Ann Rehabil Med.
2022 Dec;46(6):320-328. 10.5535/arm.22105.
Treadmill Exercise as a Preventive Measure Against Age-Related Anxiety and Social Behavioral Disorders in Rats: When Is It Worth Starting?
- Affiliations
-
- 1Department of Molecular and Cellular Physiology, Graduate School of Medicine, Ehime University, Toon, Japan
- KMID: 2537163
- DOI: http://doi.org/10.5535/arm.22105
Abstract
Objective
To determine the appropriate time points to start regular exercise which could reduce age-related anxiety and impaired social behavior.
Methods
For this study, 8-week-old male Wistar rats were divided into three groups: no exercise (NoEX), short-term exercise (S-Ex), and long-term exercise (L-Ex) groups. S-Ex-group rats started treadmill exercise at 12 months of age, while L-Ex rats started from at 2 months of age. Exercise rats were forced to walk on the treadmill three times per week, with 1- to 2-day intervals for 10 minutes during the first 2 weeks, at 10 m/min until 17 months of age, and at 8 m/min thereafter. At 19 months of age, behavioral tests were performed to assess the effects of exercise on age-induced behavioral change as well as quantitative polymerase chain reaction were done to uncover the mechanism behind the behavioral changes.
Results
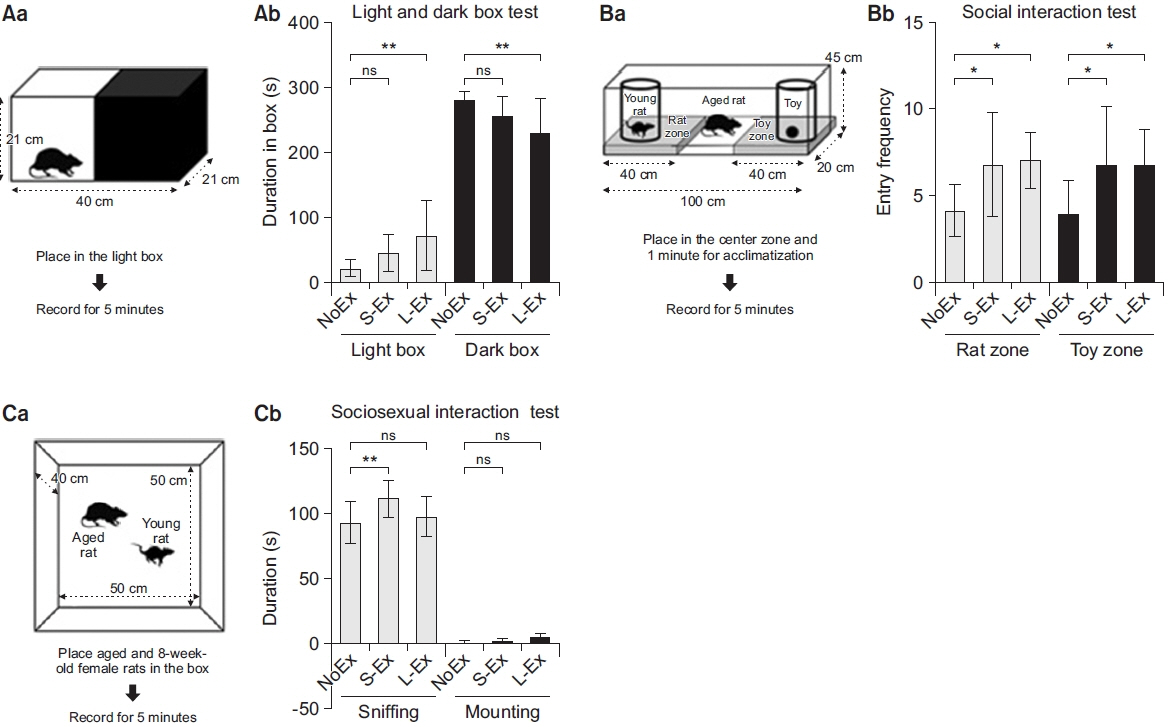
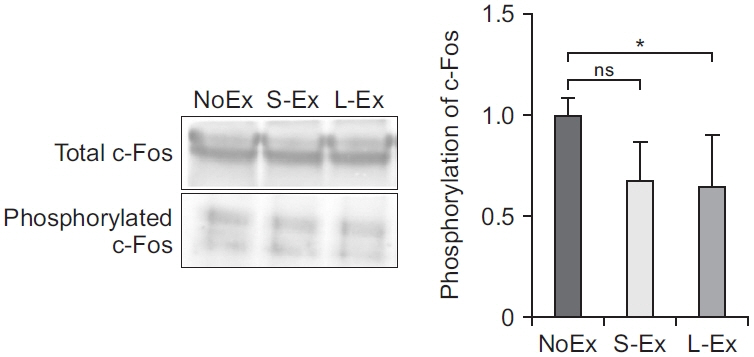
Anxiety-like behavior was improved by long-term exercise. Additionally, rats belonging to the S-Ex and L-Ex groups showed improved social behavior and increased curiosity about interesting objects. The qPCR data showed that treadmill exercise suppressed the expression of immediate-early genes in the prefrontal cortex of the aged rats.
Conclusion
This study suggests that long-term exercise represses early response genes, and in this way, it increases resistance to stress, diminishes anxiety-related behavior, and improves social behavior. These findings underscore the need to consider appropriate time to start exercise to prevent stress induced anxiety related behavior.
Keyword
Figure
Reference
-
1. Beard JR, Officer A, de Carvalho IA, Sadana R, Pot AM, Michel JP, et al. The world report on ageing and health: a policy framework for healthy ageing. Lancet. 2016; 387:2145–54.2. Cotman CW, Berchtold NC. Exercise: a behavioral intervention to enhance brain health and plasticity. Trends Neurosci. 2002; 25:295–301.3. Billinger SA, Arena R, Bernhardt J, Eng JJ, Franklin BA, Johnson CM, et al. Physical activity and exercise recommendations for stroke survivors: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke. 2014; 45:2532–53.4. Kandola A, Ashdown-Franks G, Hendrikse J, Sabiston CM, Stubbs B. Physical activity and depression: towards understanding the antidepressant mechanisms of physical activity. Neurosci Biobehav Rev. 2019; 107:525–39.5. Reid KJ, Baron KG, Lu B, Naylor E, Wolfe L, Zee PC. Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Med. 2010; 11:934–40.6. Rezaee Z, Marandi SM, Esfarjani F. Age-related biochemical dysfunction in 6-OHDA model rats subject to induced-endurance exercise. Arch Gerontol Geriatr. 2022; 98:104554.7. O'Callaghan RM, Griffin EW, Kelly AM. Long-term treadmill exposure protects against age-related neurodegenerative change in the rat hippocampus. Hippocampus. 2009; 19:1019–29.8. Fabel K, Wolf SA, Ehninger D, Babu H, Leal-Galicia P, Kempermann G. Additive effects of physical exercise and environmental enrichment on adult hippocampal neurogenesis in mice. Front Neurosci. 2009; 3:50.9. Van der Borght K, Havekes R, Bos T, Eggen BJ, Van der Zee EA. Exercise improves memory acquisition and retrieval in the Y-maze task: relationship with hippocampal neurogenesis. Behav Neurosci. 2007; 121:324–34.10. Vecchio LM, Meng Y, Xhima K, Lipsman N, Hamani C, Aubert I. The neuroprotective effects of exercise: maintaining a healthy brain throughout aging. Brain Plast. 2018; 4:17–52.11. Pietrelli A, Di Nardo M, Masucci A, Brusco A, Basso N, Matkovic L. Lifelong aerobic exercise reduces the stress response in rats. Neuroscience. 2018; 376:94–107.12. Asl NA, Sheikhzade F, Torchi M, Roshangar L, Khamnei S. Long-term regular exercise promotes memory and learning in young but not in older rats. Pathophysiology. 2008; 15:9–12.13. Sengupta P. The laboratory rat : relating its age with human’s. Int J Prev Med. 2013; 4:624–30.14. Chen D, Zhang Y, Zhang M, Chang J, Zeng Z, Kou X, et al. Exercise attenuates brain aging by rescuing down-regulated Wnt/β-catenin signaling in aged rats. Front Aging Neurosci. 2020; 12:105.15. Baek DJ, Lee CB, Baek SS. Effect of treadmill exercise on social interaction and tyrosine hydroxylase expression in the attention-deficit/hyperactivity disorder rats. J Exerc Rehabil. 2014; 10:252–7.16. Perkins AE, Doremus-Fitzwater TL, Spencer RL, Varlinskaya EI, Conti MM, Bishop C, et al. A working model for the assessment of disruptions in social behavior among aged rats: the role of sex differences, social recognition, and sensorimotor processes. Exp Gerontol. 2016; 76:46–57.17. Bourin M, Petit-Demouliere B, Dhonnchadha BN, Hascoet M. Animal models of anxiety in mice. Fundam Clin Pharmacol. 2007; 21:567–74.18. Kaidanovich-Beilin O, Lipina T, Vukobradovic I, Roder J, Woodgett JR. Assessment of social interaction behaviors. J Vis Exp. 2011; (48):2473.19. Clapcote SJ, Lipina TV, Millar JK, Mackie S, Christie S, Ogawa F, et al. Behavioral phenotypes of Disc1 missense mutations in mice. Neuron. 2007; 54:387–402.20. Portillo W, Unda N, Camacho FJ, Sanchez M, Corona R, Arzate DM, et al. Sexual activity increases the number of newborn cells in the accessory olfactory bulb of male rats. Front Neuroanat. 2012; 6:25.21. Hong W, Kennedy A, Burgos-Artizzu XP, Zelikowsky M, Navonne SG, Perona P, et al. Automated measurement of mouse social behaviors using depth sensing, video tracking, and machine learning. Proc Natl Acad Sci U S A. 2015; 112:E5351–60.22. Taguchi S, Choudhury ME, Miyanishi K, Nakanishi Y, Kameda K, Abe N, et al. Aggravating effects of treadmill exercises during the early-onset period in a rat traumatic brain injury model: when should rehabilitation exercises be initiated? IBRO Rep. 2019; 7:82–9.23. Chung HY, Kim DH, Lee EK, Chung KW, Chung S, Lee B, et al. Redefining chronic inflammation in aging and age-related diseases: proposal of the senoinflammation concept. Aging Dis. 2019; 10:367–82.24. Mattson MP. Hormesis and disease resistance: activation of cellular stress response pathways. Hum Exp Toxicol. 2008; 27:155–62.25. Steptoe A, Shankar A, Demakakos P, Wardle J. Social isolation, loneliness, and all-cause mortality in older men and women. Proc Natl Acad Sci U S A. 2013; 110:5797–801.26. Friedman A, Hueske E, Drammis SM, Toro Arana SE, Nelson ED, Carter CW, et al. Striosomes mediate value-based learning vulnerable in age and a Huntington’s disease model. Cell. 2020; 183:918–34.e49.27. Altun M, Bergman E, Edstrom E, Johnson H, Ulfhake B. Behavioral impairments of the aging rat. Physiol Behav. 2007; 92:911–23.28. Silva BA, Mattucci C, Krzywkowski P, Murana E, Illarionova A, Grinevich V, et al. Independent hypothalamic circuits for social and predator fear. Nat Neurosci. 2013; 16:1731–3.29. Kim TW, Lim BV, Kim K, Seo JH, Kim CJ. Treadmill exercise alleviates stress-induced impairment of social interaction through 5-hydroxytryptamine 1A receptor activation in rats. J Exerc Rehabil. 2015; 11:192–7.30. Wu T, Luo Y, Broster LS, Gu R, Luo YJ. The impact of anxiety on social decision-making: behavioral and electrodermal findings. Soc Neurosci. 2013; 8:11–21.31. Spruijt BM, Meyerson BJ, Hoglund U. Aging and sociosexual behavior in the male rat. Behav Brain Res. 1989; 32:51–61.32. Senba E, Ueyama T. Stress-induced expression of immediate early genes in the brain and peripheral organs of the rat. Neurosci Res. 1997; 29:183–207.33. Weinberg MS, Girotti M, Spencer RL. Restraint-induced fra-2 and c-fos expression in the rat forebrain: relationship to stress duration. Neuroscience. 2007; 150:478–86.34. Perkins AE, Woodruff ER, Chun LE, Spencer RL, Varlinskaya E, Deak T. Analysis of c-Fos induction in response to social interaction in male and female Fisher 344 rats. Brain Res. 2017; 1672:113–21.35. Bland ST, Schmid MJ, Der-Avakian A, Watkins LR, Spencer RL, Maier SF. Expression of c-fos and BDNF mRNA in subregions of the prefrontal cortex of male and female rats after acute uncontrollable stress. Brain Res. 2005; 1051:90–9.36. Seo JH, Kim TW, Kim CJ, Sung YH, Lee SJ. Treadmill exercise during pregnancy ameliorates post-traumatic stress disorder-induced anxiety-like responses in maternal rats. Mol Med Rep. 2013; 7:389–95.37. Kubik S, Miyashita T, Guzowski JF. Using immediate-early genes to map hippocampal subregional functions. Learn Mem. 2007; 14:758–70.38. Smith MA. Hippocampal vulnerability to stress and aging: possible role of neurotrophic factors. Behav Brain Res. 1996; 78:25–36.39. Droste SK, Gesing A, Ulbricht S, Muller MB, Linthorst AC, Reul JM. Effects of long-term voluntary exercise on the mouse hypothalamic-pituitary-adrenocortical axis. Endocrinology. 2003; 144:3012–23.40. Hare BD, Beierle JA, Toufexis DJ, Hammack SE, Falls WA. Exercise-associated changes in the corticosterone response to acute restraint stress: evidence for increased adrenal sensitivity and reduced corticosterone response duration. Neuropsychopharmacology. 2014; 39:1262–9.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Relationships Between Exercise Components and Social Anxiety Levels Among Chinese College Students
- Oxytocin and Vasopressin Levels and Related Factors in Adolescents with Social Phobia and Other Anxiety Disorders
- Postnatal Treadmill Exercise Alleviates Prenatal Stress-Induced Anxiety in Offspring Rats by Enhancing Cell Proliferation Through 5-Hydroxytryptamine 1A Receptor Activation
- Physical Exercise Counteracts Stress-induced Upregulation of Melanin-concentrating Hormone in the Brain and Stress-induced Persisting Anxiety-like Behaviors
- Effect of Cognitive-Behavioral Treatment in Children with Anxiety Disorder: A Preliminary Study