Cancer Res Treat.
2022 Jul;54(3):744-752. 10.4143/crt.2021.772.
Predictive Value of Interstitial Lung Abnormalities for Postoperative Pulmonary Complications in Elderly Patients with Early-stage Lung Cancer
- Affiliations
-
- 1Lung and Esophageal Cancer Clinic, Chonnam National University, Hwasun Hospital, Hwasun, Korea
- 2Department of Radiology, Chonnam National University Medical School, Chonnam National University Hospital, Gwangju, Korea
- 3Department of Internal Medicine, Chonnam National University Medical School, Chonnam National University Hospital, Gwangju, Korea
- 4Department of Thoracic and Cardiovascular Surgery, Chonnam National University Medical School, Chonnam National University Hospital, Gwangju, Korea
- 5Department of Radiology, Research Institute of Clinical Medicine of Jeonbuk National University–Biomedical Research Institute of Jeonbuk National University Hospital, Jeonju, Korea
- KMID: 2531321
- DOI: http://doi.org/10.4143/crt.2021.772
Abstract
- Purpose
Identifying pretreatment interstitial lung abnormalities (ILAs) is important because of their predictive value for complications after lung cancer treatment. This study aimed to assess the predictive value of ILAs for postoperative pulmonary complications (PPCs) in elderly patients undergoing curative resection for early-stage non-small cell lung cancer (NSCLC).
Materials and Methods
Elderly patients (age ≥ 70 years) who underwent curative resection for pathologic stage I or II NSCLC with normal preoperative spirometry results (pre-bronchodilator forced expiratory volume in 1 s to forced vital capacity [FVC] ratio > 0.70 and FVC ≥ 80% of the predicted value) between January 2012 and December 2019 were retrospectively identified. Univariable and multivariable regression analyses were performed to assess risk factors for PPCs. The Kaplan–Meier method and log-rank test were used to analyze the relationship between ILAs and postoperative mortality. One-way analysis of variance was performed to assess the correlation between ILAs and hospital stay duration.
Results
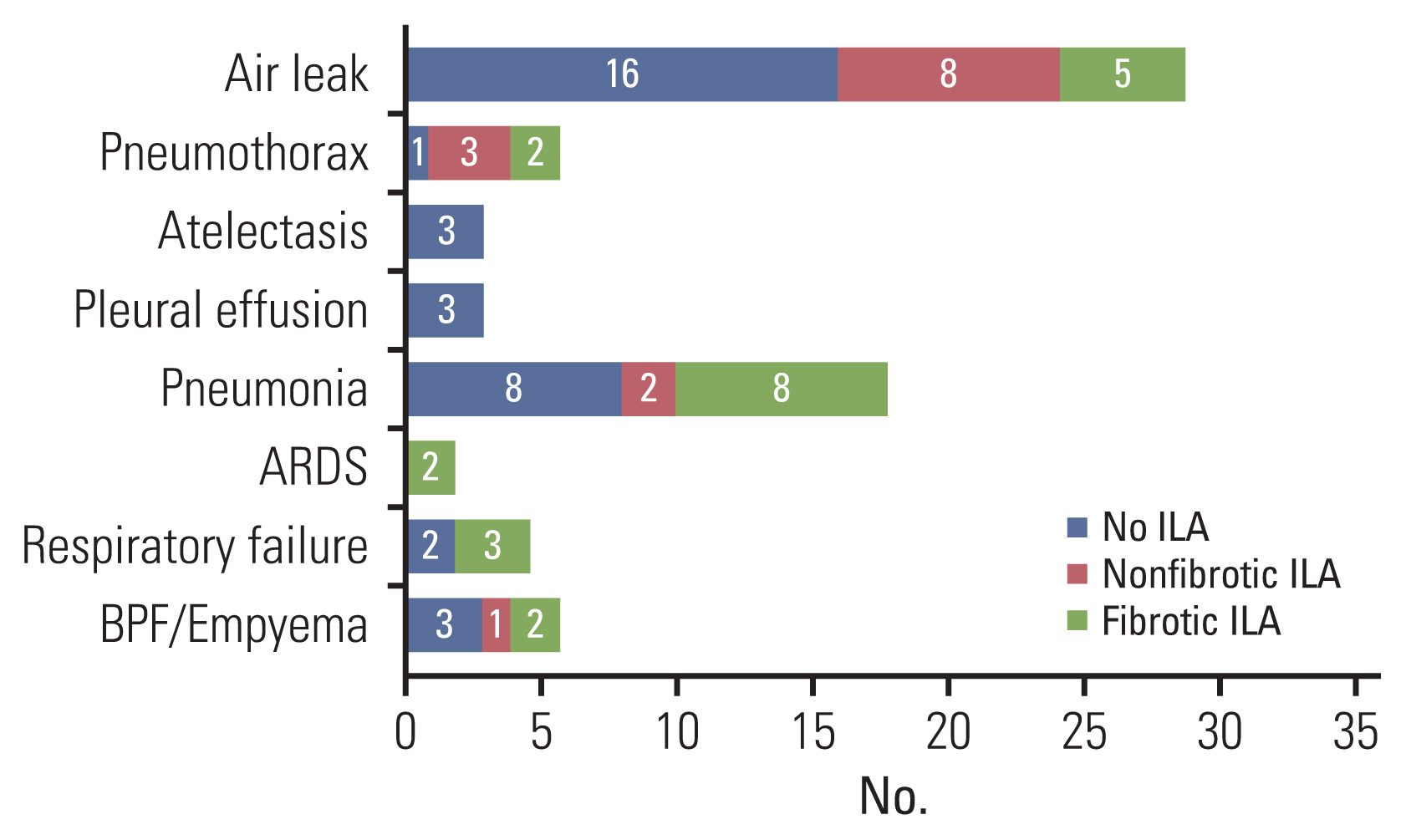
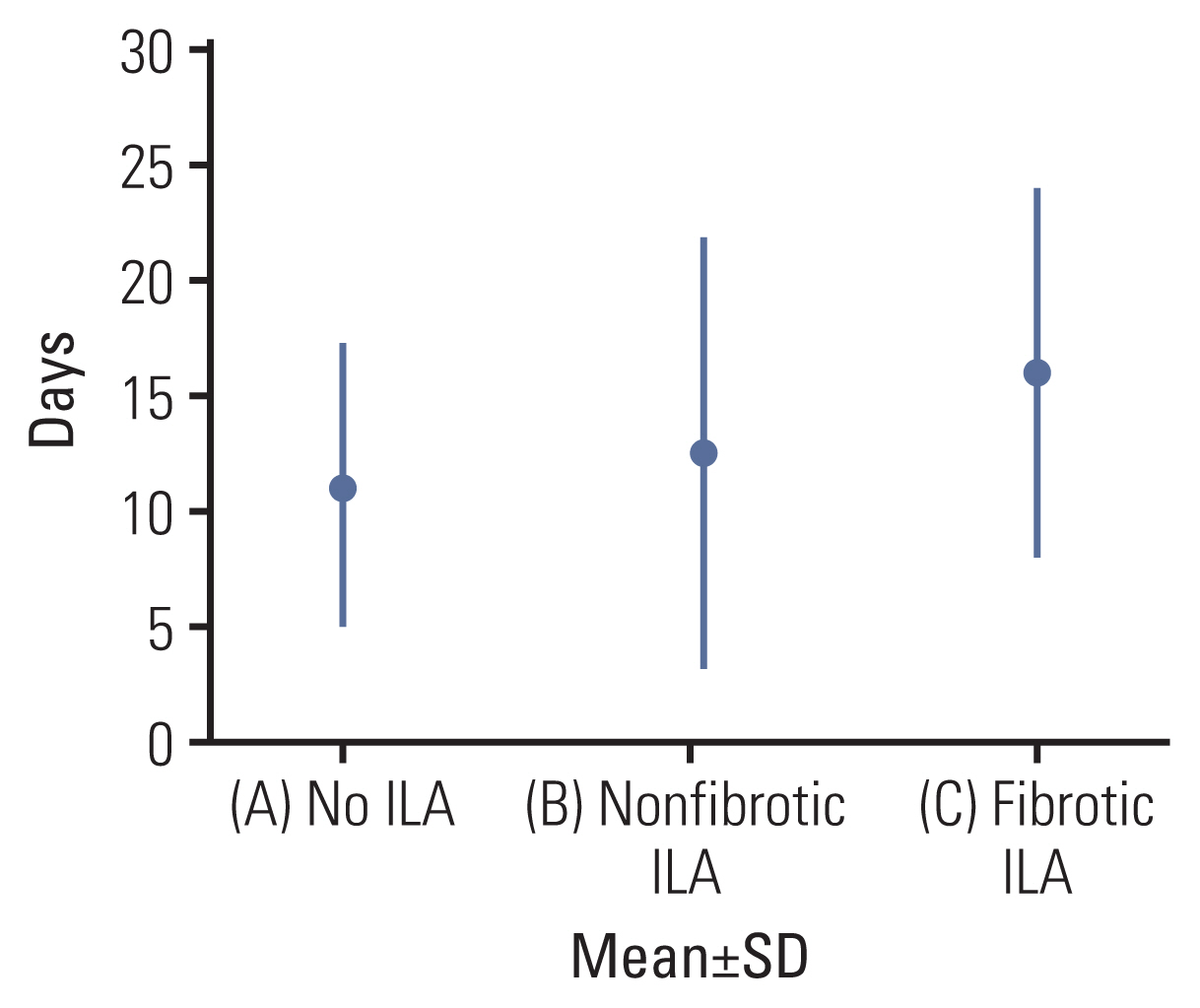
A total of 262 patients (median age, 73 [interquartile range, 71–76] years; 132 male) were evaluated. A multivariable logistic regression model revealed that, among several relevant risk factors, fibrotic ILAs independently predicted both overall PPCs (adjusted odds ratio [OR], 4.84; 95% confidence interval [CI], 1.35–17.38; p=0.016) and major PPCs (adjusted OR, 8.72; 95% CI, 1.71–44.38; p=0.009). Fibrotic ILAs were significantly associated with higher postoperative mortality and longer hospital stay (F=5.21, p=0.006).
Conclusion
Pretreatment fibrotic ILAs are associated with PPCs, higher postoperative mortality, and longer hospital stay.
Keyword
Figure
Reference
-
References
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2020. CA Cancer J Clin. 2020; 70:7–30.
Article2. Hong S, Won YJ, Lee JJ, Jung KW, Kong HJ, Im JS, et al. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2018. Cancer Res Treat. 2021; 53:301–15.
Article3. Howlader N, Noone AM, Krapcho M, Miller D, Brest A, Yu M, et al. SEER cancer statistics review, 1975–2017. Bethesda, MD: National Cancer Institute;2020.4. Kamel MK, Port JL. Oncologic considerations in the elderly. Curr Opin Anaesthesiol. 2018; 31:6–10.
Article5. de Koning HJ, van der Aalst CM, de Jong PA, Scholten ET, Nackaerts K, Heuvelmans MA, et al. Reduced lung-cancer mortality with volume CT screening in a randomized trial. N Engl J Med. 2020; 382:503–13.
Article6. Rueth NM, Parsons HM, Habermann EB, Groth SS, Virnig BA, Tuttle TM, et al. Surgical treatment of lung cancer: predicting postoperative morbidity in the elderly population. J Thorac Cardiovasc Surg. 2012; 143:1314–23.
Article7. Agostini P, Cieslik H, Rathinam S, Bishay E, Kalkat MS, Rajesh PB, et al. Postoperative pulmonary complications following thoracic surgery: are there any modifiable risk factors? Thorax. 2010; 65:815–8.
Article8. Brunelli A, Charloux A, Bolliger CT, Rocco G, Sculier JP, Varela G, et al. ERS/ESTS clinical guidelines on fitness for radical therapy in lung cancer patients (surgery and chemo-radiotherapy). Eur Respir J. 2009; 34:17–41.
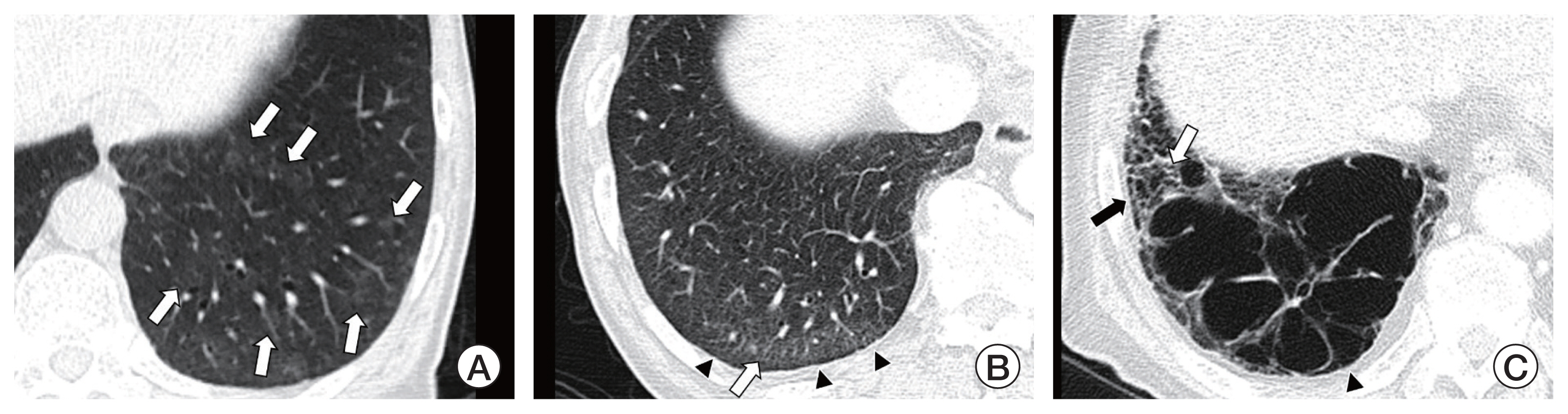
Article9. Hatabu H, Hunninghake GM, Richeldi L, Brown KK, Wells AU, Remy-Jardin M, et al. Interstitial lung abnormalities detected incidentally on CT: a Position Paper from the Fleischner Society. Lancet Respir Med. 2020; 8:726–37.
Article10. Whittaker Brown SA, Padilla M, Mhango G, Powell C, Salvatore M, Henschke C, et al. Interstitial lung abnormalities and lung cancer risk in the National Lung Screening Trial. Chest. 2019; 156:1195–203.
Article11. Nishino M, Cardarella S, Dahlberg SE, Araki T, Lydon C, Jackman DM, et al. Interstitial lung abnormalities in treatment-naive advanced non-small-cell lung cancer patients are associated with shorter survival. Eur J Radiol. 2015; 84:998–1004.
Article12. Yamaguchi S, Ohguri T, Ide S, Aoki T, Imada H, Yahara K, et al. Stereotactic body radiotherapy for lung tumors in patients with subclinical interstitial lung disease: the potential risk of extensive radiation pneumonitis. Lung Cancer. 2013; 82:260–5.
Article13. Hino T, Lee KS, Han J, Hata A, Ishigami K, Hatabu H. Spectrum of pulmonary fibrosis from interstitial lung abnormality to usual interstitial pneumonia: importance of identification and quantification of traction bronchiectasis in patient management. Korean J Radiol. 2021; 22:811–28.
Article14. Miskovic A, Lumb AB. Postoperative pulmonary complications. Br J Anaesth. 2017; 118:317–34.
Article15. Pei G, Zhou S, Han Y, Liu Z, Xu S. Risk factors for postoperative complications after lung resection for non-small cell lung cancer in elderly patients at a single institution in China. J Thorac Dis. 2014; 6:1230–8.16. Putman RK, Hatabu H, Araki T, Gudmundsson G, Gao W, Nishino M, et al. Association between interstitial lung abnormalities and all-cause mortality. JAMA. 2016; 315:672–81.17. Hatabu H, Hunninghake GM, Lynch DA. Interstitial lung abnormality: recognition and perspectives. Radiology. 2019; 291:1–3.
Article18. Jin GY, Lynch D, Chawla A, Garg K, Tammemagi MC, Sahin H, et al. Interstitial lung abnormalities in a CT lung cancer screening population: prevalence and progression rate. Radiology. 2013; 268:563–71.
Article19. Putman RK, Gudmundsson G, Axelsson GT, Hida T, Honda O, Araki T, et al. Imaging patterns are associated with interstitial lung abnormality progression and mortality. Am J Respir Crit Care Med. 2019; 200:175–83.
Article20. Iwasawa T, Okudela K, Takemura T, Fukuda T, Matsushita S, Baba T, et al. Computer-aided quantification of pulmonary fibrosis in patients with lung cancer: relationship to disease-free survival. Radiology. 2019; 292:489–98.
Article21. Im Y, Park HY, Shin S, Shin SH, Lee H, Ahn JH, et al. Prevalence of and risk factors for pulmonary complications after curative resection in otherwise healthy elderly patients with early stage lung cancer. Respir Res. 2019; 20:136.
Article22. Patel NM, Kulkarni T, Dilling D, Scholand MB. Interstitial, Diffuse Lung Disease Network Steering Committee. Preoperative evaluation of patients with interstitial lung disease. Chest. 2019; 156:826–33.
Article23. Chae KJ, Jin GY, Goo JM, Chung MJ. Interstitial lung abnormalities: what radiologists should know. Korean J Radiol. 2021; 22:454–63.
Article24. Miller ER, Putman RK, Vivero M, Hung Y, Araki T, Nishino M, et al. Histopathology of interstitial lung abnormalities in the context of lung nodule resections. Am J Respir Crit Care Med. 2018; 197:955–8.
Article25. McMillan RR, Berger A, Sima CS, Lou F, Dycoco J, Rusch V, et al. Thirty-day mortality underestimates the risk of early death after major resections for thoracic malignancies. Ann Thorac Surg. 2014; 98:1769–74.
Article26. Naccache JM, Gibiot Q, Monnet I, Antoine M, Wislez M, Chouaid C, et al. Lung cancer and interstitial lung disease: a literature review. J Thorac Dis. 2018; 10:3829–44.
Article27. von Meyenfeldt EM, Marres GM, van Thiel E, Damhuis RAM. Variation in length of hospital stay after lung cancer surgery in the Netherlands. Eur J Cardiothorac Surg. 2018; 54:560–4.
Article28. Towe CW, Khil A, Ho VP, Perry Y, Argote-Greene L, Wu KM, et al. Early discharge after lung resection is safe: 10-year experience. J Thorac Dis. 2018; 10:5870–8.
Article29. Fiore JF Jr, Bejjani J, Conrad K, Niculiseanu P, Landry T, Lee L, et al. Systematic review of the influence of enhanced recovery pathways in elective lung resection. J Thorac Cardiovasc Surg. 2016; 151:708–15.
Article30. Chen A, Karwoski RA, Gierada DS, Bartholmai BJ, Koo CW. Quantitative CT analysis of diffuse lung disease. Radiographics. 2020; 40:28–43.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Evaluation of Postoperative Acute Respiratory Failure in Primary Lung Cancer with Interstitial Pneumonia
- Reported Lung Adenocarcinoma with Idiopathic Pulmonary Fibrosis after Open Lung Biopsy
- Validation of Prediction Models for Postoperative Pulmonary Complications after Lung Resection: A Retrospective Study
- Chronic Obstructive Pulmonary Disease Combined with Interstitial Lung Disease
- A Case of Non-Specific Interstitial Pneumonia Associated with Primary Lung Adenocarcinoma