Korean J Physiol Pharmacol.
2022 Jul;26(4):239-253. 10.4196/kjpp.2022.26.4.239.
MicroRNA-21 promotes epithelial-mesenchymal transition and migration of human bronchial epithelial cells by targeting poly (ADP-ribose) polymerase-1 and activating PI3K/AKT signaling
- Affiliations
-
- 1Department of The Second Clinical College, Dalian Medical University, Dalian 116000, China
- 2Department of Respiratory and Critical Care Medicine, The Affiliated Changzhou No.2 People’s Hospital of Nanjing Medical University, Changzhou 213003, China
- KMID: 2530943
- DOI: http://doi.org/10.4196/kjpp.2022.26.4.239
Abstract
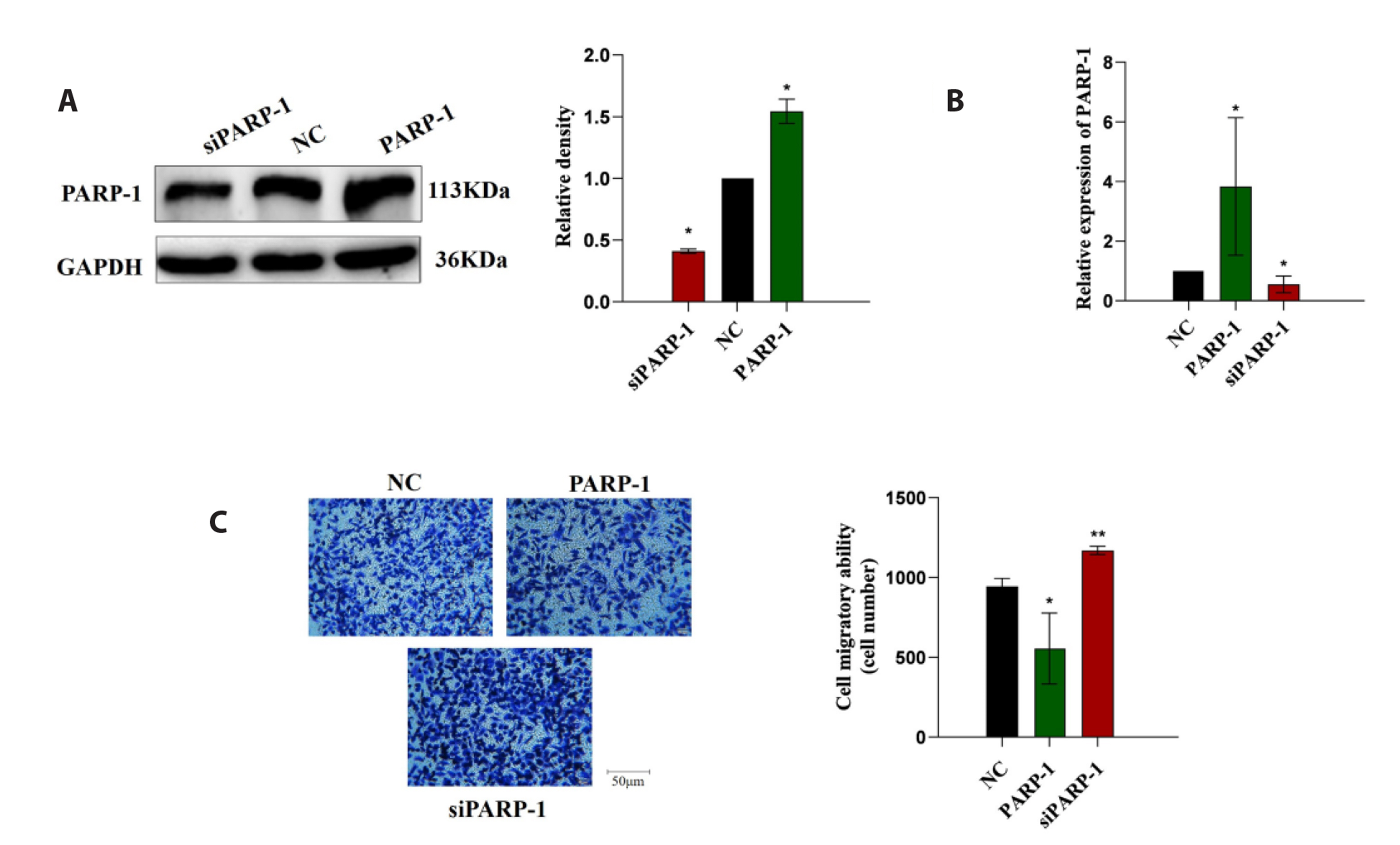
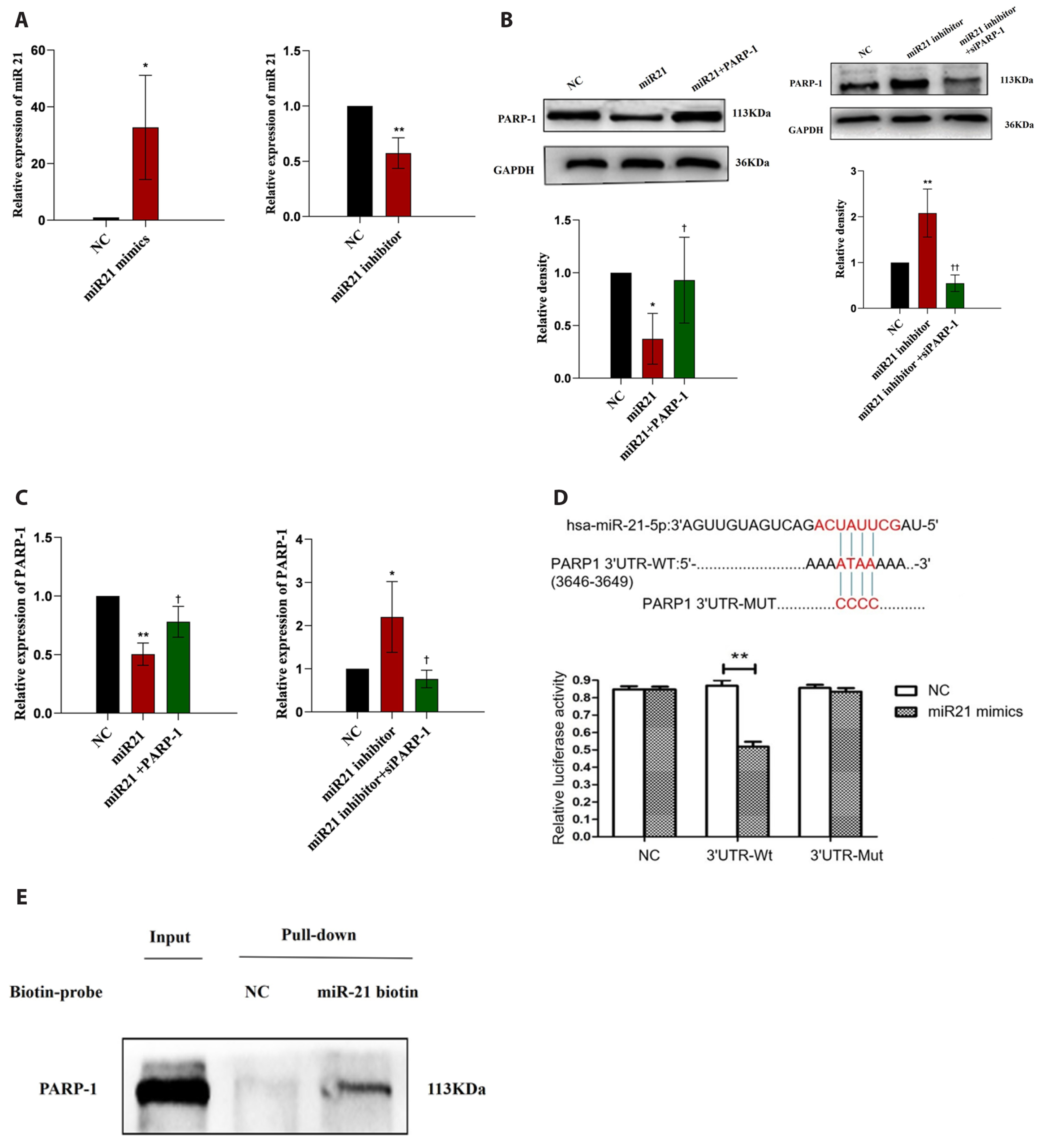
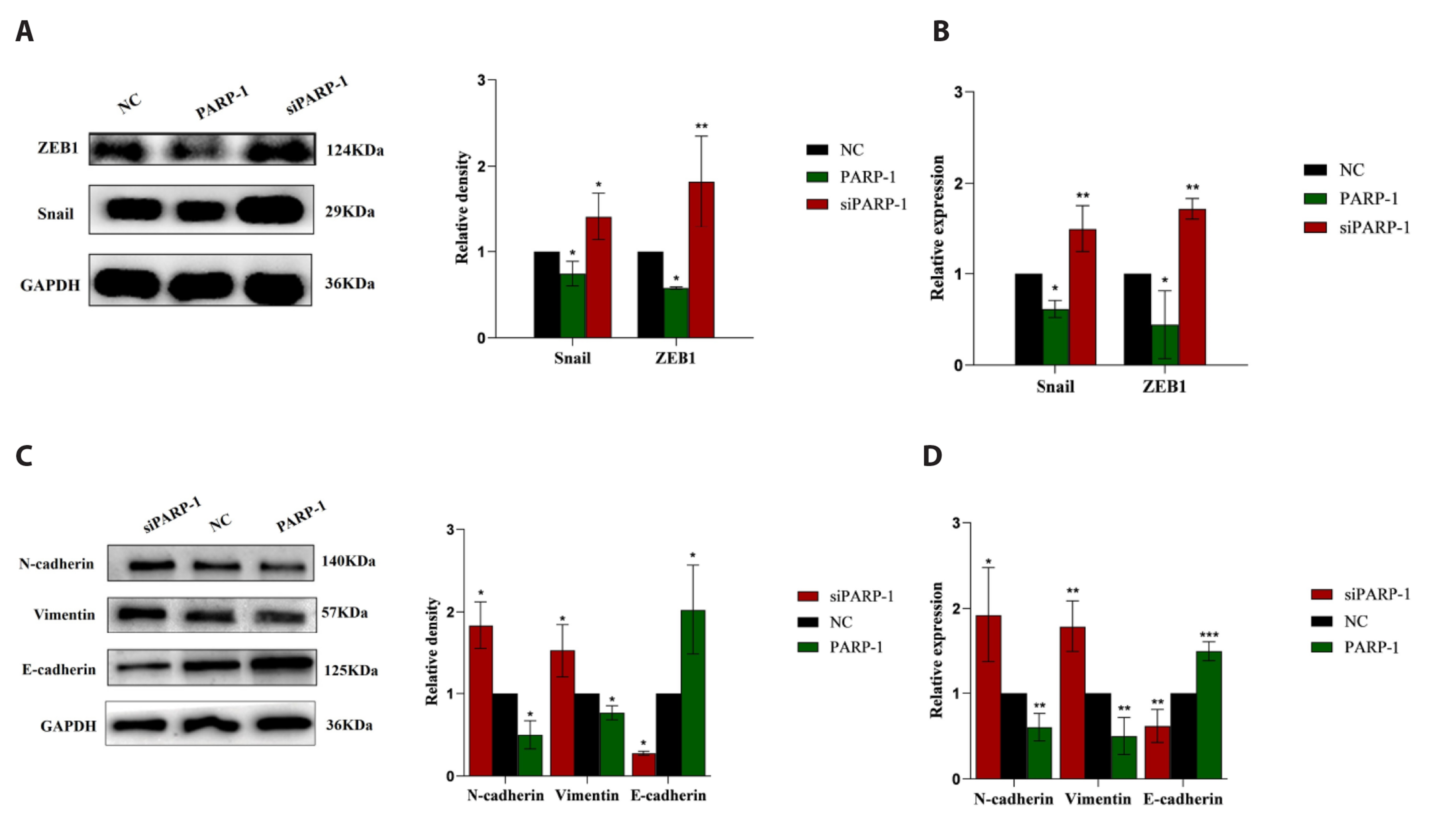
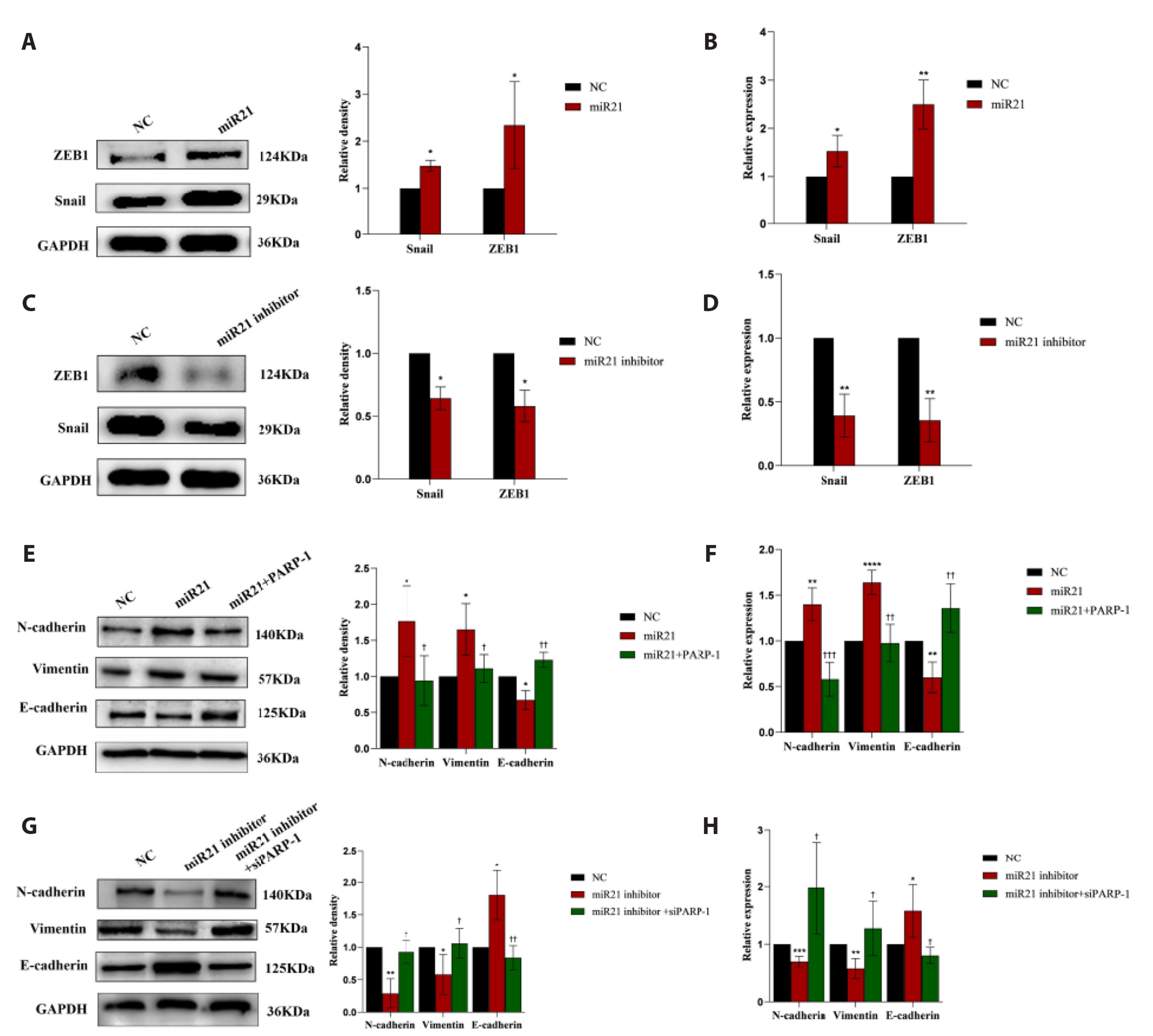
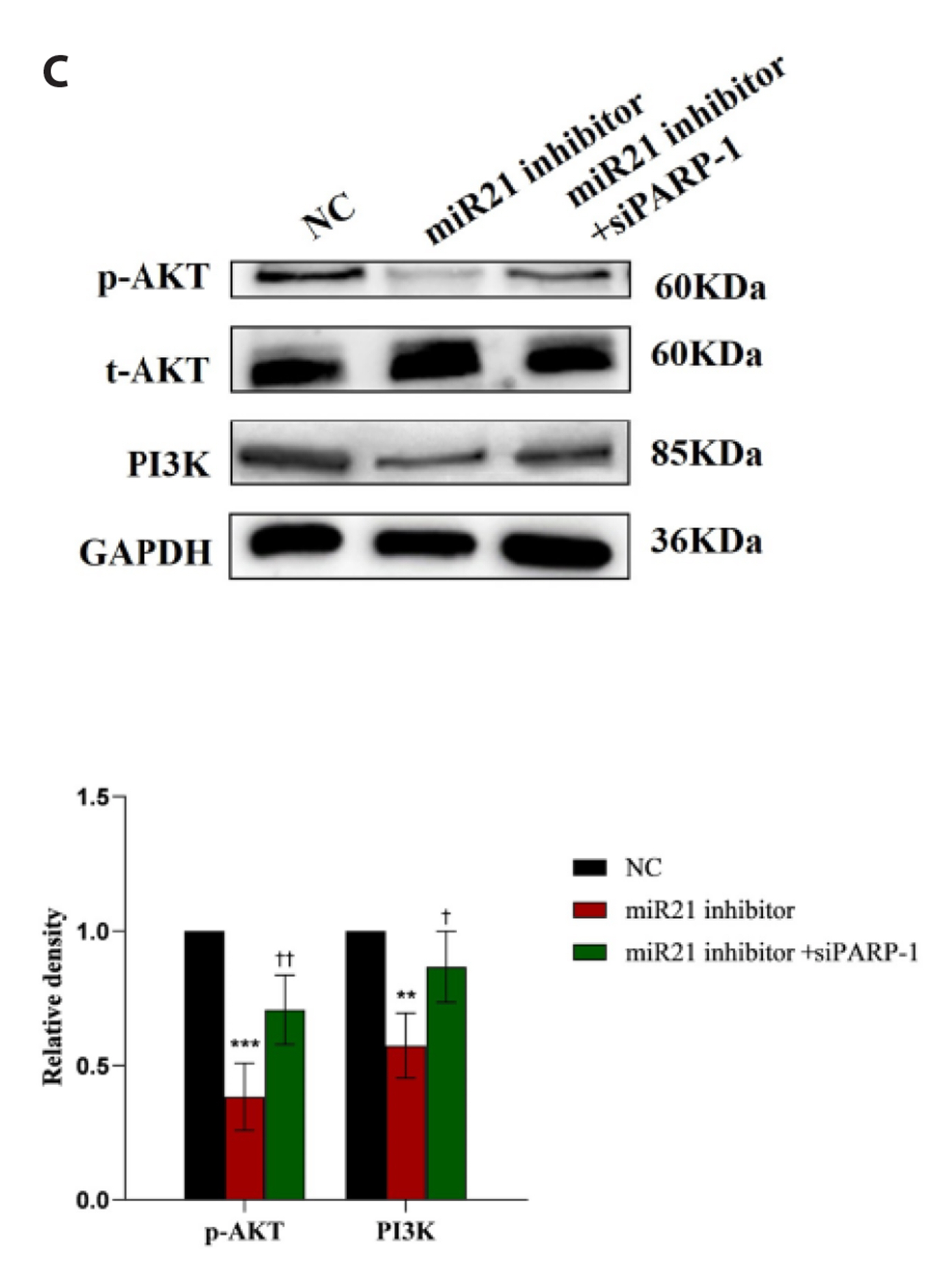
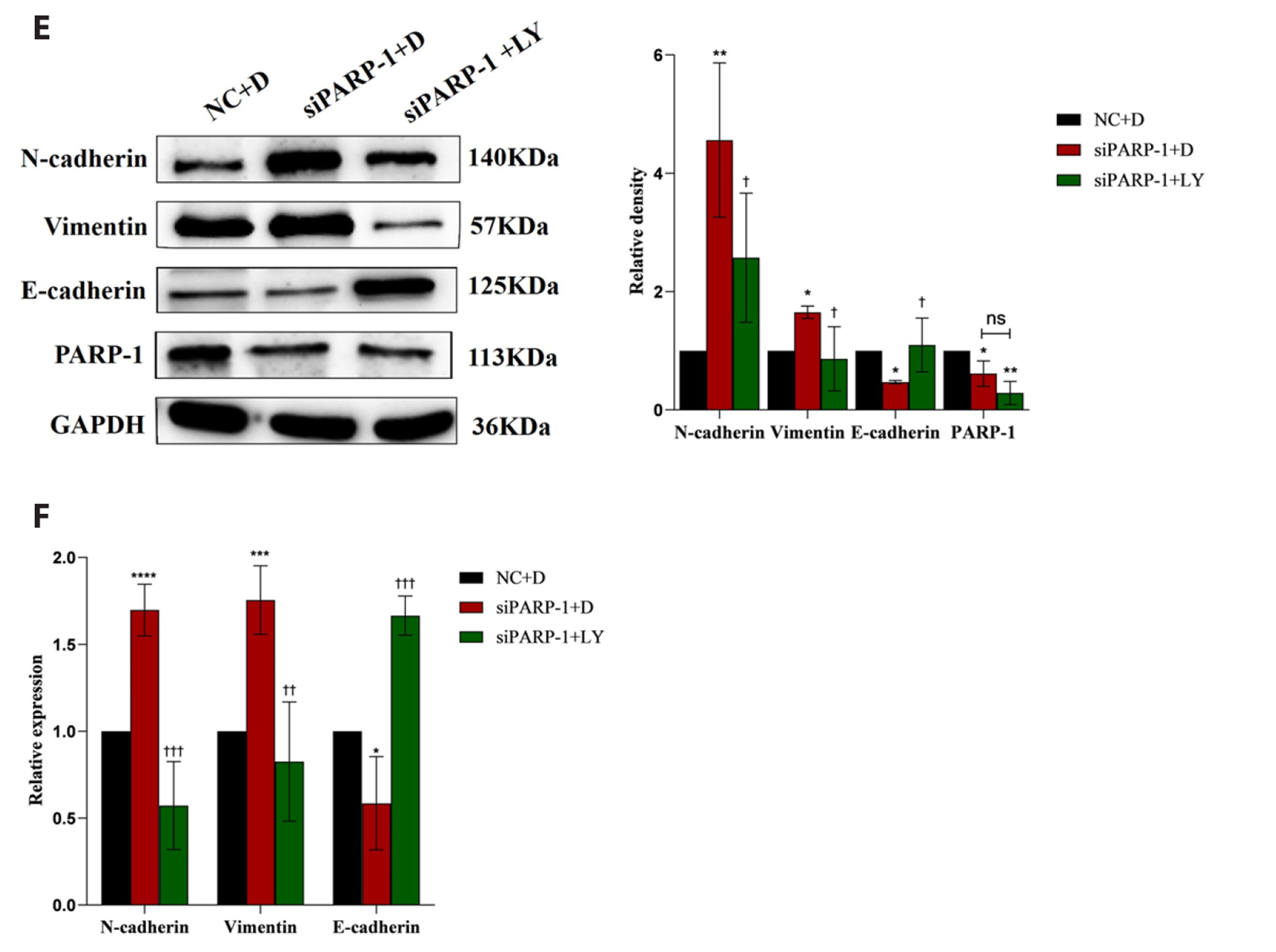
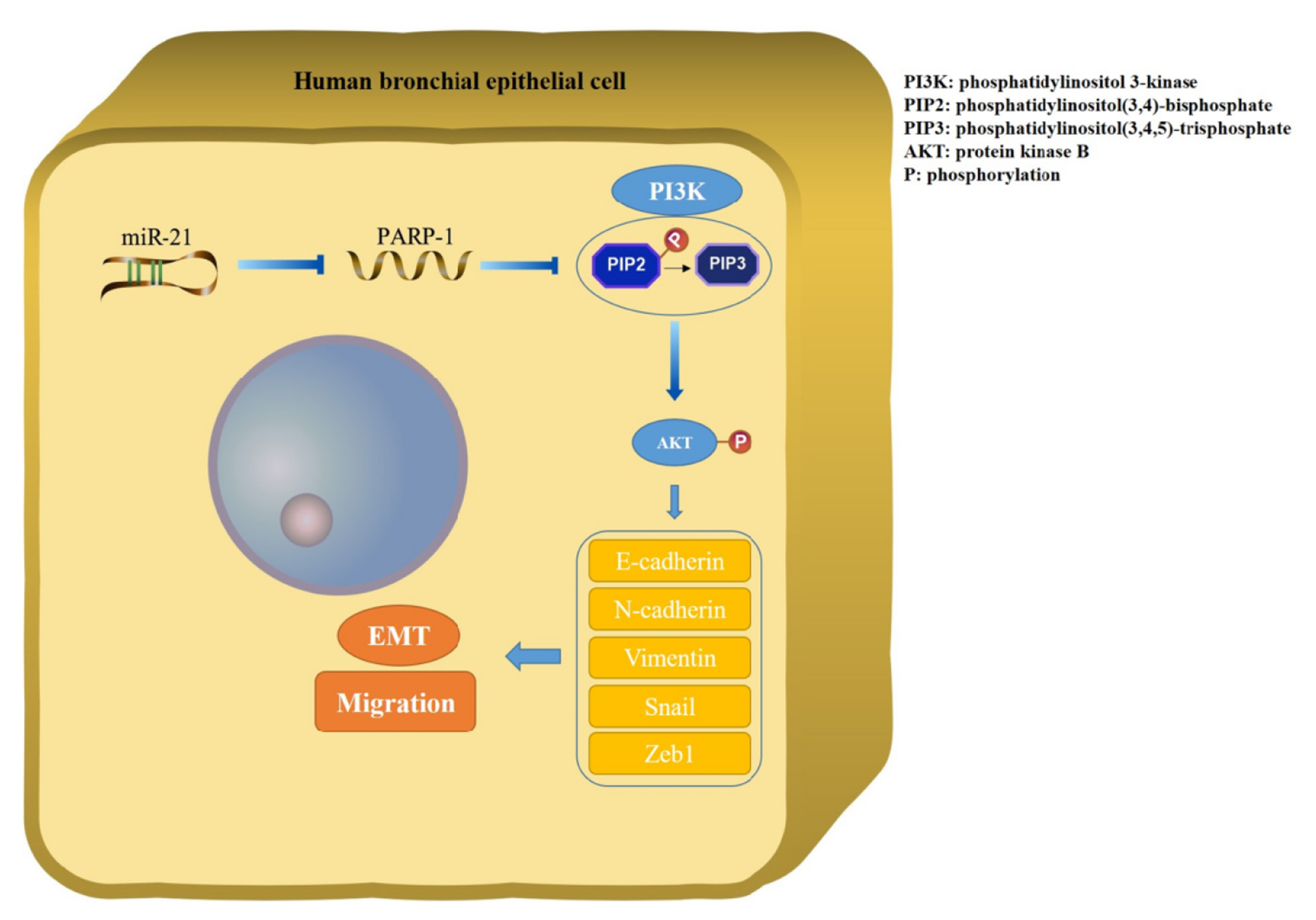
- Epithelial-mesenchymal transition (EMT ) is known to be involved in airway remodeling and fibrosis of bronchial asthma. However, the molecular mechanisms leading to EMT have yet to be fully clarified. The current study was designed to reveal the potential mechanism of microRNA-21 (miR-21) and poly (ADP-ribose) polymerase-1 (PARP-1) affecting EMT through the PI3K/AKT signaling pathway. Human bronchial epithelial cells (16HBE cells) were transfected with miR-21 mimics/inhibitors and PARP-1 plasmid/small interfering RNA (siRNA). A dual luciferase reporter assay and biotin-labeled RNA pull-down experiments were conducted to verify the targeting relationship between miR-21 mimics and PARP-1. The migration ability of 16HBE cells was evaluated by Transwell assay. Quantitative real-time polymerase chain reaction and Western blotting experiments were applied to determine the expression of Snail, ZEB1, E-cadherin, N-cadherin, Vimentin, and PARP-1. The effects of the PI3K inhibitor LY294002 on the migration of 16HBE cells and EMT were investigated. Overexpression of miR-21 mimics induced migration and EMT of 16HBE cells, which was significantly inhibited by overexpression of PARP-1. Our findings showed that PARP-1 was a direct target of miR-21, and that miR-21 targeted PARP-1 to promote migration and EMT of 16HBE cells through the PI3K/AKT signaling pathway. Using LY294002 to block PI3K/AKT signaling pathway resulted in a significant reduction in the migration and EMT of 16HBE cells. These results suggest that miR-21 promotes EMT and migration of HBE cells by targeting PARP-1. Additionally, the PI3K/AKT signaling pathway might be involved in this mechanism, which could indicate its usefulness as a therapeutic target for asthma.
Keyword
Figure
Reference
-
1. De Ferrari L, Chiappori A, Bagnasco D, Riccio AM, Passalacqua G, Canonica GW. 2016; Molecular phenotyping and biomarker development: are we on our way towards targeted therapy for severe asthma? Expert Rev Respir Med. 10:29–38. DOI: 10.1586/17476348.2016.1111763. PMID: 26566089. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84946866022&origin=inward.
Article2. Loffredo LF, Abdala-Valencia H, Anekalla KR, Cuervo-Pardo L, Gottardi CJ, Berdnikovs S. 2017; Beyond epithelial-to-mesenchymal transition: common suppression of differentiation programs underlies epithelial barrier dysfunction in mild, moderate, and severe asthma. Allergy. 72:1988–2004. DOI: 10.1111/all.13222. PMID: 28599074. PMCID: PMC5698119. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85021803995&origin=inward.
Article3. Jeffery PK. 2001; Remodeling in asthma and chronic obstructive lung disease. Am J Respir Crit Care Med. 164(10 Pt 2):S28–S38. DOI: 10.1164/ajrccm.164.supplement_2.2106061. PMID: 11734464. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0035891664&origin=inward.4. Larriba MJ, García de Herreros A, Muñoz A. 2016; Vitamin D and the epithelial to mesenchymal transition. Stem Cells Int. 2016:6213872. DOI: 10.1155/2016/6213872. PMID: 26880977. PMCID: PMC4736588. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84961353431&origin=inward.
Article5. Xia H, Xue J, Xu H, Lin M, Shi M, Sun Q, Xiao T, Dai X, Wu L, Li J, Xiang Q, Tang H, Bian Q, Liu Q. 2019; Andrographolide antagonizes the cigarette smoke-induced epithelial-mesenchymal transition and pulmonary dysfunction through anti-inflammatory inhibiting HOTAIR. Toxicology. 422:84–94. DOI: 10.1016/j.tox.2019.05.009. PMID: 31128153. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85066128653&origin=inward.
Article6. Haddad A, Gaudet M, Plesa M, Allakhverdi Z, Mogas AK, Audusseau S, Baglole CJ, Eidelman DH, Olivenstein R, Ludwig MS, Hamid Q. 2019; Neutrophils from severe asthmatic patients induce epithelial to mesenchymal transition in healthy bronchial epithelial cells. Respir Res. 20:234. DOI: 10.1186/s12931-019-1186-8. PMID: 31665016. PMCID: PMC6819645. PMID: f8111fbb356345d28571017c5e74b51b. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85074433350&origin=inward.
Article7. Hackett TL. 2012; Epithelial-mesenchymal transition in the pathophysiology of airway remodelling in asthma. Curr Opin Allergy Clin Immunol. 12:53–59. DOI: 10.1097/ACI.0b013e32834ec6eb. PMID: 22217512. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84856896936&origin=inward.
Article8. Pain M, Bermudez O, Lacoste P, Royer PJ, Botturi K, Tissot A, Brouard S, Eickelberg O, Magnan A. 2014; Tissue remodelling in chronic bronchial diseases: from the epithelial to mesenchymal phenotype. Eur Respir Rev. 23:118–130. DOI: 10.1183/09059180.00004413. PMID: 24591669.
Article9. Moret-Tatay I, Iborra M, Cerrillo E, Tortosa L, Nos P, Beltrán B. 2016; Possible biomarkers in blood for Crohn's disease: oxidative stress and microRNAs-current evidences and further aspects to unravel. Oxid Med Cell Longev. 2016:2325162. DOI: 10.1155/2016/2325162. PMID: 26823944. PMCID: PMC4707323. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84957061022&origin=inward.
Article10. Hammad Mahmoud Hammad R, Hamed DHED, Eldosoky MAER, Ahmad AAES, Osman HM, Abd Elgalil HM, Mahmoud Hassan MM. 2018; Plasma microRNA-21, microRNA-146a and IL-13 expression in asthmatic children. Innate Immun. 24:171–179. DOI: 10.1177/1753425918763521. PMID: 29635981. PMCID: PMC6852388. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85045521025&origin=inward.
Article11. Gurtan AM, Sharp PA. 2013; The role of miRNAs in regulating gene expression networks. J Mol Biol. 425:3582–3600. DOI: 10.1016/j.jmb.2013.03.007. PMID: 23500488. PMCID: PMC3757117. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84883805211&origin=inward.
Article12. Rebane A, Akdis CA. 2014; MicroRNAs in allergy and asthma. Curr Allergy Asthma Rep. 14:424. DOI: 10.1007/s11882-014-0424-x. PMID: 24504527. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84893156260&origin=inward.
Article13. Liu Y, Yang K, Shi H, Xu J, Zhang D, Wu Y, Zhou S, Sun X. 2015; MiR-21 modulates human airway smooth muscle cell proliferation and migration in asthma through regulation of PTEN expression. Exp Lung Res. 41:535–545. DOI: 10.3109/01902148.2015.1090501. PMID: 26651881. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84949768176&origin=inward.
Article14. Krishnakumar R, Kraus WL. 2010; The PARP side of the nucleus: molecular actions, physiological outcomes, and clinical targets. Mol Cell. 39:8–24. DOI: 10.1016/j.molcel.2010.06.017. PMID: 20603072. PMCID: PMC2923840. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=77954274504&origin=inward.
Article15. Naura AS, Hans CP, Zerfaoui M, You D, Cormier SA, Oumouna M, Boulares AH. 2008; Post-allergen challenge inhibition of poly(ADP-ribose) polymerase harbors therapeutic potential for treatment of allergic airway inflammation. Clin Exp Allergy. 38:839–846. DOI: 10.1111/j.1365-2222.2008.02943.x. PMID: 18261157. PMCID: PMC2740645. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=42249103606&origin=inward.
Article16. Naura AS, Zerfaoui M, Kim H, Abd Elmageed ZY, Rodriguez PC, Hans CP, Ju J, Errami Y, Park J, Ochoa AC, Boulares AH. 2010; Requirement for inducible nitric oxide synthase in chronic allergen exposure-induced pulmonary fibrosis but not inflammation. J Immunol. 185:3076–3085. DOI: 10.4049/jimmunol.0904214. PMID: 20668217. PMCID: PMC3077076. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=78049413973&origin=inward.
Article17. Lucarini L, Pini A, Gerace E, Pellicciari R, Masini E, Moroni F. 2014; Poly(ADP-ribose) polymerase inhibition with HYDAMTIQ reduces allergen-induced asthma-like reaction, bronchial hyper-reactivity and airway remodelling. J Cell Mol Med. 18:468–479. DOI: 10.1111/jcmm.12197. PMID: 24444146. PMCID: PMC3955153. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84894567125&origin=inward.
Article18. Kim KK, Kugler MC, Wolters PJ, Robillard L, Galvez MG, Brumwell AN, Sheppard D, Chapman HA. 2006; Alveolar epithelial cell mesenchymal transition develops in vivo during pulmonary fibrosis and is regulated by the extracellular matrix. Proc Natl Acad Sci U S A. 103:13180–13185. DOI: 10.1073/pnas.0605669103. PMID: 16924102. PMCID: PMC1551904. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=33748375828&origin=inward.
Article19. Hamidi A, Song J, Thakur N, Itoh S, Marcusson A, Bergh A, Heldin CH, Landström M. 2017; TGF-β promotes PI3K-AKT signaling and prostate cancer cell migration through the TRAF6-mediated ubiquitylation of p85α. Sci Signal. 10:eaal4186. DOI: 10.1126/scisignal.aal4186. PMID: 28676490. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85022054230&origin=inward.
Article20. Mao Z, Shi Y, Cao Q, Chen Y, Sun Y, Liu Z, Zhang Q, Huang M. 2018; Transcriptional regulation on the gene expression signature in combined allergic rhinitis and asthma syndrome. Epigenomics. 10:119–131. DOI: 10.2217/epi-2017-0072. PMID: 29334241. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85041135177&origin=inward.
Article21. Suzuki A, Yamaguchi MT, Ohteki T, Sasaki T, Kaisho T, Kimura Y, Yoshida R, Wakeham A, Higuchi T, Fukumoto M, Tsubata T, Ohashi PS, Koyasu S, Penninger JM, Nakano T, Mak TW. 2001; T cell-specific loss of Pten leads to defects in central and peripheral tolerance. Immunity. 14:523–534. DOI: 10.1016/S1074-7613(01)00134-0. PMID: 11371355. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=0034995242&origin=inward.
Article22. Kim SR, Park HJ, Lee KB, Kim HJ, Jeong JS, Cho SH, Lee YC. 2020; Epithelial PI3K-δ promotes house dust mite-induced allergic asthma in NLRP3 inflammasome-dependent and -independent manners. Allergy Asthma Immunol Res. 12:338–358. DOI: 10.4168/aair.2020.12.2.338. PMID: 32009326. PMCID: PMC6997282. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85082684615&origin=inward.
Article23. Cohen L, E X, Tarsi J, Ramkumar T, Horiuchi TK, Cochran R, DeMartino S, Schechtman KB, Hussain I, Holtzman MJ, Castro M. 2007; Epithelial cell proliferation contributes to airway remodeling in severe asthma. Am J Respir Crit Care Med. 176:138–145. DOI: 10.1164/rccm.200607-1062OC. PMID: 17463414. PMCID: PMC1994213. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=34447318645&origin=inward.
Article24. Liu G, Zhu R, Li B. 2005; TNF-alpha and IL-8 of the patients with allergic asthma. J Huazhong Univ Sci Technolog Med Sci. 25:274–275. 309DOI: 10.1007/BF02828140. PMID: 16201269.25. Pfeffer SR, Yang CH, Pfeffer LM. 2015; The role of miR-21 in cancer. Drug Dev Res. 76:270–277. DOI: 10.1002/ddr.21257. PMID: 26082192. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84941259771&origin=inward.
Article26. Wang G, Zhou Y, Chen W, Yang Y, Ye J, Ou H, Wu H. 2020; miR-21-5p promotes lung adenocarcinoma cell proliferation, migration and invasion via targeting WWC2. Cancer Biomark. 28:549–559. DOI: 10.3233/CBM-201489. PMID: 32623387. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85089709669&origin=inward.
Article27. Liu L, Pan Y, Zhai C, Zhu Y, Ke R, Shi W, Wang J, Yan X, Su X, Song Y, Gao L, Li M. 2018; Activation of peroxisome proliferation-activated receptor-γ inhibits transforming growth factor-β1-induced airway smooth muscle cell proliferation by suppressing Smad-miR-21 signaling. J Cell Physiol. 234:669–681. DOI: 10.1002/jcp.26839. PMID: 30132829. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85052377345&origin=inward.
Article28. Kim RY, Horvat JC, Pinkerton JW, Starkey MR, Essilfie AT, Mayall JR, Nair PM, Hansbro NG, Jones B, Haw TJ, Sunkara KP, Nguyen TH, Jarnicki AG, Keely S, Mattes J, Adcock IM, Foster PS, Hansbro PM. 2017; MicroRNA-21 drives severe, steroid-insensitive experimental asthma by amplifying phosphoinositide 3-kinase-mediated suppression of histone deacetylase 2. J Allergy Clin Immunol. 139:519–532. DOI: 10.1016/j.jaci.2016.04.038. PMID: 27448447. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84979590841&origin=inward.
Article29. Liu JH, Li C, Zhang CH, Zhang ZH. 2020; LncRNA-CASC7 enhances corticosteroid sensitivity via inhibiting the PI3K/AKT signaling pathway by targeting miR-21 in severe asthma. Pulmonology. 26:18–26. DOI: 10.1016/j.pulmoe.2019.07.001. PMID: 31412983. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85070301507&origin=inward.
Article30. Yu ZW, Xu YQ, Zhang XJ, Pan JR, Xiang HX, Gu XH, Ji SB, Qian J. 2019; Mutual regulation between miR-21 and the TGFβ/Smad signaling pathway in human bronchial fibroblasts promotes airway remodeling. J Asthma. 56:341–349. DOI: 10.1080/02770903.2018.1455859. PMID: 29621415. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85045072483&origin=inward.
Article31. Yamada M, Kubo H, Ota C, Takahashi T, Tando Y, Suzuki T, Fujino N, Makiguchi T, Takagi K, Suzuki T, Ichinose M. 2013; The increase of microRNA-21 during lung fibrosis and its contribution to epithelial-mesenchymal transition in pulmonary epithelial cells. Respir Res. 14:95. DOI: 10.1186/1465-9921-14-95. PMID: 24063588. PMCID: PMC3849377. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84884494821&origin=inward.
Article32. Gao Y, Lu J, Zhang Y, Chen Y, Gu Z, Jiang X. 2013; Baicalein attenuates bleomycin-induced pulmonary fibrosis in rats through inhibition of miR-21. Pulm Pharmacol Ther. 26:649–654. DOI: 10.1016/j.pupt.2013.03.006. PMID: 23523661. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84888009848&origin=inward.
Article33. Dai L, Chen F, Zheng Y, Zhang D, Qian B, Ji H, Long F, Cretoiu D. 2019; miR-21 regulates growth and EMT in lung cancer cells via PTEN/Akt/GSK3β signaling. Front Biosci (Landmark Ed). 24:1426–1439. DOI: 10.2741/4788. PMID: 31136988. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85067277602&origin=inward.34. Oumouna M, Datta R, Oumouna-Benachour K, Suzuki Y, Hans C, Matthews K, Fallon K, Boulares H. 2006; Poly(ADP-ribose) polymerase-1 inhibition prevents eosinophil recruitment by modulating Th2 cytokines in a murine model of allergic airway inflammation: a potential specific effect on IL-5. J Immunol. 177:6489–6496. Erratum. DOI: 10.4049/jimmunol.177.9.6489. PMID: 17056581. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=33750314513&origin=inward.
Article35. Echeverri Tirado LC, Ghonim MA, Wang J, Al-Khami AA, Wyczechowska D, Luu HH, Kim H, Sanchez-Pino MD, Yélamos J, Yassin LM, Boulares AH. 2019; PARP-1 is critical for recruitment of dendritic cells to the lung in a mouse model of asthma but dispensable for their differentiation and function. Mediators Inflamm. 2019:1656484. DOI: 10.1155/2019/1656484. PMID: 31178661. PMCID: PMC6507252. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85067541537&origin=inward.
Article36. Ghonim MA, Pyakurel K, Ibba SV, Wang J, Rodriguez P, Al-Khami AA, Lammi MR, Kim H, Zea AH, Davis C, Okpechi S, Wyczechowska D, Al-Ghareeb K, Mansy MS, Ochoa A, Naura AS, Boulares AH. 2015; PARP is activated in human asthma and its inhibition by olaparib blocks house dust mite-induced disease in mice. Clin Sci (Lond). 129:951–962. DOI: 10.1042/CS20150122. PMID: 26205779. PMCID: PMC4613510. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=84952012202&origin=inward.
Article37. Holguin F, Cardet JC, Chung KF, Diver S, Ferreira DS, Fitzpatrick A, Gaga M, Kellermeyer L, Khurana S, Knight S, McDonald VM, Morgan RL, Ortega VE, Rigau D, Subbarao P, Tonia T, Adcock IM, Bleecker ER, Brightling C, Boulet LP, et al. 2020; Management of severe asthma: a European Respiratory Society/American Thoracic Society guideline. Eur Respir J. 55:1900588. DOI: 10.1183/13993003.00588-2019. PMID: 31558662. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85077155298&origin=inward.
Article38. Saglani S. 2017; Childhood severe asthma: New insights on remodelling and biomarkers. Paediatr Respir Rev. 24:11–13. DOI: 10.1016/j.prrv.2017.06.001. PMID: 28697969. PMID: https://www.scopus.com/inward/record.uri?partnerID=HzOxMe3b&scp=85022059264&origin=inward.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- LETM1 Promotes Gastric Cancer Cell Proliferation, Migration, and Invasion via the PI3K/Akt Signaling Pathway
- Inhibition of PI3K/Akt signaling suppresses epithelial-to-mesenchymal transition in hepatocellular carcinoma through the Snail/GSK-3/beta-catenin pathway
- Vitexin Inhibits Gastric Cancer Growth and Metastasis through HMGB1-mediated Inactivation of the PI3K/ AKT/mTOR/HIF-1α Signaling Pathway
- Targeting epithelial-mesenchymal transition pathway in hepatocellular carcinoma
- MSCs-Derived miR-150-5p-Expressing Exosomes Promote Skin Wound Healing by Activating PI3K/AKT Pathway through PTEN