. 2006; 37(6):1503–1507. PMID: 16675740.
Article18. Feig DI, Kang DH, Johnson RJ. Uric acid and cardiovascular risk. N Engl J Med. 2008; 359(17):1811–1821. PMID: 18946066.
Article19. Dehghan A, van Hoek M, Sijbrands EJ, Hofman A, Witteman JC. High serum uric acid as a novel risk factor for type 2 diabetes. Diabetes Care. 2008; 31(2):361–362. PMID: 17977935.
Article20. Ross R. Atherosclerosis--an inflammatory disease. N Engl J Med. 1999; 340(2):115–126. PMID: 9887164.21. Hotamisligil GS. Inflammation and metabolic disorders. Nature. 2006; 444(7121):860–867. PMID: 17167474.
Article22. Marchesini G, Brizi M, Bianchi G, Tomassetti S, Bugianesi E, Lenzi M, et al. Nonalcoholic fatty liver disease: a feature of the metabolic syndrome. Diabetes. 2001; 50(8):1844–1850. PMID: 11473047.23. Li Y, Xu C, Yu C, Xu L, Miao M. Association of serum uric acid level with non-alcoholic fatty liver disease: a cross-sectional study. J Hepatol. 2009; 50(5):1029–1034. PMID: 19299029.
Article24. Lee YJ, Lee HR, Lee JH, Shin YH, Shim JY. Association between serum uric acid and non-alcoholic fatty liver disease in Korean adults. Clin Chem Lab Med. 2010; 48(2):175–180. PMID: 19961393.
Article25. Afzali A, Weiss NS, Boyko EJ, Ioannou GN. Association between serum uric acid level and chronic liver disease in the United States. Hepatology. 2010; 52(2):578–589. PMID: 20683957.
Article26. Sirota JC, McFann K, Targher G, Johnson RJ, Chonchol M, Jalal DI. Elevated serum uric acid levels are associated with non-alcoholic fatty liver disease independently of metabolic syndrome features in the United States: liver ultrasound data from the National Health and Nutrition Examination Survey. Metabolism. 2013; 62(3):392–399. PMID: 23036645.
Article27. Chen S, Guo X, Yu S, Sun G, Yang H, Li Z, et al. Association between Serum Uric Acid and Elevated Alanine Aminotransferase in the General Population. Int J Environ Res Public Health. 2016; 13(9):841. PMID: 27563918.
Article28. Molla NH, Kathak RR, Sumon AH, Barman Z, Mou AD, Hasan A, et al. Assessment of the relationship between serum uric acid levels and liver enzymes activity in Bangladeshi adults. Sci Rep. 2021; 11(1):20114. PMID: 34635716.
Article29. Deb S, Sakharkar P. A population based study of liver function amongst adults with hyperuricemia and gout in the United States. Diseases. 2021; 9(3):61. PMID: 34562968.
Article30. Duan H, Zhang R, Chen X, Yu G, Song C, Jiang Y, et al. Associations of uric acid with liver steatosis and fibrosis applying vibration controlled transient elastography in the United States: a nationwide cross-section study. Front Endocrinol (Lausanne). 2022; 13:930224. PMID: 35813625.
Article31. Lee K. Relationship between uric acid and hepatic steatosis among Koreans. Diabetes Metab. 2009; 35(6):447–451. PMID: 19879789.
Article32. Kweon S, Kim Y, Jang MJ, Kim Y, Kim K, Choi S, et al. Data resource profile: the Korea national health and nutrition examination survey (KNHANES). Int J Epidemiol. 2014; 43(1):69–77. PMID: 24585853.
Article33. Zhu Y, Pandya BJ, Choi HK. Prevalence of gout and hyperuricemia in the US general population: the National Health and Nutrition Examination Survey 2007-2008. Arthritis Rheum. 2011; 63(10):3136–3141. PMID: 21800283.
Article34. Lee JH, Kim D, Kim HJ, Lee CH, Yang JI, Kim W, et al. Hepatic steatosis index: a simple screening tool reflecting nonalcoholic fatty liver disease. Dig Liver Dis. 2010; 42(7):503–508. PMID: 19766548.
Article35. Kang SH, Lee HW, Yoo JJ, Cho Y, Kim SU, Lee TH, et al. KASL clinical practice guidelines: management of nonalcoholic fatty liver disease. Clin Mol Hepatol. 2021; 27(3):363–401. PMID: 34154309.
Article36. Sun W, Cui H, Li N, Wei Y, Lai S, Yang Y, et al. Comparison of FIB-4 index, NAFLD fibrosis score and BARD score for prediction of advanced fibrosis in adult patients with non-alcoholic fatty liver disease: a meta-analysis study. Hepatol Res. 2016; 46(9):862–870. PMID: 26763834.
Article37. Shih MH, Lazo M, Liu SH, Bonekamp S, Hernaez R, Clark JM. Association between serum uric acid and nonalcoholic fatty liver disease in the US population. J Formos Med Assoc. 2015; 114(4):314–320. PMID: 25839764.
Article38. Kawamoto R, Kohara K, Tabara Y, Miki T, Otsuka N. Serum gamma-glutamyl transferase levels are associated with metabolic syndrome in community-dwelling individuals. J Atheroscler Thromb. 2009; 16(4):355–362. PMID: 19755788.
Article39. Zhang JX, Xiang GD, Xiang L, Dong J. Serum gamma-glutamyl transferase is associated with the elevated uric acid levels in normotensive Chinese adults. Clin Chim Acta. 2015; 441:122–126. PMID: 25545230.
Article40. Chen SC, Huang YF, Wang JD. Hyperferritinemia and hyperuricemia may be associated with liver function abnormality in obese adolescents. PLoS One. 2012; 7(10):e48645. PMID: 23119080.
Article41. Baldwin W, McRae S, Marek G, Wymer D, Pannu V, Baylis C, et al. Hyperuricemia as a mediator of the proinflammatory endocrine imbalance in the adipose tissue in a murine model of the metabolic syndrome. Diabetes. 2011; 60(4):1258–1269. PMID: 21346177.
Article42. Yang H, Li D, Song X, Liu F, Wang X, Ma Q, et al. Joint associations of serum uric acid and ALT with NAFLD in elderly men and women: a Chinese cross-sectional study. J Transl Med. 2018; 16(1):285. PMID: 30333032.
Article43. Liu J, Xu C, Ying L, Zang S, Zhuang Z, Lv H, et al. Relationship of serum uric acid level with non-alcoholic fatty liver disease and its inflammation progression in non-obese adults. Hepatol Res. 2017; 47(3):E104–E112. PMID: 27172177.
Article44. Sandra S, Lesmana CR, Purnamasari D, Kurniawan J, Gani RA. Hyperuricemia as an independent risk factor for non-alcoholic fatty liver disease (NAFLD) progression evaluated using controlled attenuation parameter-transient elastography: lesson learnt from tertiary referral center. Diabetes Metab Syndr. 2019; 13(1):424–428. PMID: 30641737.
Article45. Modan M, Halkin H, Karasik A, Lusky A. Elevated serum uric acid--a facet of hyperinsulinaemia. Diabetologia. 1987; 30(9):713–718. PMID: 3322912.
Article46. Quiñones Galvan A, Natali A, Baldi S, Frascerra S, Sanna G, Ciociaro D, et al. Effect of insulin on uric acid excretion in humans. Am J Physiol. 1995; 268(1 Pt 1):E1–E5. PMID: 7840165.47. Friedman SL, Neuschwander-Tetri BA, Rinella M, Sanyal AJ. Mechanisms of NAFLD development and therapeutic strategies. Nat Med. 2018; 24(7):908–922. PMID: 29967350.
Article48. Neuschwander-Tetri BA. Non-alcoholic fatty liver disease. BMC Med. 2017; 15(1):45. PMID: 28241825.
Article49. Chen Z, Tian R, She Z, Cai J, Li H. Role of oxidative stress in the pathogenesis of nonalcoholic fatty liver disease. Free Radic Biol Med. 2020; 152:116–141. PMID: 32156524.
Article50. Choi YJ, Shin HS, Choi HS, Park JW, Jo I, Oh ES, et al. Uric acid induces fat accumulation via generation of endoplasmic reticulum stress and SREBP-1c activation in hepatocytes. Lab Invest. 2014; 94(10):1114–1125. PMID: 25111690.
Article51. Szabo G, Csak T. Inflammasomes in liver diseases. J Hepatol. 2012; 57(3):642–654. PMID: 22634126.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Clinical Significance of Hypouricemia in Patients with Liver Cirrhosis
- Noninvasive serum biomarkers for liver steatosis in nonalcoholic fatty liver disease: Current and future developments
- Hepatic and Splenic Volumetry Could Be Used as an Imaging Parameter to Evaluate Fibrosis Grades of the Diffuse Liver Disease Including Nonalcoholic Fatty Liver Disease
- The Relationship between Normal Serum Uric Acid and Nonalcoholic Fatty Liver Disease
- The diagnosis of nonalcoholic fatty liver disease