Korean Circ J.
2023 Jun;53(6):351-366. 10.4070/kcj.2022.0351.
Historical Review and Future of Cardiac Xenotransplantation
- Affiliations
-
- 1Department of Pathology, Seoul National University Hospital, Seoul National University College of Medicine, Seoul, Korea
- 2Department of Thoracic and Cardiovascular Surgery, Konkuk University Medical Center, Konkuk University School of Medicine, Seoul, Korea
- 3Division of Cardiology, Incheon Sejong Hospital, Incheon, Korea
- 4Department of Thoracic and Cardiovascular Surgery, Chonnam National University Hospital and Medical School, Gwangju, Korea
- 5Department of Pathology and Translational Genomics, Sungkyunkwan University School of Medicine, Samsung Medical Center, Seoul, Korea
- 6Department of Thoracic and Cardiovascular Surgery, Bucheon Sejong Hospital, Bucheon, Korea
- 7Department of Pathology, Incheon Sejong Hospital, Incheon, Korea
- KMID: 2543049
- DOI: http://doi.org/10.4070/kcj.2022.0351
Abstract
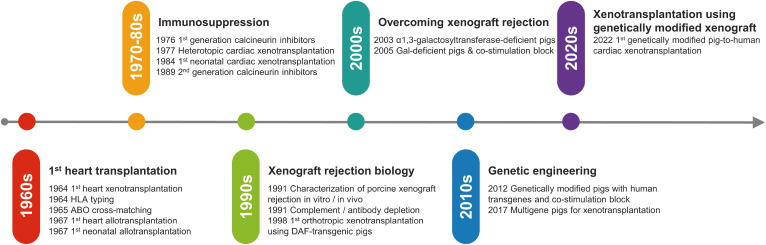
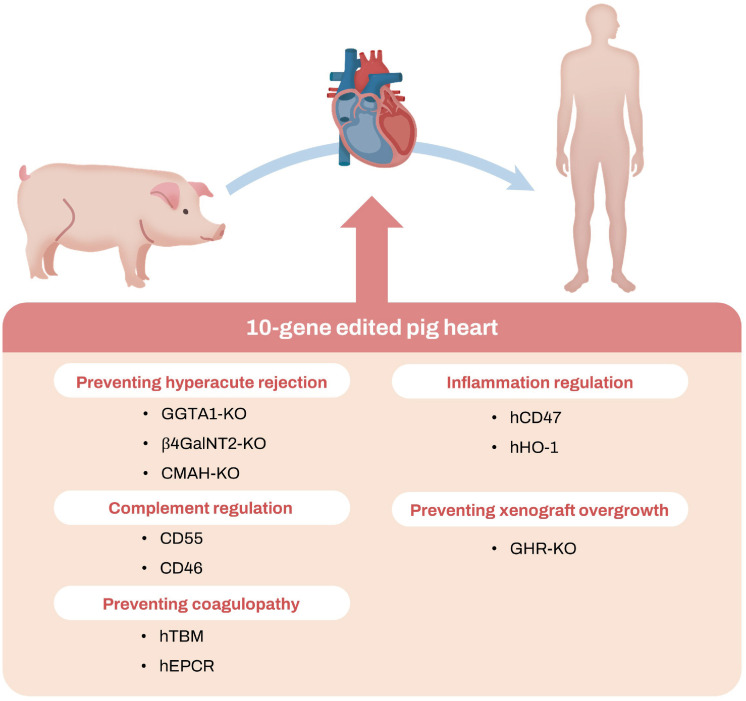
- Along with the development of immunosuppressive drugs, major advances on xenotransplantation were achieved by understanding the immunobiology of xenograft rejection. Most importantly, three predominant carbohydrate antigens on porcine endothelial cells were key elements provoking hyperacute rejection: α1,3-galactose, SDa blood group antigen, and N-glycolylneuraminic acid. Preformed antibodies binding to the porcine major xenoantigen causes complement activation and endothelial cell activation, leading to xenograft injury and intravascular thrombosis. Recent advances in genetic engineering enabled knock-outs of these major xenoantigens, thus producing xenografts with less hyperacute rejection rates. Another milestone in the history of xenotransplantation was the development of co-stimulation blockaded strategy. Unlike allotransplantation, xenotransplantation requires blockade of CD40-CD40L pathway to prevent T-cell dependent B-cell activation and antibody production. In 2010s, advanced genetic engineering of xenograft by inducing the expression of multiple human transgenes became available. So-called ‘multi-gene’ xenografts expressing human transgenes such as thrombomodulin and endothelial protein C receptor were introduced, which resulted in the reduction of thrombotic events and improvement of xenograft survival. Still, there are many limitations to clinical translation of cardiac xenotransplantation. Along with technical challenges, zoonotic infection and physiological discordances are major obstacles. Social barriers including healthcare costs also need to be addressed. Although there are several remaining obstacles to overcome, xenotransplantation would surely become the novel option for millions of patients with end-stage heart failure who have limited options to traditional therapeutics.
Figure
Reference
-
1. Pierson RN 3rd, Fishman JA, Lewis GD, et al. Progress toward cardiac xenotransplantation. Circulation. 2020; 142:1389–1398. PMID: 33017208.2. Griffith BP, Goerlich CE, Singh AK, et al. Genetically modified porcine-to-human cardiac xenotransplantation. N Engl J Med. 2022; 387:35–44. PMID: 35731912.3. Hardy JD, Kurrus FD, Chavez CM, et al. Heart transplantation in man: developmental studies and report of a case. JAMA. 1964; 188:1132–1140. PMID: 14163110.4. Terasaki PI, McClelland JD. Microdroplet assay of human serum cytotoxins. Nature. 1964; 204:998–1000.5. Terasaki PI, Vredevoe DL, Mickey MR, et al. Serotyping for homotransplantation. VII. Selection of kidney donors for thirty-two recipients. Ann N Y Acad Sci. 1966; 129:500–520. PMID: 21165168.6. Starzl TE, Marchioro TL, Porter KA, Iwasaki Y, Cerilli GJ. The use of heterologous antilymphoid agents in canine renal and liver homotransplantation and in human renal homotransplantation. Surg Gynecol Obstet. 1967; 124:301–308. PMID: 4163340.7. Barnard CN. The operation. A human cardiac transplant: an interim report of a successful operation performed at Groote Schuur Hospital, Cape Town. S Afr Med J. 1967; 41:1271–1274. PMID: 4170370.8. Kantrowitz A, Haller JD, Joos H, Cerruti MM, Carstensen HE. Transplantation of the heart in an infant and an adult. Am J Cardiol. 1968; 22:782–790. PMID: 4880223.9. Calne RY, White DJ, Rolles K, Smith DP, Herbertson BM. Prolonged survival of pig orthotopic heart grafts treated with cyclosporin A. Lancet. 1978; 1:1183–1185. PMID: 77948.10. Borel JF, Feurer C, Gubler HU, Stähelin H. Biological effects of cyclosporin A: a new antilymphocytic agent. Agents Actions. 1976; 6:468–475. PMID: 8969.11. Griffith BP, Hardesty RL, Deeb GM, Starzl TE, Bahnson HT. Cardiac transplantation with cyclosporin A and prednisone. Ann Surg. 1982; 196:324–329. PMID: 7051996.12. Bailey LL, Nehlsen-Cannarella SL, Concepcion W, et al. Baboon-to-human cardiac xenotransplantation in a neonate. JAMA. 1985; 254:3321–3329. PMID: 2933538.13. Kino T, Hatanaka H, Miyata S, et al. FK-506, a novel immunosuppressant isolated from a Streptomyces. II. Immunosuppressive effect of FK-506 in vitro. J Antibiot (Tokyo). 1987; 40:1256–1265. PMID: 2445722.14. Armitage JM, Kormos RL, Fung J, et al. Preliminary experience with FK506 in thoracic transplantation. Transplantation. 1991; 52:164–167. PMID: 1713363.15. Dalmasso AP, Platt JL, Bach FH. Reaction of complement with endothelial cells in a model of xenotransplantation. Clin Exp Immunol. 1991; 86(Suppl 1):31–35. PMID: 1718641.16. White DJ, Cozzi E, Langford G, et al. The control of hyperacute rejection by genetic engineering of the donor species. Eye (Lond). 1995; 9:185–189. PMID: 7556715.17. Byrne GW, McCurry KR, Martin MJ, McClellan SM, Platt JL, Logan JS. Transgenic pigs expressing human CD59 and decay-accelerating factor produce an intrinsic barrier to complement-mediated damage. Transplantation. 1997; 63:149–155. PMID: 9000677.18. Phelps CJ, Koike C, Vaught TD, et al. Production of α 1,3-galactosyltransferase-deficient pigs. Science. 2003; 299:411–414. PMID: 12493821.19. Kuwaki K, Tseng YL, Dor FJ, et al. Heart transplantation in baboons using α1,3-galactosyltransferase gene-knockout pigs as donors: initial experience. Nat Med. 2005; 11:29–31. PMID: 15619628.20. Lowe M, Badell IR, Thompson P, et al. A novel monoclonal antibody to CD40 prolongs islet allograft survival. Am J Transplant. 2012; 12:2079–2087. PMID: 22845909.21. Niu D, Wei HJ, Lin L, et al. Inactivation of porcine endogenous retrovirus in pigs using CRISPR-Cas9. Science. 2017; 357:1303–1307. PMID: 28798043.22. Längin M, Mayr T, Reichart B, et al. Consistent success in life-supporting porcine cardiac xenotransplantation. Nature. 2018; 564:430–433. PMID: 30518863.23. Hinrichs A, Riedel EO, Klymiuk N, et al. Growth hormone receptor knockout to reduce the size of donor pigs for preclinical xenotransplantation studies. Xenotransplantation. 2021; 28:e12664. PMID: 33241624.24. Mohiuddin MM, Goerlich CE, Singh AK, et al. Progressive genetic modifications of porcine cardiac xenografts extend survival to 9 months. Xenotransplantation. 2022; 29:e12744. PMID: 35357044.25. Hitchings GH, Elion GB. The chemistry and biochemistry of purine analogs. Ann N Y Acad Sci. 1954; 60:195–199. PMID: 14350523.26. Calne RY, Alexandre GP, Murray JE. A study of the effects of drugs in prolonging survival of homologous renal transplants in dogs. Ann N Y Acad Sci. 1962; 99:743–761. PMID: 14017936.27. Socha WW, Moor-Jankowski J, Ruffié J. Blood groups of primates: present status, theoretical implications and practical applications: a review. J Med Primatol. 1984; 13:11–40. PMID: 6389882.28. Bailey LL, Nehlsen-Cannarella SL, Doroshow RW, et al. Cardiac allotransplantation in newborns as therapy for hypoplastic left heart syndrome. N Engl J Med. 1986; 315:949–951. PMID: 3531854.29. Platt JL, Lindman BJ, Geller RL, et al. The role of natural antibodies in the activation of xenogenic endothelial cells. Transplantation. 1991; 52:1037–1043. PMID: 1750066.30. Sandrin MS, McKenzie IF. Gal α (1,3)Gal, the major xenoantigen(s) recognised in pigs by human natural antibodies. Immunol Rev. 1994; 141:169–190. PMID: 7532618.31. Good AH, Cooper DK, Malcolm AJ, et al. Identification of carbohydrate structures that bind human antiporcine antibodies: implications for discordant xenografting in humans. Transplant Proc. 1992; 24:559–562. PMID: 1566430.32. Byrne GW, Stalboerger PG, Du Z, Davis TR, McGregor CG. Identification of new carbohydrate and membrane protein antigens in cardiac xenotransplantation. Transplantation. 2011; 91:287–292. PMID: 21119562.33. Rose AG, Cooper DK, Human PA, Reichenspurner H, Reichart B. Histopathology of hyperacute rejection of the heart: experimental and clinical observations in allografts and xenografts. J Heart Lung Transplant. 1991; 10:223–234. PMID: 2031919.34. Beerli RR, Dreier B, Barbas CF 3rd. Positive and negative regulation of endogenous genes by designed transcription factors. Proc Natl Acad Sci U S A. 2000; 97:1495–1500. PMID: 10660690.35. Zhang F, Cong L, Lodato S, Kosuri S, Church GM, Arlotta P. Efficient construction of sequence-specific TAL effectors for modulating mammalian transcription. Nat Biotechnol. 2011; 29:149–153. PMID: 21248753.36. Jinek M, Chylinski K, Fonfara I, Hauer M, Doudna JA, Charpentier E. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 2012; 337:816–821. PMID: 22745249.37. Doudna JA, Charpentier E. Genome editing. The new frontier of genome engineering with CRISPR-Cas9. Science. 2014; 346:1258096. PMID: 25430774.38. Yamada K, Yazawa K, Shimizu A, et al. Marked prolongation of porcine renal xenograft survival in baboons through the use of α1,3-galactosyltransferase gene-knockout donors and the cotransplantation of vascularized thymic tissue. Nat Med. 2005; 11:32–34. PMID: 15619627.39. Estrada JL, Martens G, Li P, et al. Evaluation of human and non-human primate antibody binding to pig cells lacking GGTA1/CMAH/β4GalNT2 genes. Xenotransplantation. 2015; 22:194–202. PMID: 25728481.40. Gorsuch WB, Chrysanthou E, Schwaeble WJ, Stahl GL. The complement system in ischemia-reperfusion injuries. Immunobiology. 2012; 217:1026–1033. PMID: 22964228.41. Garcia BL, Zwarthoff SA, Rooijakkers SH, Geisbrecht BV. Novel evasion mechanisms of the classical complement pathway. J Immunol. 2016; 197:2051–2060. PMID: 27591336.42. Waterworth PD, Dunning J, Tolan M, et al. Life-supporting pig-to-baboon heart xenotransplantation. J Heart Lung Transplant. 1998; 17:1201–1207. PMID: 9883761.43. Diamond LE, Quinn CM, Martin MJ, Lawson J, Platt JL, Logan JS. A human CD46 transgenic pig model system for the study of discordant xenotransplantation. Transplantation. 2001; 71:132–142. PMID: 11211178.44. Loveland BE, Milland J, Kyriakou P, et al. Characterization of a CD46 transgenic pig and protection of transgenic kidneys against hyperacute rejection in non-immunosuppressed baboons. Xenotransplantation. 2004; 11:171–183. PMID: 14962279.45. Platt JL, Vercellotti GM, Lindman BJ, Oegema TR Jr, Bach FH, Dalmasso AP. Release of heparan sulfate from endothelial cells. Implications for pathogenesis of hyperacute rejection. J Exp Med. 1990; 171:1363–1368. PMID: 2139104.46. Roussel JC, Moran CJ, Salvaris EJ, Nandurkar HH, d’Apice AJ, Cowan PJ. Pig thrombomodulin binds human thrombin but is a poor cofactor for activation of human protein C and TAFI. Am J Transplant. 2008; 8:1101–1112. PMID: 18444940.47. Iwase H, Ekser B, Hara H, et al. Regulation of human platelet aggregation by genetically modified pig endothelial cells and thrombin inhibition. Xenotransplantation. 2014; 21:72–83. PMID: 24188473.48. Bongoni AK, Klymiuk N, Wolf E, Ayares D, Rieben R, Cowan PJ. Transgenic expression of human thrombomodulin inhibits HMGB1-induced porcine aortic endothelial cell activation. Transplantation. 2016; 100:1871–1879. PMID: 27077599.49. Bühler L, Awwad M, Basker M, et al. High-dose porcine hematopoietic cell transplantation combined with CD40 ligand blockade in baboons prevents an induced anti-pig humoral response. Transplantation. 2000; 69:2296–2304. PMID: 10868629.50. Elgueta R, Benson MJ, de Vries VC, Wasiuk A, Guo Y, Noelle RJ. Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol Rev. 2009; 229:152–172. PMID: 19426221.51. Ezzelarab MB, Ekser B, Isse K, et al. Increased soluble CD154 (CD40 ligand) levels in xenograft recipients correlate with the development of de novo anti-pig IgG antibodies. Transplantation. 2014; 97:502–508. PMID: 24487395.52. Noelle RJ, Roy M, Shepherd DM, Stamenkovic I, Ledbetter JA, Aruffo A. A 39-kDa protein on activated helper T cells binds CD40 and transduces the signal for cognate activation of B cells. Proc Natl Acad Sci U S A. 1992; 89:6550–6554. PMID: 1378631.53. Yamamoto T, Hara H, Foote J, et al. Life-supporting kidney xenotransplantation from genetically engineered pigs in baboons: a comparison of two immunosuppressive regimens. Transplantation. 2019; 103:2090–2104. PMID: 31283686.54. Kim EJ, Kwun J, Gibby AC, et al. Costimulation blockade alters germinal center responses and prevents antibody-mediated rejection. Am J Transplant. 2014; 14:59–69. PMID: 24354871.55. Mohiuddin MM, Singh AK, Corcoran PC, et al. Chimeric 2C10R4 anti-CD40 antibody therapy is critical for long-term survival of GTKO.hCD46.hTBM pig-to-primate cardiac xenograft. Nat Commun. 2016; 7:11138. PMID: 27045379.56. Dorling A, Lombardi G, Binns R, Lechler RI. Detection of primary direct and indirect human anti-porcine T cell responses using a porcine dendritic cell population. Eur J Immunol. 1996; 26:1378–1387. PMID: 8647220.57. Martens GR, Reyes LM, Li P, et al. Humoral reactivity of renal transplant-waitlisted patients to cells from GGTA1/CMAH/B4GalNT2, and SLA class I knockout pigs. Transplantation. 2017; 101:e86–e92. PMID: 28114170.58. Barclay AN, Brown MH. The SIRP family of receptors and immune regulation. Nat Rev Immunol. 2006; 6:457–464. PMID: 16691243.59. Chan JL, Singh AK, Corcoran PC, et al. Encouraging experience using multi-transgenic xenografts in a pig-to-baboon cardiac xenotransplantation model. Xenotransplantation. 2017; 24:e12330.60. Park CG, Shin JS, Min BH, Kim H, Yeom SC, Ahn C. Current status of xenotransplantation in South Korea. Xenotransplantation. 2019; 26:e12488. PMID: 30697818.61. Lee SJ, Kim JS, Chee HK, et al. Seven years of experiences of preclinical experiments of xeno-heart transplantation of pig to non-human primate (Cynomolgus monkey). Transplant Proc. 2018; 50:1167–1171. PMID: 29731087.62. Berry GJ, Burke MM, Andersen C, et al. The 2013 International Society for Heart and Lung Transplantation Working Formulation for the standardization of nomenclature in the pathologic diagnosis of antibody-mediated rejection in heart transplantation. J Heart Lung Transplant. 2013; 32:1147–1162. PMID: 24263017.63. Stewart S, Winters GL, Fishbein MC, et al. Revision of the 1990 working formulation for the standardization of nomenclature in the diagnosis of heart rejection. J Heart Lung Transplant. 2005; 24:1710–1720. PMID: 16297770.64. Boulet J, Cunningham JW, Mehra MR. Cardiac xenotransplantation: challenges, evolution, and advances. JACC Basic Transl Sci. 2022; 7:716–729. PMID: 35958689.65. Längin M, Reichart B, Steen S, et al. Cold non-ischemic heart preservation with continuous perfusion prevents early graft failure in orthotopic pig-to-baboon xenotransplantation. Xenotransplantation. 2021; 28:e12636. PMID: 32841431.66. DiChiacchio L, Singh AK, Lewis B, et al. Early experience with preclinical perioperative cardiac xenograft dysfunction in a single program. Ann Thorac Surg. 2020; 109:1357–1361. PMID: 31589847.67. Nilsson J, Jernryd V, Qin G, et al. A nonrandomized open-label phase 2 trial of nonischemic heart preservation for human heart transplantation. Nat Commun. 2020; 11:2976. PMID: 32532991.68. Goerlich CE, Griffith B, Hanna P, et al. The growth of xenotransplanted hearts can be reduced with growth hormone receptor knockout pig donors. J Thorac Cardiovasc Surg. 2023; 165:e69–e81. PMID: 34579956.69. Fishman JA, Scobie L, Takeuchi Y. Xenotransplantation-associated infectious risk: a WHO consultation. Xenotransplantation. 2012; 19:72–81. PMID: 22497509.70. Switzer WM, Michler RE, Shanmugam V, et al. Lack of cross-species transmission of porcine endogenous retrovirus infection to nonhuman primate recipients of porcine cells, tissues, or organs. Transplantation. 2001; 71:959–965. PMID: 11349732.71. Valdes-Gonzalez R, Dorantes LM, Bracho-Blanchet E, Rodríguez-Ventura A, White DJ. No evidence of porcine endogenous retrovirus in patients with type 1 diabetes after long-term porcine islet xenotransplantation. J Med Virol. 2010; 82:331–334. PMID: 20029803.72. Porrett PM, Orandi BJ, Kumar V, et al. First clinical-grade porcine kidney xenotransplant using a human decedent model. Am J Transplant. 2022; 22:1037–1053. PMID: 35049121.73. Denner J. Recent progress in xenotransplantation, with emphasis on virological safety. Ann Transplant. 2016; 21:717–727. PMID: 27872471.74. Yamada K, Tasaki M, Sekijima M, et al. Porcine cytomegalovirus infection is associated with early rejection of kidney grafts in a pig to baboon xenotransplantation model. Transplantation. 2014; 98:411–418. PMID: 25243511.75. Denner J, Längin M, Reichart B, et al. Impact of porcine cytomegalovirus on long-term orthotopic cardiac xenotransplant survival. Sci Rep. 2020; 10:17531. PMID: 33067513.76. Cengiz N, Wareham CS. Ethical considerations in xenotransplantation: a review. Curr Opin Organ Transplant. 2020; 25:483–488. PMID: 32833703.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Overcoming Immunological Barriers in Xenotransplantation: A Historical Review of Previous Research and Future Directions
- Current Status of Solid Organ Xenotransplantation
- Current status of pancreatic islet xenotransplantation
- Current Status and Future Perspectives of Xenotransplantation
- Transplantation Immunology from the Historical Perspective