Intest Res.
2022 Apr;20(2):213-223. 10.5217/ir.2021.00031.
Drug therapy and monitoring for inflammatory bowel disease: a multinational questionnaire investigation in Asia
- Affiliations
-
- 1Division of Gastroenterology and Hepatology, Key Laboratory of Gastroenterology and Hepatology, Ministry of Health, Inflammatory Bowel Disease Research Center, Renji Hospital, School of Medicine, Shanghai Jiao Tong University, Shanghai Institute of Digestive Disease, Shanghai, China
- 2Department of Gastroenterology, Xijing Hospital, Air Force Medical University, Xi’an, China
- 3Division of Gastroenterology, Peking Union Medical College Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China
- KMID: 2529567
- DOI: http://doi.org/10.5217/ir.2021.00031
Abstract
- Background/Aims
The incidence and prevalence of inflammatory bowel disease (IBD) is rising in Asia recently. The study aimed to obtain a comprehensive understanding of the current status of drug therapy and monitoring for IBD in Asia.
Methods
A questionnaire investigation on drug therapy and monitoring for IBD was conducted right before the 6th Annual Meeting of Asian Organization for Crohn’s & Colitis. Questionnaires were provided to Asian physicians to fill out via emails between March and May 2018.
Results
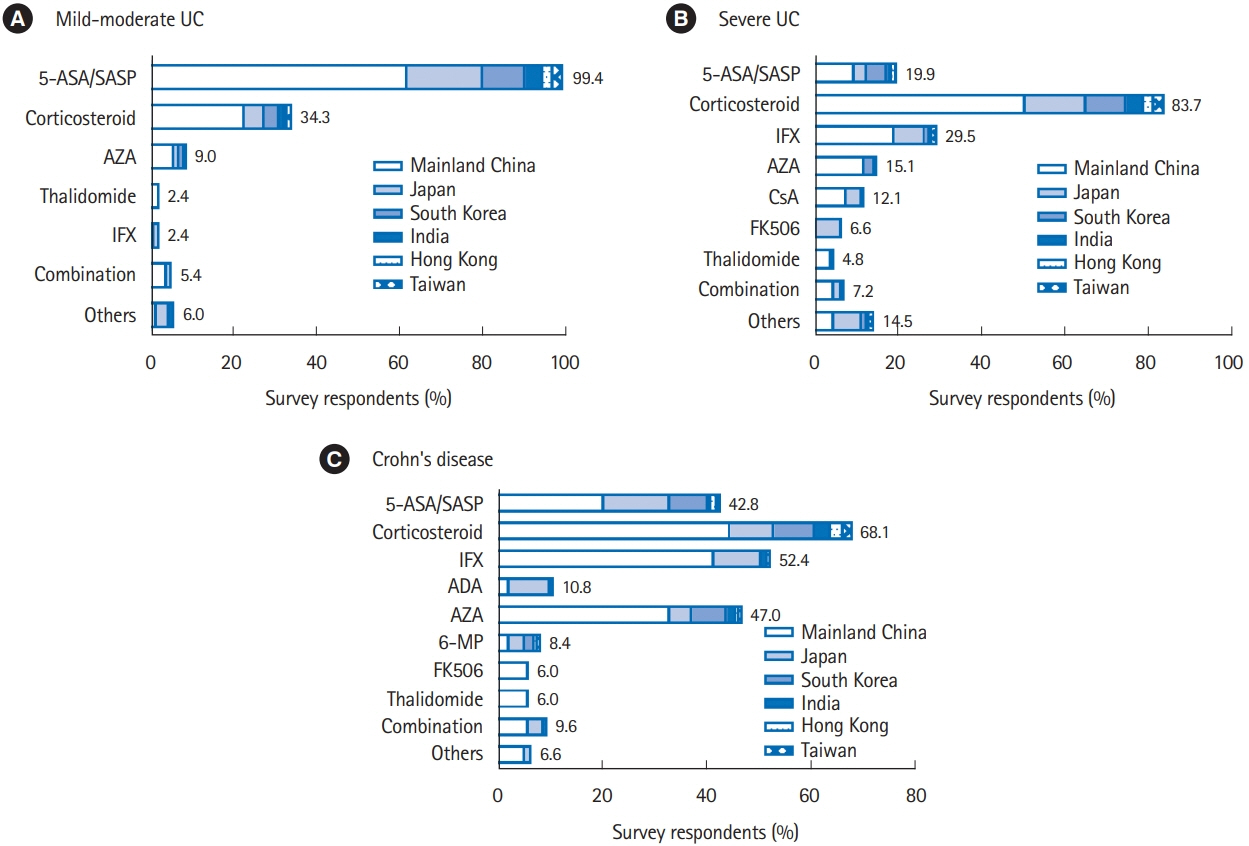
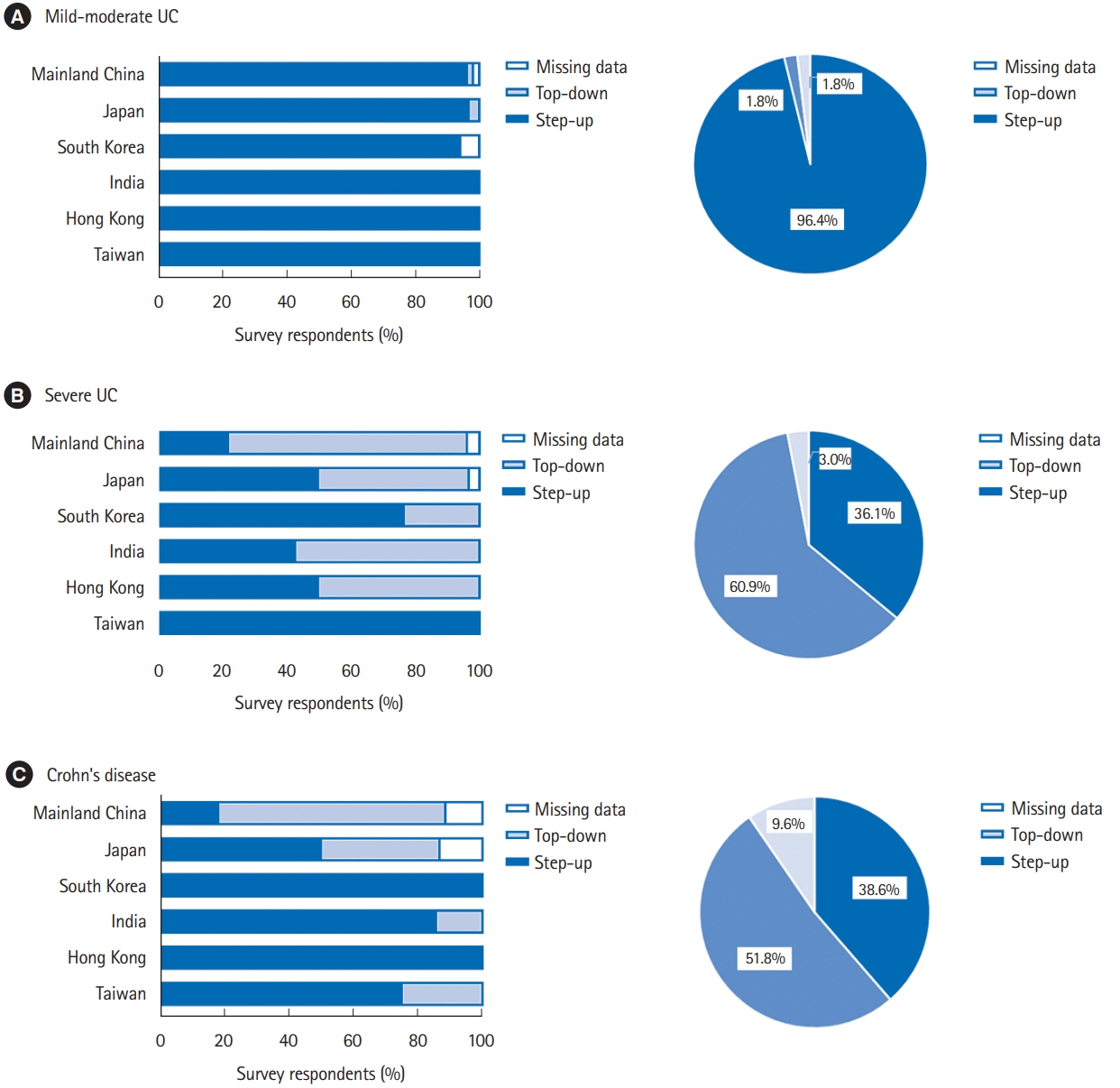
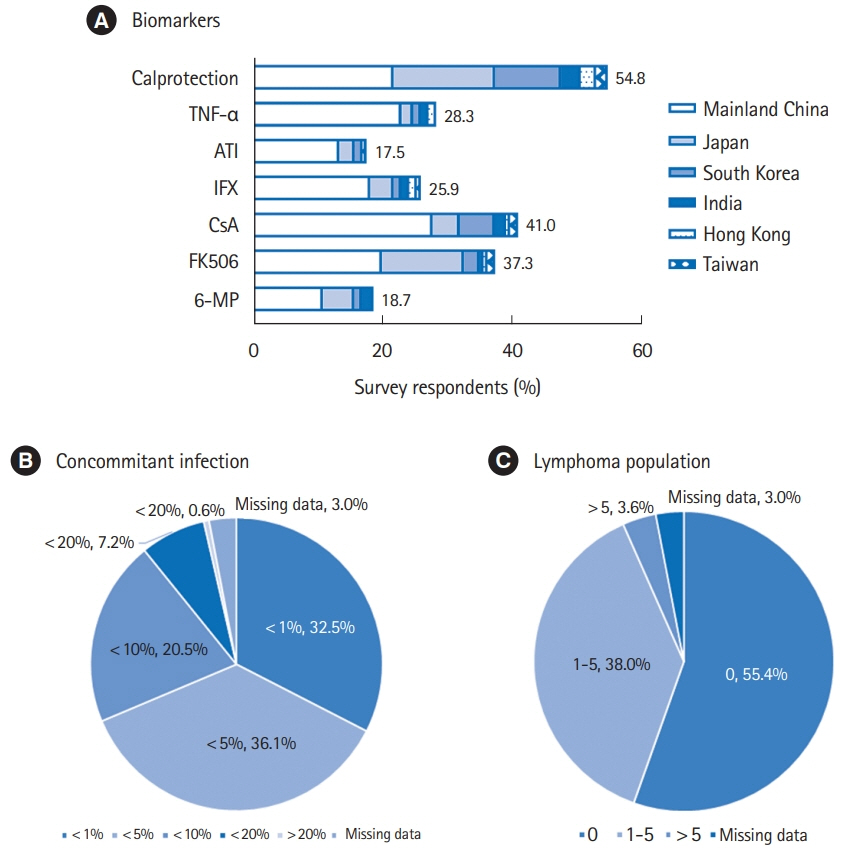
In total, responses of 166 physicians from 129 medical centers were included for analysis. Among the surveyed regions, the most average number of IBD specialist gastroenterologists and nurses was 4.8 per center in Taiwan and 2.5 per center in Mainland China, respectively. 5-Aminosalicylic acid/sulfasalazine (99.4%) was the most preferred first-line choice for mild-moderate ulcerative colitis (UC), meanwhile corticosteroid (83.7%) was widely applied for severe UC. The first-line medication for Crohn’s disease (CD) markedly varied as corticosteroid (68.1%) was the most favored in Mainland China, Japan, and South Korea, followed by infliximab (52.4%) and azathioprine (47.0%). Step-up strategy was preferred in mild-moderate UC (96.4%), while 51.8% of the physicians selected top-down treatment for CD. Only 25.9% and 17.5% of the physicians could test blood concentration of infliximab and antibody to infliximab in their hospitals, respectively.
Conclusions
The current status of drug therapy and monitoring for IBD in Asia possesses commonalities as well as differences. Asian recommendations, IBD specialist teams and practice of therapeutic drug monitoring are required to improve IBD management in Asia.
Figure
Reference
-
1. Ng SC, Shi HY, Hamidi N, et al. Worldwide incidence and prevalence of inflammatory bowel disease in the 21st century: a systematic review of population-based studies. Lancet. 2017; 390:2769–2778.
Article2. Goh K, Xiao SD. Inflammatory bowel disease: a survey of the epidemiology in Asia. J Dig Dis. 2009; 10:1–6.
Article3. Ng SC. Epidemiology of inflammatory bowel disease: focus on Asia. Best Pract Res Clin Gastroenterol. 2014; 28:363–372.
Article4. Ouyang Q, Tandon R, Goh KL, et al. Management consensus of inflammatory bowel disease for the Asia-Pacific region. J Gastroenterol Hepatol. 2006; 21:1772–1782.
Article5. Ooi CJ, Fock KM, Makharia GK, et al. The Asia-Pacific consensus on ulcerative colitis. J Gastroenterol Hepatol. 2010; 25:453–468.
Article6. Ooi CJ, Makharia GK, Hilmi I, et al. Asia Pacific Consensus Statements on Crohn’s disease. Part 1: Definition, diagnosis, and epidemiology: (Asia Pacific Crohn’s Disease Consensus--Part 1). J Gastroenterol Hepatol. 2016; 31:45–55.
Article7. Prideaux L, Kamm MA, De Cruz PP, Chan FK, Ng SC. Inflammatory bowel disease in Asia: a systematic review. J Gastroenterol Hepatol. 2012; 27:1266–1280.
Article8. Hida N, Nakamura S, Hahm KB, et al. A questionnaire-based survey on the diagnosis and management of inflammatory bowel disease in East Asian countries in 2012. Digestion. 2014; 89:88–103.
Article9. Simian D, Quera R. Towards an integral management of inflammatory bowel disease. Rev Med Chil. 2016; 144:488–495.10. Pineton de Chambrun G, Blanc P, Peyrin-Biroulet L. Current evidence supporting mucosal healing and deep remission as important treatment goals for inflammatory bowel disease. Expert Rev Gastroenterol Hepatol. 2016; 10:915–927.
Article11. Hindryckx P, Vande Casteele N, Novak G, et al. The expanding therapeutic armamentarium for inflammatory bowel disease: how to choose the right drug[s] for our patients? J Crohns Colitis. 2018; 12:105–119.
Article12. Rubin DT, Uluscu O, Sederman R. Response to biologic therapy in Crohn’s disease is improved with early treatment: an analysis of health claims data. Inflamm Bowel Dis. 2012; 18:2225–2231.
Article13. Lee WJ, Briars L, Lee TA, Calip GS, Suda KJ, Schumock GT. Top-down versus step-up prescribing strategies for tumor necrosis factor alpha inhibitors in children and young adults with inflammatory bowel disease. Inflamm Bowel Dis. 2016; 22:2410–7.
Article14. Feagan BG, Macdonald JK. Oral 5-aminosalicylic acid for induction of remission in ulcerative colitis. Cochrane Database Syst Rev. 2012; 10–CD000543.
Article15. Wang Y, Parker CE, Feagan BG, MacDonald JK. Oral 5-aminosalicylic acid for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2016; 2016–CD000544.
Article16. Nakase H, Keum B, Ye BD, Park SJ, Koo HS, Eun CS. Treatment of inflammatory bowel disease in Asia: the results of a multinational web-based survey in the 2(nd) Asian Organization of Crohn’s and Colitis (AOCC) meeting in Seoul. Intest Res. 2016; 14:231–239.
Article17. Ford AC, Khan KJ, Achkar JP, Moayyedi P. Efficacy of oral vs. topical, or combined oral and topical 5-aminosalicylates, in ulcerative colitis: systematic review and meta-analysis. Am J Gastroenterol. 2012; 107:167–176.
Article18. Akobeng AK, Gardener E. Oral 5-aminosalicylic acid for maintenance of medically-induced remission in Crohn’s disease. Cochrane Database Syst Rev. 2005; (1):CD003715.
Article19. Qiu X, Ma J, Wang K, Zhang H. Chemopreventive effects of 5-aminosalicylic acid on inflammatory bowel disease-associated colorectal cancer and dysplasia: a systematic review with meta-analysis. Oncotarget. 2017; 8:1031–1045.
Article20. Carrat F, Seksik P, Colombel JF, Peyrin-Biroulet L, Beaugerie L; CESAME Study Group. The effects of aminosalicylates or thiopurines on the risk of colorectal cancer in inflammatory bowel disease. Aliment Pharmacol Ther. 2017; 45:533–541.
Article21. Chang S, Hanauer S. Optimizing pharmacologic management of inflammatory bowel disease. Expert Rev Clin Pharmacol. 2017; 10:595–607.
Article22. Ford AC, Bernstein CN, Khan KJ, et al. Glucocorticosteroid therapy in inflammatory bowel disease: systematic review and meta-analysis. Am J Gastroenterol. 2011; 106:590–599.
Article23. Sandborn WJ, Bosworth B, Zakko S, et al. Budesonide foam induces remission in patients with mild to moderate ulcerative proctitis and ulcerative proctosigmoiditis. Gastroenterology. 2015; 148:740–750.
Article24. Terdiman JP, Gruss CB, Heidelbaugh JJ, Sultan S, Falck-Ytter YT; AGA Institute Clinical Practice and Quality Management Committee. American Gastroenterological Association Institute guideline on the use of thiopurines, methotrexate, and anti-TNF-α biologic drugs for the induction and maintenance of remission in inflammatory Crohn’s disease. Gastroenterology. 2013; 145:1459–1463.
Article25. Colombel JF, Sandborn WJ, Reinisch W, et al. Infliximab, azathioprine, or combination therapy for Crohn’s disease. N Engl J Med. 2010; 362:1383–1395.
Article26. Panaccione R, Ghosh S, Middleton S, et al. Combination therapy with infliximab and azathioprine is superior to monotherapy with either agent in ulcerative colitis. Gastroenterology. 2014; 146:392–400.
Article27. Sampaio EP, Sarno EN, Galilly R, Cohn ZA, Kaplan G. Thalidomide selectively inhibits tumor necrosis factor alpha production by stimulated human monocytes. J Exp Med. 1991; 173:699–703.
Article28. Moreira AL, Sampaio EP, Zmuidzinas A, Frindt P, Smith KA, Kaplan G. Thalidomide exerts its inhibitory action on tumor necrosis factor alpha by enhancing mRNA degradation. J Exp Med. 1993; 177:1675–1680.
Article29. Moller DR, Wysocka M, Greenlee BM, et al. Inhibition of IL-12 production by thalidomide. J Immunol. 1997; 159:5157–5161.
Article30. Yang C, Singh P, Singh H, Le ML, El-Matary W. Systematic review: thalidomide and thalidomide analogues for treatment of inflammatory bowel disease. Aliment Pharmacol Ther. 2015; 41:1079–1093.
Article31. Simon M, Pariente B, Lambert J, et al. Long-term outcomes of thalidomide therapy for adults with refractory Crohn’s disease. Clin Gastroenterol Hepatol. 2016; 14:966–972.
Article32. Lazzerini M, Martelossi S, Magazzù G, et al. Effect of thalidomide on clinical remission in children and adolescents with refractory Crohn disease: a randomized clinical trial. JAMA. 2013; 310:2164–2173.
Article33. Kornbluth A. Infliximab approved for use in Crohn’s disease: a report on the FDA GI Advisory Committee conference. Inflamm Bowel Dis. 1998; 4:328–329.
Article34. Tamm R, Mägi R, Tremmel R, et al. Polymorphic variation in TPMT is the principal determinant of TPMT phenotype: a meta-analysis of three genome-wide association studies. Clin Pharmacol Ther. 2017; 101:684–695.
Article35. Zhu X, Wang XD, Chao K, et al. NUDT15 polymorphisms are better than thiopurine S-methyltransferase as predictor of risk for thiopurine-induced leukopenia in Chinese patients with Crohn’s disease. Aliment Pharmacol Ther. 2016; 44:967–975.
Article36. Dart RJ, Irving PM. Optimising use of thiopurines in inflammatory bowel disease. Expert Rev Clin Immunol. 2017; 13:877–888.
Article37. Spencer EA, Dubinsky MC. Therapeutic drug monitoring in inflammatory bowel disease: history and future directions. Pediatr Clin North Am. 2017; 64:1309–1326.38. Ma C, Battat R, Jairath V, Vande Casteele N. Advances in therapeutic drug monitoring for small-molecule and biologic therapies in inflammatory bowel disease. Curr Treat Options Gastroenterol. 2019; 17:127–145.
Article39. Gonczi L, Kurti Z, Rutka M, et al. Drug persistence and need for dose intensification to adalimumab therapy; the importance of therapeutic drug monitoring in inflammatory bowel diseases. BMC Gastroenterol. 2017; 17:97.
Article40. Papamichael K, Vajravelu RK, Vaughn BP, Osterman MT, Cheifetz AS. Proactive infliximab monitoring following reactive testing is associated with better clinical outcomes than reactive testing alone in patients with inflammatory bowel disease. J Crohns Colitis. 2018; 12:804–810.
Article41. Papamichael K, Juncadella A, Wong D, et al. Proactive therapeutic drug monitoring of adalimumab is associated with better long-term outcomes compared with standard of care in patients with inflammatory bowel disease. J Crohns Colitis. 2019; 13:976–981.
Article42. Lega S, Phan BL, Rosenthal CJ, et al. Proactively optimized infliximab monotherapy is as effective as combination therapy in IBD. Inflamm Bowel Dis. 2019; 25:134–141.
Article43. Papamichael K, Vande Casteele N, Ferrante M, Gils A, Cheifetz AS. Therapeutic drug monitoring during induction of antitumor necrosis factor therapy in inflammatory bowel disease: defining a therapeutic drug window. Inflamm Bowel Dis. 2017; 23:1510–1515.
Article44. Restellini S, Chao CY, Lakatos PL, et al. Therapeutic drug monitoring guides the management of Crohn’s patients with secondary loss of response to adalimumab. Inflamm Bowel Dis. 2018; 24:1531–1538.
Article45. Guidi L, Pugliese D, Panici Tonucci T, et al. Therapeutic drug monitoring is more cost-effective than a clinically based approach in the management of loss of response to infliximab in inflammatory bowel disease: an observational multicentre study. J Crohns Colitis. 2018; 12:1079–1088.
Article46. Deora V, Kozak J, El-Kalla M, Huynh HQ, El-Matary W. Therapeutic drug monitoring was helpful in guiding the decisionmaking process for children receiving infliximab for inflammatory bowel disease. Acta Paediatr. 2017; 106:1863–1867.
Article47. Hoseyni H, Xu Y, Zhou H. Therapeutic drug monitoring of biologics for inflammatory bowel disease: an answer to optimized treatment? J Clin Pharmacol. 2018; 58:864–876.
Article48. Campbell JP, Burton E, Wymer S, Shaw M, Vaughn BP. Out-of-pocket cost is a barrier to therapeutic drug monitoring in inflammatory bowel disease. Dig Dis Sci. 2017; 62:3336–3343.
Article49. Grossberg LB, Papamichael K, Feuerstein JD, Siegel CA, Ullman TA, Cheifetz AS. A survey study of gastroenterologists’ attitudes and barriers toward therapeutic drug monitoring of anti-TNF therapy in inflammatory bowel disease. Inflamm Bowel Dis. 2017; 24:191–197.
Article50. Siegel CA. Refocusing IBD patient management: personalized, proactive, and patient-centered care. Am J Gastroenterol. 2018; 113:1440–1443.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Monitoring and Safety of Azathioprine Therapy in Inflammatory Bowel Disease
- Inflammatory Bowel Disease in Asia: The Challenges and Opportunities
- Update on the epidemiology of inflammatory bowel disease in Asia: where are we now?
- Precision medicine for pediatric inflammatory bowel disease: a perspective
- Treatment of inflammatory bowel disease in Asia: the results of a multinational web-based survey in the 2nd Asian Organization of Crohn's and Colitis (AOCC) meeting in Seoul