Korean J healthc assoc Infect Control Prev.
2023 Jun;28(1):126-134. 10.14192/kjicp.2023.28.1.126.
Trend of bb-lactam Resistance in Escherichia coli and Klebsiella pneumoniae Bacteremia and Clinical Characteristics of Cefotaxime-susceptible Extended-spectrum bb-lactamase-producing Isolates
- Affiliations
-
- 1Division of Infectious Diseases, Department of Internal Medicine, Hallym University Sacred Heart Hospital, Hallym University College of Medicine, Anyang, Korea
- 2Department of Infectious Diseases, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 3Department of Laboratory Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2543143
- DOI: http://doi.org/10.14192/kjicp.2023.28.1.126
Abstract
- Background
This study aimed to evaluate the trend of β-lactam resistance in Escherichia coli and Klebsiella pneumoniae bacteremia, the prevalence of cefotaxime-susceptible extendedspectrum β-lactamase-producing (ESBL) E. coli and K. pneumoniae, and the clinical characteristics of these infections.
Methods
A retrospective study was conducted in a tertiary hospital in Korea between 2011 and 2020. All patients with E. coli or K. pneumoniae bacteremia were identified, along with the results for the antibiotic susceptibility and ESBL production test for each isolate.
Results
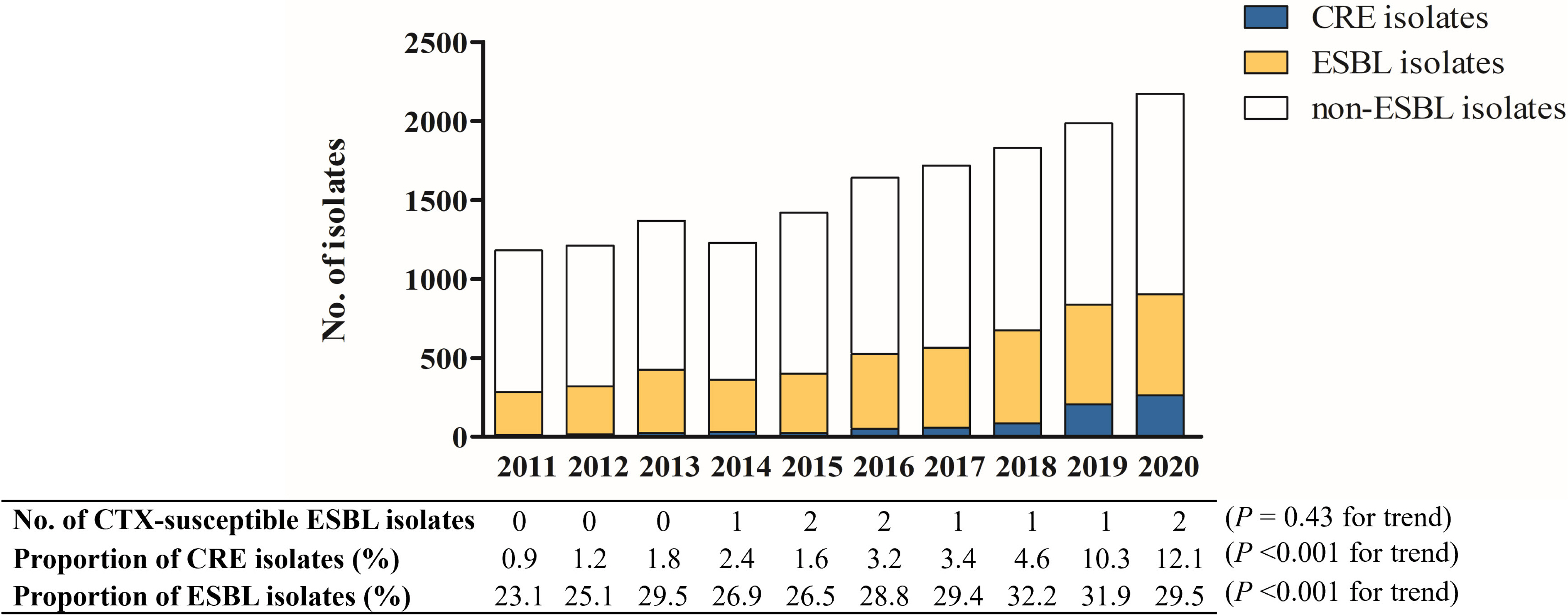
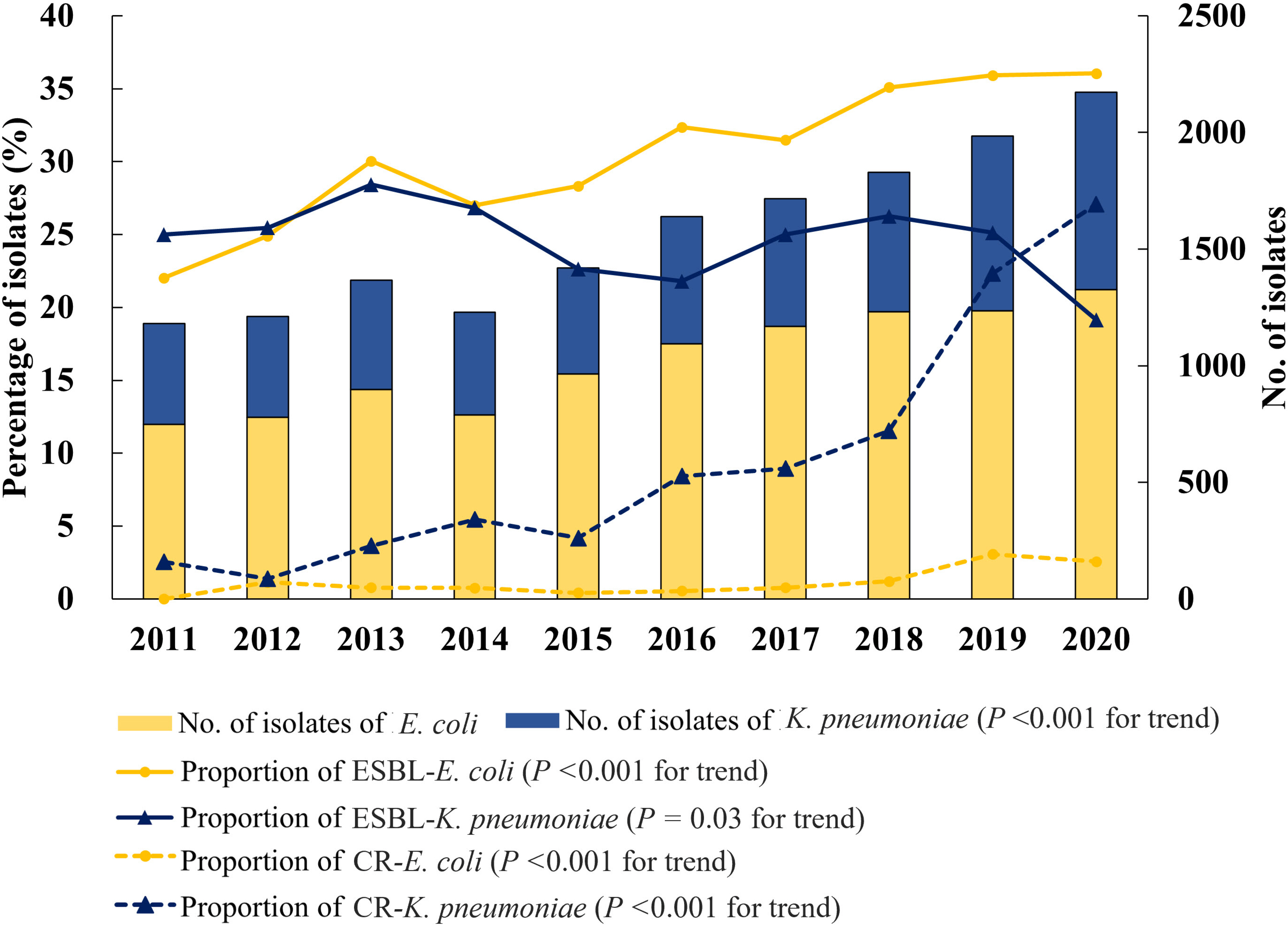
Of 15 706 E. coli and K. pneumoniae bacteremia episodes, only 10 (0.06%) were caused by cefotaxime-susceptible ESBL-producing isolates, and their prevalence remained low during the study period. The proportion of ESBL-producing isolates gradually increased from 23.1% to 29.5%, whereas the proportion of carbapenem-resistant isolates rapidly increased from 0.9% to 12.1%. Furthermore, the proportion of carbapenem-resistant K. pneumoniae increased significantly and surpassed that of carbapenem-susceptible ESBL-producing K. pneumoniae in 2020. In a matched case-control study, the baseline characteristics of patients with cefotaxime-susceptible ESBL E. coli and K. pneumoniae bacteremia were similar to those with cefotaxime-resistant infections. There was no significant difference in 30-day mortality between patients with cefotaxime-susceptible and cefotaxime-resistant ESBL E. coli and K. pneumoniae bacteremia.
Conclusion
The prevalence of cefotaxime-susceptible ESBL E. coli and K. pneumoniae bacteremia and its mortality were very low, supporting the current guidelines for elective ESBL testing. The rapidly increasing carbapenem resistance is a concern.
Figure
Reference
-
1. Bennett JE, Dolin R, Blaser MJ. 2019. Mandell, Douglas, and Bennett's principles and practice of infectious diseases. 9th ed. Elsevier;Philadelphia: p. 270.2. Kadri SS. 2020; Key takeaways from the U.S. CDC's 2019 antibiotic resistance threats report for frontline providers. Crit Care Med. 48:939–45. DOI: 10.1097/CCM.0000000000004371. PMID: 32282351. PMCID: PMC7176261.
Article3. Knothe H, Shah P, Krcmery V, Antal M, Mitsuhashi S. 1983; Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime in clinical isolates of Klebsiella pneumoniae and Serratia marcescens. Infection. 11:315–7. DOI: 10.1007/BF01641355. PMID: 6321357.
Article4. Diekema DJ, Hsueh PR, Mendes RE, Pfaller MA, Rolston KV, Sader HS, et al. 2019; The microbiology of bloodstream infection: 20-year trends from the SENTRY antimicrobial surveillance program. Antimicrob Agents Chemother. 63:e00355–19. DOI: 10.1128/AAC.00355-19. PMID: 31010862. PMCID: PMC6591610.
Article5. Clinical. 2010. Performance standards for antimicrobial susceptibility testing. CLSI supplement M100. 20th ed. Clinical and Laboratory Standards Institute;Wayne: DOI: 10.1016/s0196-4399(01)88009-0.6. Dudley MN, Ambrose PG, Bhavnani SM, Craig WA, Ferraro MJ. Dudley MN, Ambrose PG, Bhavnani SM, Craig WA, Ferraro MJ, Jones RN. Antimicrobial Susceptibility Testing Subcommittee of the Clinical and Laboratory Standards Institute. 2013; Background and rationale for revised clinical and laboratory standards institute interpretive criteria (Breakpoints) for Enterobacteriaceae and Pseudomonas aeruginosa: I. Cephalosporins and Aztreonam. Clin Infect Dis. 56:1301–9. DOI: 10.1093/cid/cit017. PMID: 23334813.7. Andes D, Craig WA. 2005; Treatment of infections with ESBL-producing organisms: pharmacokinetic and pharmacodynamic considerations. Clin Microbiol Infect. 11 Suppl 6:10–7. DOI: 10.1111/j.1469-0691.2005.01265.x. PMID: 16209701.
Article8. Paterson DL, Ko WC, Von Gottberg A, Casellas JM, Mulazimoglu L, Klugman KP, et al. 2001; Outcome of cephalosporin treatment for serious infections due to apparently susceptible organisms producing extended-spectrum beta-lactamases: implications for the clinical microbiology laboratory. J Clin Microbiol. 39:2206–12. DOI: 10.1128/JCM.39.6.2206-2212.2001. PMID: 11376058. PMCID: PMC88112.
Article9. Kang CI, Kim SH, Park WB, Lee KD, Kim HB, Kim EC, et al. 2004; Bloodstream infections due to extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae: risk factors for mortality and treatment outcome, with special emphasis on antimicrobial therapy. Antimicrob Agents Chemother. 48:4574–81. DOI: 10.1128/AAC.48.12.4574-4581.2004. PMID: 15561828. PMCID: PMC529180.
Article10. National Committee for Clinical Laboratory Standards. 2000. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard. 5th ed. National Committee for Clinical Laboratory Standards;Wayne: DOI: 10.1111/j.1469-0691.2005.01265.x.11. Hombach M, Mouttet B, Bloemberg GV. 2013; Consequences of revised CLSI and EUCAST guidelines for antibiotic susceptibility patterns of ESBL- and AmpC β-lactamase-producing clinical Enterobacteriaceae isolates. J Antimicrob Chemother. 68:2092–8. DOI: 10.1093/jac/dkt136. PMID: 23633681.
Article12. Livermore DM, Andrews JM, Hawkey PM, Ho PL, Keness Y, Doi Y, et al. 2012; Are susceptibility tests enough, or should laboratories still seek ESBLs and carbapenemases directly? J Antimicrob Chemother. 67:1569–77. DOI: 10.1093/jac/dks088. PMID: 22461311.
Article13. Hsueh PR, Ko WC, Wu JJ, Lu JJ, Wang FD, Wu HY, et al. 2010; Consensus statement on the adherence to Clinical and Laboratory Standards Institute (CLSI) Antimicrobial Susceptibility Testing Guidelines (CLSI-2010 and CLSI-2010-update) for Enterobacteriaceae in clinical microbiology laboratories in Taiwan. J Microbiol Immunol Infect. 43:452–5. DOI: 10.1016/S1684-1182(10)60070-9. PMID: 21075714.
Article14. Garner JS, Jarvis WR, Emori TG, Horan TC, Hughes JM. 1988; CDC definitions for nosocomial infections, 1988. Am J Infect Control. 16:128–40. Erratum in: Am J Infect Control 1988;16:177. DOI: 10.1016/0196-6553(88)90053-3. PMID: 2841893.
Article15. Friedman ND, Kaye KS, Stout JE, McGarry SA, Trivette SL, Briggs JP, et al. 2002; Health care--associated bloodstream infections in adults: a reason to change the accepted definition of community-acquired infections. Ann Intern Med. 137:791–7. DOI: 10.7326/0003-4819-137-10-200211190-00007. PMID: 12435215.
Article16. McCabe WR, Jackson GG. 1962; Gram-negative bacteremia: II. Clinical, laboratory, and therapeutic observations. Arch Intern Med. 110:856–64. DOI: 10.1001/archinte.1962.03620240038007.17. Charlson ME, Pompei P, Ales KL, MacKenzie CR. 1987; A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis. 40:373–83. DOI: 10.1016/0021-9681(87)90171-8. PMID: 3558716.
Article18. Knaus WA, Draper EA, Wagner DP, Zimmerman JE. 1985; APACHE II: a severity of disease classification system. Crit Care Med. 13:818–29. DOI: 10.1097/00003246-198510000-00009. PMID: 3928249.19. Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, et al. 2003; ; SCCM/ESICM/ACCP/ATS/SIS. 2001 SCCM/ESICM/ACCP/ATS/SIS International Sepsis Definitions Conference. Crit Care Med. 31:1250–6. DOI: 10.1097/01.CCM.0000050454.01978.3B. PMID: 12682500.20. Horan TC, Andrus M, Dudeck MA. 2008; CDC/NHSN surveillance definition of health care-associated infection and criteria for specific types of infections in the acute care setting. Am J Infect Control. 36:309–32. Erratum in: Am J Infect Control 2008;36:655. DOI: 10.1016/j.ajic.2008.03.002. PMID: 18538699.
Article21. Seo H, Lee SC, Chung H, Ra SH, Sung H, Kim MN, et al. 2020; Clinical and microbiological analysis of risk factors for mortality in patients with carbapenem-resistant Enterobacteriaceae bacteremia. Int J Antimicrob Agents. 56:106126. DOI: 10.1016/j.ijantimicag.2020.106126. PMID: 32755654.
Article22. Clinical. 2021. Performance standards for antimicrobial susceptibility testing. CLSI supplement M100. 31st ed. Clinical and Laboratory Standards Institute;Wayne: DOI: 10.1016/s0196-4399(01)88009-0.23. Tamma PD, Goodman KE, Harris AD, Tekle T, Roberts A, Taiwo A, et al. 2017; Comparing the outcomes of patients with carbapenemase-producing and non-carbapenemase-producing carbapenem-resistant Enterobacteriaceae bacteremia. Clin Infect Dis. 64:257–64. DOI: 10.1093/cid/ciw741. PMID: 28013264. PMCID: PMC5241781.24. Wiegand I, Geiss HK, Mack D, Stürenburg E, Seifert H. 2007; Detection of extended-spectrum beta-lactamases among Enterobacteriaceae by use of semiautomated microbiology systems and manual detection procedures. J Clin Microbiol. 45:1167–74. DOI: 10.1128/JCM.01988-06. PMID: 17287329. PMCID: PMC1865808.
Article25. Linscott AJ, Brown WJ. 2005; Evaluation of four commercially available extended-spectrum beta-lactamase phenotypic confirmation tests. J Clin Microbiol. 43:1081–5. DOI: 10.1128/JCM.43.3.1081-1085.2005. PMID: 15750065. PMCID: PMC1081251.
Article26. Infante A, Martín C, Gázquez G, Buñuel F. Ortiz de la Tabla V. 2021; Rapid identification and antimicrobial susceptibility testing of Gram-negative rod on positive blood cultures using MicroScan panels. Eur J Clin Microbiol Infect Dis. 40:151–7. DOI: 10.1007/s10096-020-04014-3. PMID: 32860091.
Article27. Chung JW, Jeon HS, Sung H, Kim MN. 2009; Evaluation of MicroScan and Phoenix system for rapid identification and susceptibility testing using direct inoculation from positive BACTEC blood culture bottles. Korean J Lab Med. 29:25–34. DOI: 10.3343/kjlm.2009.29.1.25. PMID: 19262075.
Article28. Ceken S, Iskender G, Gedik H, Duygu F, Mert D, Kaya AH, et al. 2018; Risk factors for bloodstream infections due to extended-spectrum β-lactamase producing Enterobacteriaceae in cancer patients. J Infect Dev Ctries. 12:265–72. DOI: 10.3855/jidc.9720. PMID: 31851636.
Article29. van Hout D, Verschuuren TD, Bruijning-Verhagen PCJ, Bosch T, Schürch AC, Willems RJL, et al. 2020; Extended-spectrum beta-lactamase (ESBL)-producing and non-ESBL-producing Escherichia coli isolates causing bacteremia in the Netherlands (2014-2016) differ in clonal distribution, antimicrobial resistance gene and virulence gene content. PLoS One. 15:e0227604. DOI: 10.1371/journal.pone.0227604. PMID: 31935253. PMCID: PMC6959556.30. Ku NS, Chung HS, Choi JY, Yong D, Lee K, Kim JM, et al. 2015; Clinical usefulness of the 2010 clinical and laboratory standards institute revised breakpoints for cephalosporin use in the treatment of bacteremia caused by Escherichia coli or Klebsiella spp. Biomed Res Int. 2015:831074. DOI: 10.1155/2015/831074. PMID: 25793209. PMCID: PMC4352463.31. Hong SK, Yong D, Kim K, Hong SS, Hong SG, Khosbayar T, et al. 2013; First outbreak of KPC-2-producing Klebsiella pneumoniae sequence type 258 in a hospital in South Korea. J Clin Microbiol. 51:3877–9. DOI: 10.1128/JCM.01730-13. PMID: 24006005. PMCID: PMC3889788.
Article32. Ahn YS, Bahk HJ, Lee Y. 2021; Epidemiology of carbapenem-resistant Enterobacteriaceae in Korea between 2018 and 2019. Public Health Wkly Rep. 14:413–20.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Evaluation of the Method to Screen Isolates of Extended-Spectrum -Lactamase-Producing Klebsiella pneumoniae and Escherichia coli Using Cefpodoxime Disk
- Antimicrobial Susceptibility of Escherichia coli and Klebsiella pneumoniae Blood Isolates over 5 years:Influence of Extended-Spectrum Beta-Lactamase-Producing Organisms
- In Vitro Susceptibility of piperacillin/tazobactam Against extended-spectrum beta-lactamase-producing Escherichia coli and Klebsiella pneumoniae
- The characteristics of extended-spectrum beta-lactamases in Korean isolates of Enterobacteriaceae
- Characteristics of Extended-spectrum Beta-lactam Hydrolyzing Activity of Klebsiella pneumoniae and Escherichia coli Strains Isolated from Clinical Specimens