Endocrinol Metab.
2022 Aug;37(4):630-640. 10.3803/EnM.2022.1430.
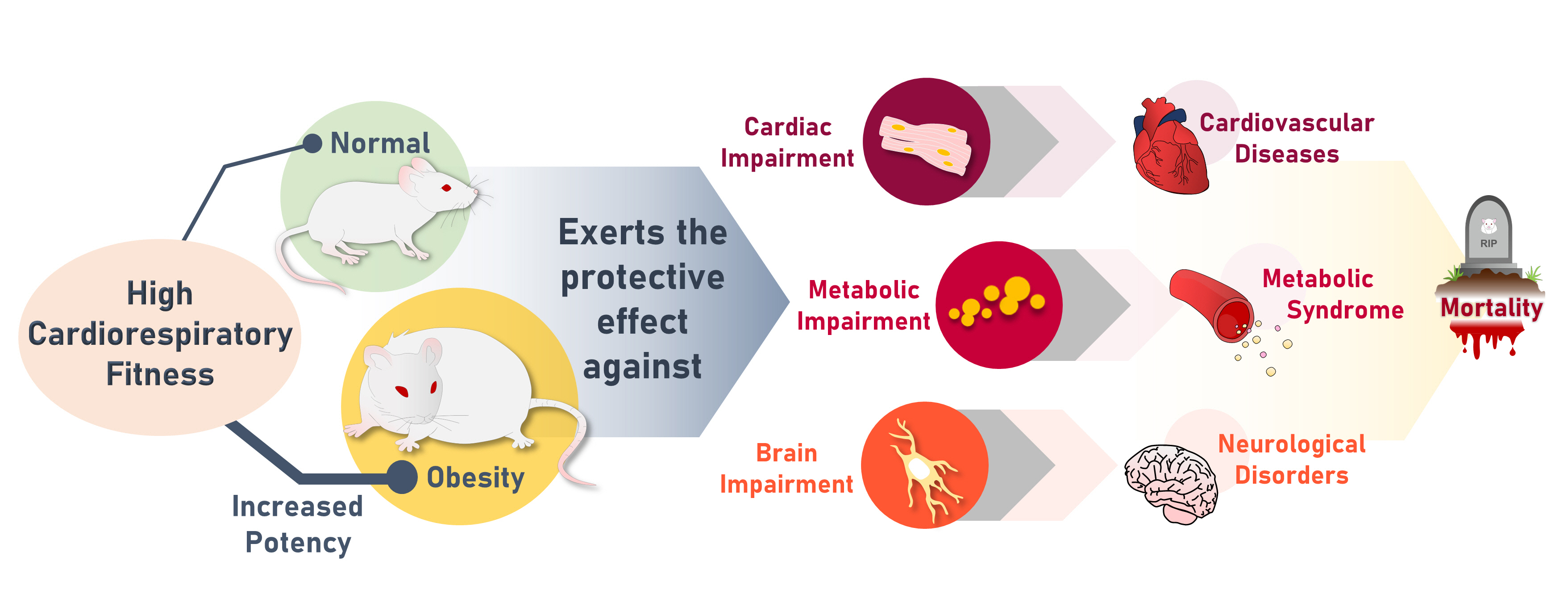
High Cardiorespiratory Fitness Protects against Molecular Impairments of Metabolism, Heart, and Brain with Higher Efficacy in Obesity-Induced Premature Aging
- Affiliations
-
- 1Neurophysiology Unit, Cardiac Electrophysiology Research and Training Center, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 2Cardiac Electrophysiology Unit, Department of Physiology, Faculty of Medicine, Chiang Mai University, Chiang Mai, Thailand
- 3Center of Excellence in Cardiac Electrophysiology Research, Chiang Mai University, Chiang Mai, Thailand
- 4Department of Oral Biology and Diagnostic Sciences, Faculty of Dentistry, Chiang Mai University, Chiang Mai, Thailand
- KMID: 2532863
- DOI: http://doi.org/10.3803/EnM.2022.1430
Abstract
- Background
High cardiorespiratory fitness (CRF) protects against age-related diseases. However, the mechanisms mediating the protective effect of high intrinsic CRF against metabolic, cardiac, and brain impairments in non-obese versus obese conditions remain incompletely understood. We aimed to identify the mechanisms through which high intrinsic CRF protects against metabolic, cardiac, and brain impairments in non-obese versus obese untrained rats.
Methods
Seven-week-old male Wistar rats were divided into two groups (n=8 per group) to receive either a normal diet or a highfat diet (HFD). At weeks 12 and 28, CRF, carbohydrate and fatty acid oxidation, cardiac function, and metabolic parameters were evaluated. At week 28, behavior tests were performed. At the end of week 28, rats were euthanized to collect heart and brain samples for molecular studies.
Results
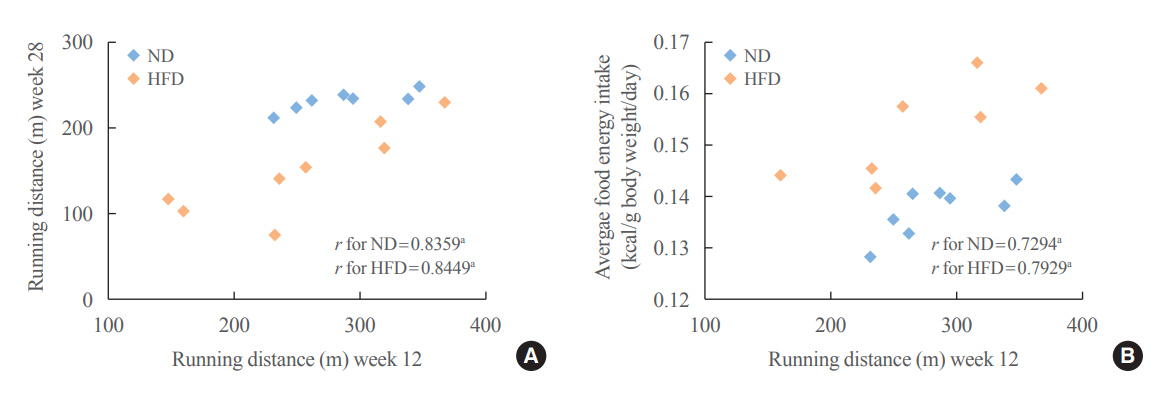
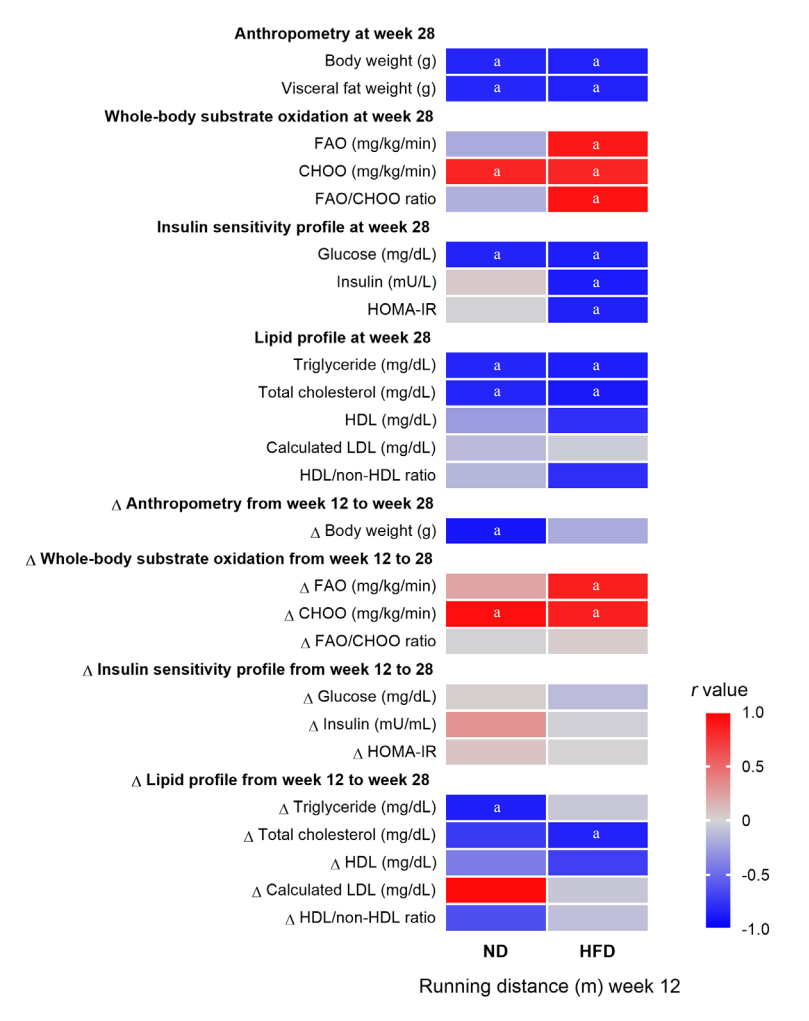
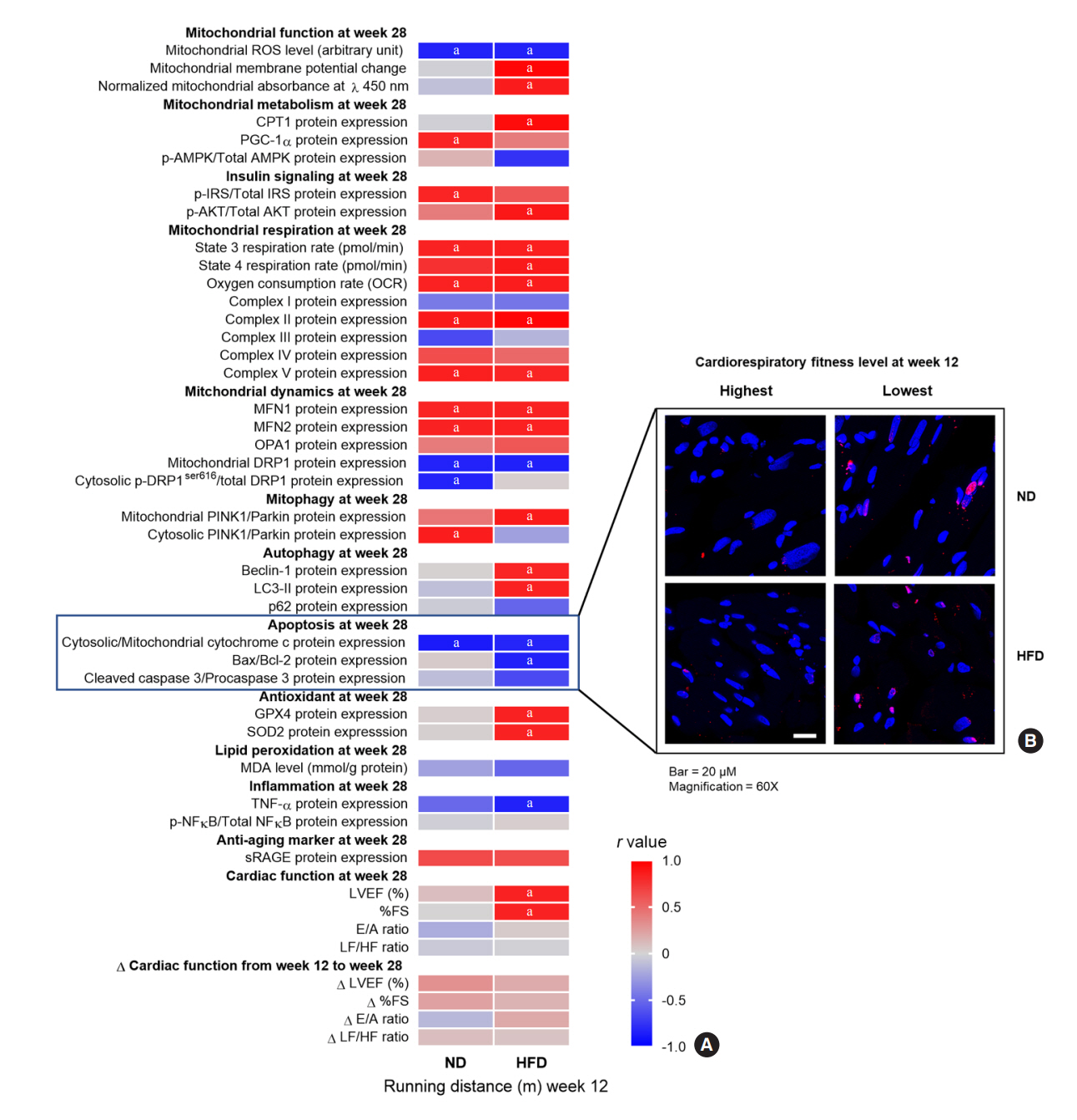
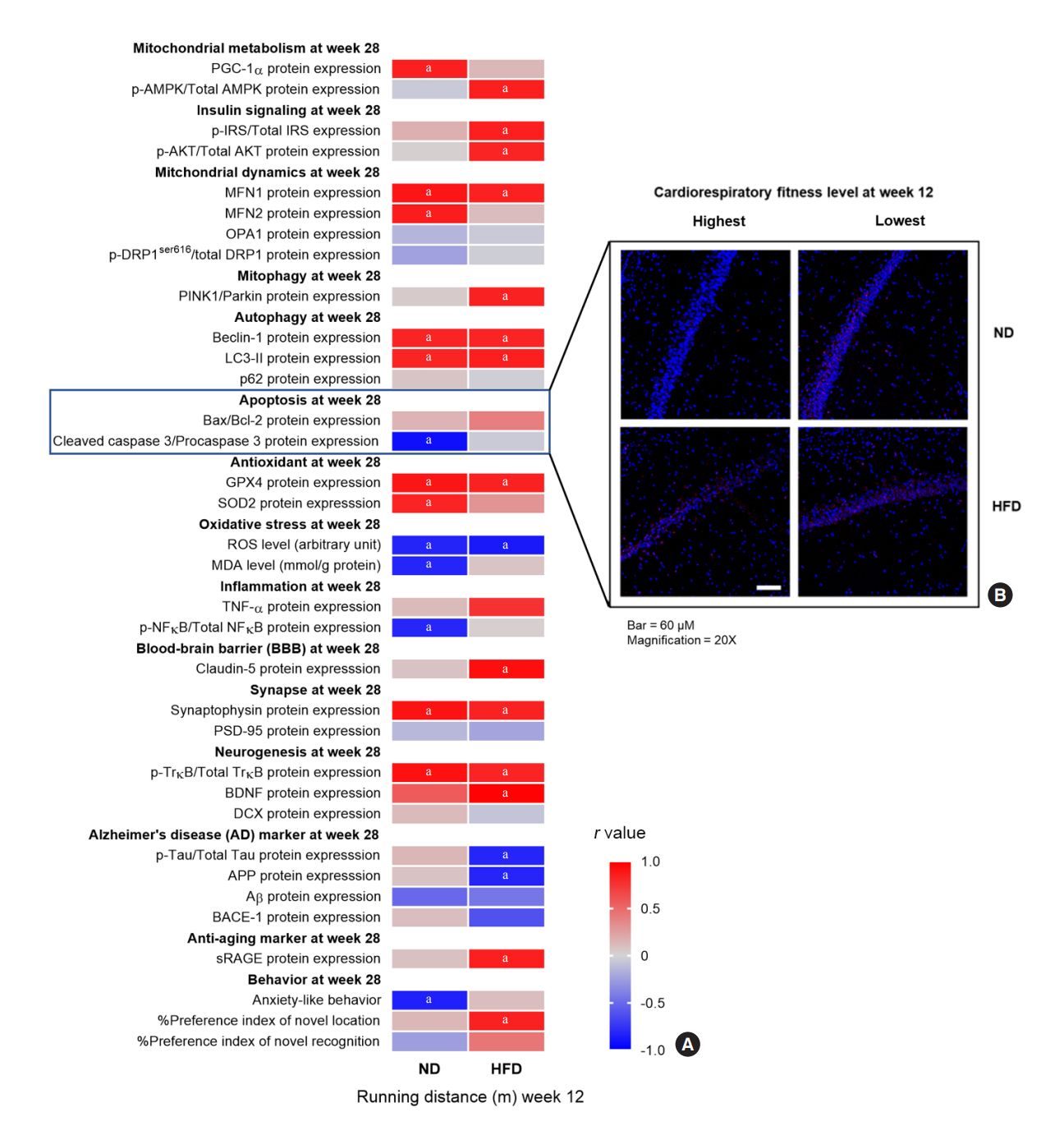
The obese rats exhibited higher values for aging-related parameters than the non-obese rats, indicating that they experienced obesity-induced premature aging. High baseline CRF levels were positively correlated with several favorable metabolic, cardiac, and brain parameters at follow-up. Specifically, the protective effects of high CRF against metabolic, cardiac, and brain impairments were mediated by the modulation of body weight and composition, the lipid profile, substrate oxidation, mitochondrial function, insulin signaling, autophagy, apoptosis, inflammation, oxidative stress, cardiac function, neurogenesis, blood-brain barrier, synaptic function, accumulation of Alzheimer’s disease-related proteins, and cognition. Interestingly, this effect was more obvious in HFD-fed rats.
Conclusion
The protective effect of high CRF is mediated by the modulation of several mechanisms. These effects exhibit greater efficacy under conditions of obesity-induced premature aging.
Keyword
Figure
Reference
-
1. Lee DC, Artero EG, Sui X, Blair SN. Mortality trends in the general population: the importance of cardiorespiratory fitness. J Psychopharmacol. 2010; 24(4 Suppl):27–35.
Article2. Schutte NM, Nederend I, Hudziak JJ, Bartels M, de Geus EJ. Twin-sibling study and meta-analysis on the heritability of maximal oxygen consumption. Physiol Genomics. 2016; 48:210–9.
Article3. Vidoni ED, Honea RA, Billinger SA, Swerdlow RH, Burns JM. Cardiorespiratory fitness is associated with atrophy in Alzheimer’s and aging over 2 years. Neurobiol Aging. 2012; 33:1624–32.
Article4. Liu R, Sui X, Laditka JN, Church TS, Colabianchi N, Hussey J, et al. Cardiorespiratory fitness as a predictor of dementia mortality in men and women. Med Sci Sports Exerc. 2012; 44:253–9.
Article5. Kodama S, Saito K, Tanaka S, Maki M, Yachi Y, Asumi M, et al. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women: a meta-analysis. JAMA. 2009; 301:2024–35.
Article6. Koch LG, Britton SL. Artificial selection for intrinsic aerobic endurance running capacity in rats. Physiol Genomics. 2001; 5:45–52.
Article7. Wisloff U, Najjar SM, Ellingsen O, Haram PM, Swoap S, Al-Share Q, et al. Cardiovascular risk factors emerge after artificial selection for low aerobic capacity. Science. 2005; 307:418–20.
Article8. Rivas DA, Lessard SJ, Saito M, Friedhuber AM, Koch LG, Britton SL, et al. Low intrinsic running capacity is associated with reduced skeletal muscle substrate oxidation and lower mitochondrial content in white skeletal muscle. Am J Physiol Regul Integr Comp Physiol. 2011; 300:R835–43.
Article9. Crissey JM, Padilla J, Vieira-Potter VJ, Thorne PK, Koch LG, Britton SL, et al. Divergent role of nitric oxide in insulin-stimulated aortic vasorelaxation between low- and high-intrinsic aerobic capacity rats. Physiol Rep. 2015; 3:e12459.
Article10. Gavini CK, Mukherjee S, Shukla C, Britton SL, Koch LG, Shi H, et al. Leanness and heightened nonresting energy expenditure: role of skeletal muscle activity thermogenesis. Am J Physiol Endocrinol Metab. 2014; 306:E635–47.
Article11. Overmyer KA, Evans CR, Qi NR, Minogue CE, Carson JJ, Chermside-Scabbo CJ, et al. Maximal oxidative capacity during exercise is associated with skeletal muscle fuel selection and dynamic changes in mitochondrial protein acetylation. Cell Metab. 2015; 21:468–78.
Article12. Salvestrini V, Sell C, Lorenzini A. Obesity may accelerate the aging process. Front Endocrinol (Lausanne). 2019; 10:266.
Article13. Hussain SO, Barbato JC, Koch LG, Metting PJ, Britton SL. Cardiac function in rats selectively bred for low- and high-capacity running. Am J Physiol Regul Integr Comp Physiol. 2001; 281:R1787–91.
Article14. Koch LG, Kemi OJ, Qi N, Leng SX, Bijma P, Gilligan LJ, et al. Intrinsic aerobic capacity sets a divide for aging and longevity. Circ Res. 2011; 109:1162–72.
Article15. Burghardt PR, Flagel SB, Burghardt KJ, Britton SL, GerardKoch L, Watson SJ, et al. Risk-assessment and coping strategies segregate with divergent intrinsic aerobic capacity in rats. Neuropsychopharmacology. 2011; 36:390–401.
Article16. Choi J, Chandrasekaran K, Demarest TG, Kristian T, Xu S, Vijaykumar K, et al. Brain diabetic neurodegeneration segregates with low intrinsic aerobic capacity. Ann Clin Transl Neurol. 2014; 1:589–604.
Article17. Dai DF, Rabinovitch PS, Ungvari Z. Mitochondria and cardiovascular aging. Circ Res. 2012; 110:1109–24.
Article18. Bonomini F, Rodella LF, Rezzani R. Metabolic syndrome, aging and involvement of oxidative stress. Aging Dis. 2015; 6:109–20.
Article19. Grimm A, Eckert A. Brain aging and neurodegeneration: from a mitochondrial point of view. J Neurochem. 2017; 143:418–31.
Article20. Thorpe RJ Jr, Ferraro KF. Aging, obesity, and mortality: misplaced concern about obese older people? Res Aging. 2004; 26:108–29.21. Tam BT, Morais JA, Santosa S. Obesity and ageing: two sides of the same coin. Obes Rev. 2020; 21:e12991.
Article22. Sengupta P. The laboratory rat: relating its age with human’s. Int J Prev Med. 2013; 4:624–30.23. Morris EM, Jackman MR, Johnson GC, Liu TW, Lopez JL, Kearney ML, et al. Intrinsic aerobic capacity impacts susceptibility to acute high-fat diet-induced hepatic steatosis. Am J Physiol Endocrinol Metab. 2014; 307:E355–64.24. Thyfault JP, Morris EM. Intrinsic (genetic) aerobic fitness impacts susceptibility for metabolic disease. Exerc Sport Sci Rev. 2017; 45:7–15.
Article25. Harman SM, Blackman MR. The effects of growth hormone and sex steroid on lean body mass, fat mass, muscle strength, cardiovascular endurance and adverse events in healthy elderly women and men. Horm Res. 2003; 60(Suppl 1):121–4.
Article26. Brown M. Skeletal muscle and bone: effect of sex steroids and aging. Adv Physiol Educ. 2008; 32:120–6.
Article27. Zeiher J, Ombrellaro KJ, Perumal N, Keil T, Mensink G, Finger JD. Correlates and determinants of cardiorespiratory fitness in adults: a systematic review. Sports Med Open. 2019; 5:39.
Article28. Chockalingam A, Linden MA, Del Rosario M, Govindarajan G, Dellsperger KC, Thomas TR. Exercise and weight loss improve exercise capacity independent of cardiac function in metabolic syndrome. Angiology. 2010; 61:192–7.
Article29. Lin X, Zhang X, Guo J, Roberts CK, McKenzie S, Wu WC, et al. Effects of exercise training on cardiorespiratory fitness and biomarkers of cardiometabolic health: a systematic review and meta-analysis of randomized controlled trials. J Am Heart Assoc. 2015; 4:e002014.
Article30. Frimpong E, Dafkin C, Donaldson J, Millen A, Meiring RM. The effect of home-based low-volume, high-intensity interval training on cardiorespiratory fitness, body composition and cardiometabolic health in women of normal body mass and those with overweight or obesity: protocol for a randomized controlled trial. BMC Sports Sci Med Rehabil. 2019; 11:39.
Article31. American College of Sports Medicine Position Stand. The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med Sci Sports Exerc. 1998; 30:975–91.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- How Does Obesity and Physical Activity Affect Aging?: Focused on Telomere as a Biomarker of Aging
- Association of Cardiorespiratory Fitness with Insulin Resistance, Blood Lipids and Bone Mineral Density in Young Female Adults
- Obesity Phenotypes, Lifestyle Medicine, and Population Health: Precision Needed Everywhere!
- Correlation of Sit-to-Stand Test and 6-Minute Walk Test to Illustrate Cardiorespiratory Fitness in Systolic Heart Failure Patients
- Association between Estimated Cardiorespiratory Fitness and Insulin Resistance in Korean Adults: Results from the Korea National Health and Nutrition Survey 2019–2021