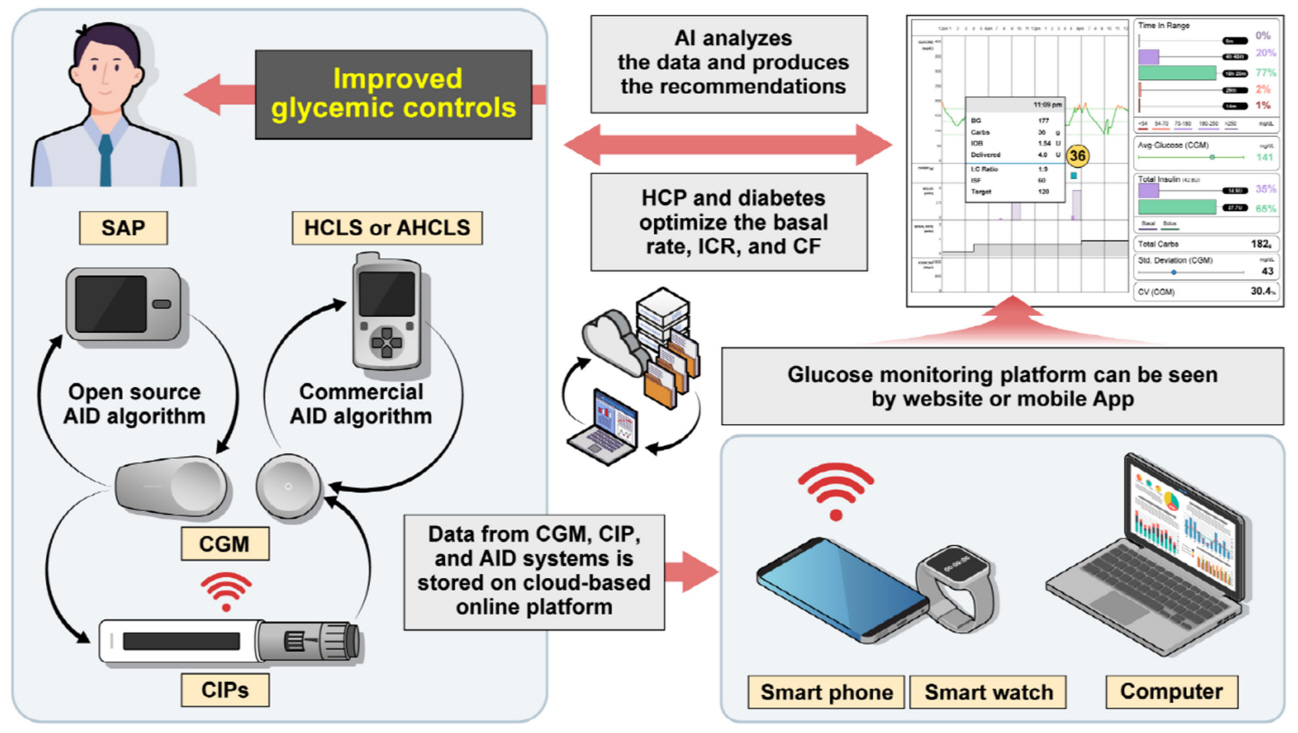
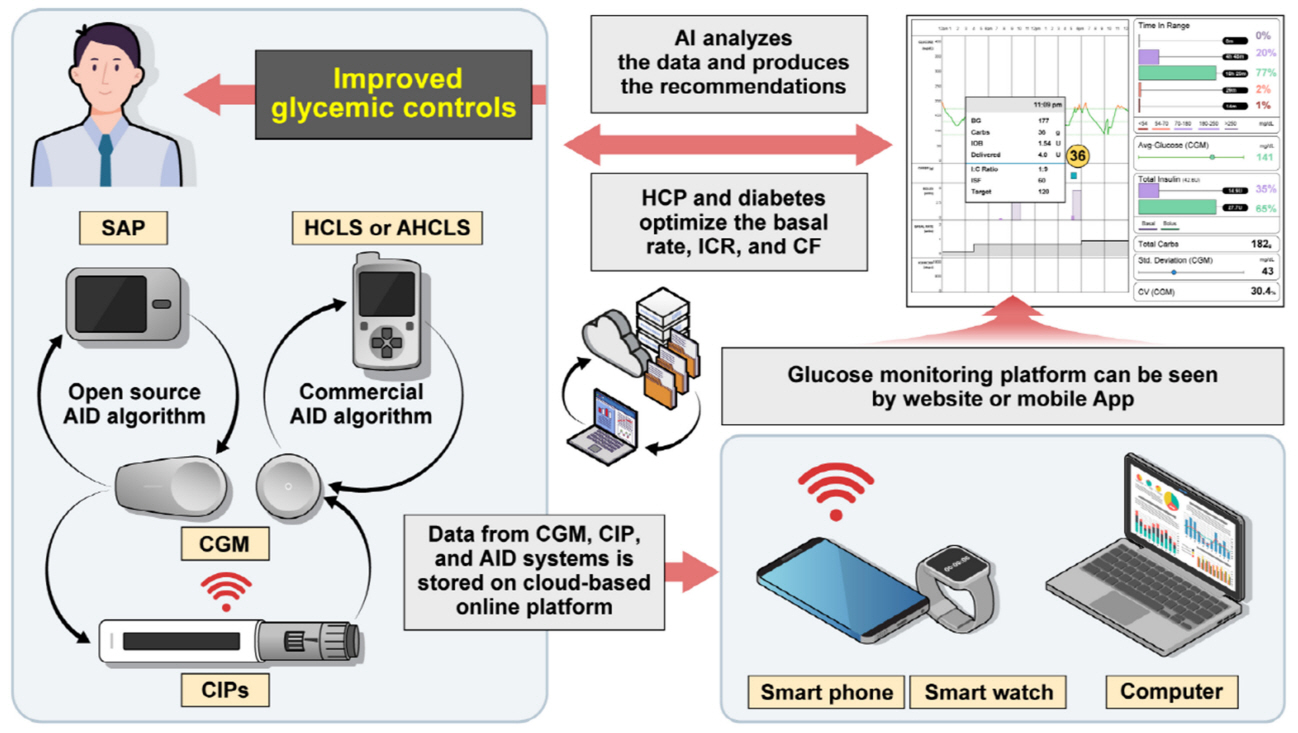
Diabetes Metab J.
2023 Jan;47(1):27-41. 10.4093/dmj.2022.0271.
Advances in Continuous Glucose Monitoring and Integrated Devices for Management of Diabetes with Insulin-Based Therapy: Improvement in Glycemic Control
- Affiliations
-
- 1Division of Endocrinology and Metabolism, Department of Internal Medicine, Yonsei University Wonju College of Medicine, Wonju, Korea
- 2Division of Endocrinology and Metabolism, Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea
- 3Department of Clinical Research Design and Evaluation, Samsung Advanced Institute for Health Sciences & Technology, Sungkyunkwan University, Seoul, Korea
- KMID: 2538928
- DOI: http://doi.org/10.4093/dmj.2022.0271
Abstract
- Continuous glucose monitoring (CGM) technology has evolved over the past decade with the integration of various devices including insulin pumps, connected insulin pens (CIPs), automated insulin delivery (AID) systems, and virtual platforms. CGM has shown consistent benefits in glycemic outcomes in type 1 diabetes mellitus (T1DM) and type 2 diabetes mellitus (T2DM) treated with insulin. Moreover, the combined effect of CGM and education have been shown to improve glycemic outcomes more than CGM alone. Now a CIP is the expected future technology that does not need to be worn all day like insulin pumps and helps to calculate insulin doses with a built-in bolus calculator. Although only a few clinical trials have assessed the effectiveness of CIPs, they consistently show benefits in glycemic outcomes by reducing missed doses of insulin and improving problematic adherence. AID systems and virtual platforms made it possible to achieve target glycosylated hemoglobin in diabetes while minimizing hypoglycemia, which has always been challenging in T1DM. Now fully automatic AID systems and tools for diabetes decisions based on artificial intelligence are in development. These advances in technology could reduce the burden associated with insulin treatment for diabetes.
Keyword
Figure
Cited by 2 articles
-
Navigating the Seas of Glycemic Control: The Role of Continuous Glucose Monitoring in Type 1 Diabetes Mellitus
Jun Sung Moon
Diabetes Metab J. 2023;47(3):345-346. doi: 10.4093/dmj.2023.0125.The Benefits Of Continuous Glucose Monitoring In Pregnancy
Jee Hee Yoo, Jae Hyeon Kim
Endocrinol Metab. 2023;38(5):472-481. doi: 10.3803/EnM.2023.1805.
Reference
-
1. Yoo JH, Kim JH. Time in range from continuous glucose monitoring: a novel metric for glycemic control. Diabetes Metab J. 2020; 44:828–39.
Article2. Lin R, Brown F, James S, Jones J, Ekinci E. Continuous glucose monitoring: a review of the evidence in type 1 and 2 diabetes mellitus. Diabet Med. 2021; 38:e14528.
Article3. Klonoff DC, Kerr D. Smart pens will improve insulin therapy. J Diabetes Sci Technol. 2018; 12:551–3.
Article4. Fleming GA, Petrie JR, Bergenstal RM, Holl RW, Peters AL, Heinemann L. Diabetes digital app technology: benefits, challenges, and recommendations: a consensus report by the European Association for the Study of Diabetes (EASD) and the American Diabetes Association (ADA) diabetes technology working group. Diabetes Care. 2020; 43:250–60.
Article5. Beck RW, Bergenstal RM, Laffel LM, Pickup JC. Advances in technology for management of type 1 diabetes. Lancet. 2019; 394:1265–73.
Article6. Clarke SF, Foster JR. A history of blood glucose meters and their role in self-monitoring of diabetes mellitus. Br J Biomed Sci. 2012; 69:83–93.
Article7. Edelman SV. Regulation catches up to reality. J Diabetes Sci Technol. 2017; 11:160–4.
Article8. Aleppo G, Ruedy KJ, Riddlesworth TD, Kruger DF, Peters AL, Hirsch I, et al. REPLACE-BG: a randomized trial comparing continuous glucose monitoring with and without routine blood glucose monitoring in adults with well-controlled type 1 diabetes. Diabetes Care. 2017; 40:538–45.
Article9. American Diabetes Association Professional Practice Committee, Draznin B, Aroda VR, Bakris G, Benson G, Brown FM, et al. 7. Diabetes technology: standards of medical care in diabetes-2022. Diabetes Care. 2022; 45(Suppl 1):S97–112.10. Wadwa RP, Laffel LM, Shah VN, Garg SK. Accuracy of a factory-calibrated, real-time continuous glucose monitoring system during 10 days of use in youth and adults with diabetes. Diabetes Technol Ther. 2018; 20:395–402.
Article11. Bailey T, Bode BW, Christiansen MP, Klaff LJ, Alva S. The performance and usability of a factory-calibrated flash glucose monitoring system. Diabetes Technol Ther. 2015; 17:787–94.
Article12. Alva S, Bailey T, Brazg R, Budiman ES, Castorino K, Christiansen MP, et al. Accuracy of a 14-day factory-calibrated continuous glucose monitoring system with advanced algorithm in pediatric and adult population with diabetes. J Diabetes Sci Technol. 2022; 16:70–7.
Article13. Medtronic. GUARDIAN(TM) SENSOR 3. Available from: https://www.medtronic.com/us-en/healthcare-professionals/products/diabetes/continuous-glucose-monitoring-systems/guardian-sensor-3.html (cited 2022 Oct 31).14. Medtronic. InPen. Available from: https://www.medtronicdiabetes.com/products/inpen-smart-insulin-pen-system (cited 2022 Oct 31).15. Medtronic. MiniMed(TM) 780G system with Guardian(TM) 4 sensor. Available from: https://www.medtronic-diabetes.com/en-gb/insulin-pump-therapy/minimed-780g-system (cited 2022 Oct 31).16. Christiansen MP, Klaff LJ, Brazg R, Chang AR, Levy CJ, Lam D, et al. A prospective multicenter evaluation of the accuracy of a novel implanted continuous glucose sensor: PRECISE II. Diabetes Technol Ther. 2018; 20:197–206.
Article17. Garg SK, Kipnes M, Castorino K, Bailey TS, Akturk HK, Welsh JB, et al. Accuracy and safety of Dexcom G7 continuous glucose monitoring in adults with diabetes. Diabetes Technol Ther. 2022; 24:373–80.
Article18. Zipp R. Dexcom CEO Sayer on G7 FDA submission, developing a 15-day sensor, M&A plans. Available from: https://www.medtechdive.com/news/dexcom-ceo-g7-fda-15-day-sensorma/625013/ (cited 2022 Oct 31).19. Garg SK, Liljenquist D, Bode B, Christiansen MP, Bailey TS, Brazg RL, et al. Evaluation of accuracy and safety of the nextgeneration up to 180-day long-term implantable eversense continuous glucose monitoring system: the promise study. Diabetes Technol Ther. 2022; 24:84–92.
Article20. Schwarz Y, Konvalina N, Tirosh A. A pilot trial to evaluate the accuracy of a novel non-invasive glucose meter. Sensors (Basel). 2021; 21:6704.
Article21. Beck RW, Riddlesworth T, Ruedy K, Ahmann A, Bergenstal R, Haller S, et al. Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: the diamond randomized clinical trial. JAMA. 2017; 317:371–8.
Article22. Lind M, Polonsky W, Hirsch IB, Heise T, Bolinder J, Dahlqvist S, et al. Continuous glucose monitoring vs conventional therapy for glycemic control in adults with type 1 diabetes treated with multiple daily insulin injections: the gold randomized clinical trial. JAMA. 2017; 317:379–87.
Article23. Beck RW, Riddlesworth TD, Ruedy K, Ahmann A, Haller S, Kruger D, et al. Continuous glucose monitoring versus usual care in patients with type 2 diabetes receiving multiple daily insulin injections: a randomized trial. Ann Intern Med. 2017; 167:365–74.
Article24. Martens T, Beck RW, Bailey R, Ruedy KJ, Calhoun P, Peters AL, et al. Effect of continuous glucose monitoring on glycemic control in patients with type 2 diabetes treated with basal insulin: a randomized clinical trial. JAMA. 2021; 325:2262–72.25. Heinemann L, Freckmann G, Ehrmann D, Faber-Heinemann G, Guerra S, Waldenmaier D, et al. Real-time continuous glucose monitoring in adults with type 1 diabetes and impaired hypoglycaemia awareness or severe hypoglycaemia treated with multiple daily insulin injections (HypoDE): a multicentre, randomised controlled trial. Lancet. 2018; 391:1367–77.
Article26. Aleppo G, Beck RW, Bailey R, Ruedy KJ, Calhoun P, Peters AL, et al. The effect of discontinuing continuous glucose monitoring in adults with type 2 diabetes treated with basal insulin. Diabetes Care. 2021; 44:2729–37.27. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash glucose-sensing technology as a replacement for blood glucose monitoring for the management of insulintreated type 2 diabetes: a multicenter, open-label randomized controlled trial. Diabetes Ther. 2017; 8:55–73.
Article28. Bolinder J, Antuna R, Geelhoed-Duijvestijn P, Kroger J, Weitgasser R. Novel glucose-sensing technology and hypoglycaemia in type 1 diabetes: a multicentre, non-masked, randomised controlled trial. Lancet. 2016; 388:2254–63.
Article29. Oskarsson P, Antuna R, Geelhoed-Duijvestijn P, Kroger J, Weitgasser R, Bolinder J. Impact of flash glucose monitoring on hypoglycaemia in adults with type 1 diabetes managed with multiple daily injection therapy: a pre-specified subgroup analysis of the IMPACT randomised controlled trial. Diabetologia. 2018; 61:539–50.
Article30. Park C, Le QA. The effectiveness of continuous glucose monitoring in patients with type 2 diabetes: a systematic review of literature and meta-analysis. Diabetes Technol Ther. 2018; 20:613–21.
Article31. Maiorino MI, Signoriello S, Maio A, Chiodini P, Bellastella G, Scappaticcio L, et al. Effects of continuous glucose monitoring on metrics of glycemic control in diabetes: a systematic review with meta-analysis of randomized controlled trials. Diabetes Care. 2020; 43:1146–56.
Article32. American Diabetes Association Professional Practice Committee, Draznin B, Aroda VR, Bakris G, Benson G, Brown FM, et al. 6. Glycemic targets: standards of medical care in diabetes-2022. Diabetes Care. 2022; 45(Suppl 1):S83–96.33. Seyed Ahmadi S, Westman K, Pivodic A, Olafsdottir AF, Dahlqvist S, Hirsch IB, et al. The association between HbA1c and time in hypoglycemia during CGM and self-monitoring of blood glucose in people with type 1 diabetes and multiple daily insulin injections: a randomized clinical trial (GOLD-4). Diabetes Care. 2020; 43:2017–24.
Article34. Strategies to Enhance New CGM Use in Early Childhood (SENCE) Study Group. A randomized clinical trial assessing continuous glucose monitoring (CGM) use with standardized education with or without a family behavioral intervention compared with fingerstick blood glucose monitoring in very young children with type 1 diabetes. Diabetes Care. 2021; 44:464–72.35. Hermanns N, Ehrmann D, Schipfer M, Kroger J, Haak T, Kulzer B. The impact of a structured education and treatment programme (FLASH) for people with diabetes using a flash sensor-based glucose monitoring system: results of a randomized controlled trial. Diabetes Res Clin Pract. 2019; 150:111–21.
Article36. Pemberton JS, Kershaw M, Dias R, Idkowiak J, Mohamed Z, Saraff V, et al. DYNAMIC: dynamic glucose management strategies delivered through a structured education program improves time in range in a socioeconomically deprived cohort of children and young people with type 1 diabetes with a history of hypoglycemia. Pediatr Diabetes. 2021; 22:249–60.
Article37. Yaron M, Roitman E, Aharon-Hananel G, Landau Z, Ganz T, Yanuv I, et al. Effect of flash glucose monitoring technology on glycemic control and treatment satisfaction in patients with type 2 diabetes. Diabetes Care. 2019; 42:1178–84.
Article38. Yoo JH, Kim G, Lee HJ, Sim KH, Jin SM, Kim JH. Effect of structured individualized education on continuous glucose monitoring use in poorly controlled patients with type 1 diabetes: a randomized controlled trial. Diabetes Res Clin Pract. 2022; 184:109209.
Article39. Davies MJ, Gagliardino JJ, Gray LJ, Khunti K, Mohan V, Hughes R. Real-world factors affecting adherence to insulin therapy in patients with type 1 or type 2 diabetes mellitus: a systematic review. Diabet Med. 2013; 30:512–24.
Article40. Munshi MN, Slyne C, Greenberg JM, Greaves T, Lee A, Carl S, et al. Nonadherence to insulin therapy detected by Bluetooth-enabled pen cap is associated with poor glycemic control. Diabetes Care. 2019; 42:1129–31.
Article41. Edwards S, He X, Wang W, Poon JL, Meadows E, Price D, et al. Use of connected pen as a diagnostic tool to evaluate missed bolus dosing behavior in people with type 1 and type 2 diabetes. Diabetes Technol Ther. 2022; 24:61–6.
Article42. Adolfsson P, Hartvig NV, Kaas A, Moller JB, Hellman J. Increased time in range and fewer missed bolus injections after introduction of a smart connected insulin pen. Diabetes Technol Ther. 2020; 22:709–18.
Article43. Adolfsson P, Bjornsson V, Hartvig NV, Kaas A, Moller JB, Ogionwo Lange E. Improved glycemic control observed in children with type 1 diabetes following the introduction of smart insulin pens: a real-world study. Diabetes Ther. 2022; 13:43–56.
Article44. Gildon BW. InPen smart insulin pen system: product review and user experience. Diabetes Spectr. 2018; 31:354–8.
Article45. Sy SL, Munshi MM, Toschi E. Can smart pens help improve diabetes management? J Diabetes Sci Technol. 2022; 16:628–34.
Article46. Vigersky R, Smith M, Thanasekaran S, Gaetano A, Im GH, Cordero TL, et al. Impact of inpen smart insulin pen use on real-world glycemic and insulin dosing outcomes in individuals with poorly controlled diabetes. Diabetes. 2021; 70(Supplement_1):219–OR.47. Bigfoot, BIOMEDICAL. The first FDA CLEARED DOSE DECISION SUPPORT SYSTEM INTEGRATED with iCGM. Available from: https://www.bigfootbiomedical.com/ (cited 2022 Oct 31).48. Sanofi. Sanofi and Abbott partner to integrate glucose sensing and insulin delivery technologies to help change the way diabetes is managed. Available from: https://www.sanofi.com/en/media-room/press-releases/2019/2019-09-16-05-00-00-1915647 (cited 2022 Oct 31).49. Grunberger G, Sherr J, Allende M, Blevins T, Bode B, Handelsman Y, et al. American Association of Clinical Endocrinology clinical practice guideline: the use of advanced technology in the management of persons with diabetes mellitus. Endocr Pract. 2021; 27:505–37.
Article50. Brown SA, Kovatchev BP, Raghinaru D, Lum JW, Buckingham BA, Kudva YC, et al. Six-month randomized, multicenter trial of closed-loop control in type 1 diabetes. N Engl J Med. 2019; 381:1707–17.
Article51. Breton MD, Kanapka LG, Beck RW, Ekhlaspour L, Forlenza GP, Cengiz E, et al. A randomized trial of closed-loop control in children with type 1 diabetes. N Engl J Med. 2020; 383:836–45.
Article52. Ware J, Allen JM, Boughton CK, Wilinska ME, Hartnell S, Thankamony A, et al. Randomized trial of closed-loop control in very young children with type 1 diabetes. N Engl J Med. 2022; 386:209–19.
Article53. Moon SJ, Jung I, Park CY. Current advances of artificial pancreas systems: a comprehensive review of the clinical evidence. Diabetes Metab J. 2021; 45:813–39.
Article54. Collyns OJ, Meier RA, Betts ZL, Chan DS, Frampton C, Frewen CM, et al. Improved glycemic outcomes with Medtronic MiniMed advanced hybrid closed-loop delivery: results from a randomized crossover trial comparing automated insulin delivery with predictive low glucose suspend in people with type 1 diabetes. Diabetes Care. 2021; 44:969–75.
Article55. Carlson AL, Sherr JL, Shulman DI, Garg SK, Pop-Busui R, Bode BW, et al. Safety and glycemic outcomes during the MiniMed advanced hybrid closed-loop system pivotal trial in adolescents and adults with type 1 diabetes. Diabetes Technol Ther. 2022; 24:178–89.
Article56. Petrovski G, Al Khalaf F, Campbell J, Day E, Almajaly D, Hussain K, et al. Glycemic outcomes of advanced hybrid closed loop system in children and adolescents with type 1 diabetes, previously treated with multiple daily injections (MiniMed 780G system in T1D individuals, previously treated with MDI). BMC Endocr Disord. 2022; 22:80.
Article57. de Portu S, Vorrink L, Re R, Shin J, Castaneda J, Habteab A, et al. Randomised controlled trial of advanced hybrid closed loop in an adult population with type 1 diabetes (ADAPT): study protocol and rationale. BMJ Open. 2022; 12:e050635.
Article58. Breton MD, Kovatchev BP. One year real-world use of the control-IQ advanced hybrid closed-loop technology. Diabetes Technol Ther. 2021; 23:601–8.
Article59. Brown SA, Forlenza GP, Bode BW, Pinsker JE, Levy CJ, Criego AB, et al. Multicenter trial of a tubeless, on-body automated insulin delivery system with customizable glycemic targets in pediatric and adult participants with type 1 diabetes. Diabetes Care. 2021; 44:1630–40.
Article60. Sherr JL, Bode BW, Forlenza GP, Laffel LM, Schoelwer MJ, Buckingham BA, et al. Safety and glycemic outcomes with a tubeless automated insulin delivery system in very young children with type 1 diabetes: a single-arm multicenter clinical trial. Diabetes Care. 2022; 45:1907–10.
Article61. American Diabetes Association 82nd scientificsessions. Results from insulin-only bionic pancreas trial to be reported. Available from: https://www.adameetingnews.org/live-updates/session-coverage/results-from-insulin-only-bionic-pancreastrial-to-be-reported/ (cited 2022 Oct 31).62. Braune K, Lal RA, Petruzelkova L, Scheiner G, Winterdijk P, Schmidt S, et al. Open-source automated insulin delivery: international consensus statement and practical guidance for health-care professionals. Lancet Diabetes Endocrinol. 2022; 10:58–74.
Article63. Choi MS, Lee S, Kim J, Kim G, Park SM, Kim JH. Do-it-yourself open artificial pancreas system in children and adolescents with type 1 diabetes mellitus: real-world data. Diabetes Metab J. 2022; 46:154–9.
Article64. Lum JW, Bailey RJ, Barnes-Lomen V, Naranjo D, Hood KK, Lal RA, et al. A real-world prospective study of the safety and effectiveness of the loop open source automated insulin delivery system. Diabetes Technol Ther. 2021; 23:367–75.
Article65. Petruzelkova L, Soupal J, Plasova V, Jiranova P, Neuman V, Plachy L, et al. Excellent glycemic control maintained by opensource hybrid closed-loop AndroidAPS during and after sustained physical activity. Diabetes Technol Ther. 2018; 20:744–50.
Article66. Burside MJ, Lewis DM, Crocket H, Meier R, Williman J, Sanders OJ, et al. The CREATE trial: randomized clinical trial comparing open-source automated insulin delivery with sensor augmented pump therapy in type 1 diabetes. Diabetes. 2022; 71(Supplement_1):286–OR.67. Crossen S, Raymond J, Neinstein A. Top 10 tips for successfully implementing a diabetes telehealth program. Diabetes Technol Ther. 2020; 22:920–8.
Article68. Ramakrishnan P, Yan K, Balijepalli C, Druyts E. Changing face of healthcare: digital therapeutics in the management of diabetes. Curr Med Res Opin. 2021; 37:2089–91.
Article69. Kesavadev J, Saboo B, Krishna MB, Krishnan G. Evolution of insulin delivery devices: from syringes, pens, and pumps to DIY artificial pancreas. Diabetes Ther. 2020; 11:1251–69.
Article70. Gerhardsson P, Schwandt A, Witsch M, Kordonouri O, Svensson J, Forsander G, et al. The SWEET project 10-year benchmarking in 19 countries worldwide is associated with improved HbA1c and increased use of diabetes technology in youth with type 1 diabetes. Diabetes Technol Ther. 2021; 23:491–9.
Article71. Nimri R, Oron T, Muller I, Kraljevic I, Alonso MM, Keskinen P, et al. Adjustment of insulin pump settings in type 1 diabetes management: Advisor Pro device compared to physicians’ recommendations. J Diabetes Sci Technol. 2022; 16:364–72.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical Utilities of Continuous Glucose Monitoring and Insulin Pumps in Pediatric Patients with Type 1 Diabetes
- Medical Nutrition Therapy Using Continuous Glucose Monitoring System
- Continuous Glucose Monitoring Sensors for Diabetes Management: A Review of Technologies and Applications
- Application of Continuous Glucose Monitoring System (CGMS) and Patient Education
- Glucose Management Using Continuous Glucose Monitors