Nutr Res Pract.
2022 Apr;16(2):147-160. 10.4162/nrp.2022.16.2.147.
Ursolic acid improves the indoxyl sulfate-induced impairment of mitochondrial biogenesis in C2C12 cells
- Affiliations
-
- 1Department of Nutrition, Faculty of Health Science, Aomori University of Health and Welfare, Aomori 030-8505, Japan
- 2Department of Food and Human Health Sciences, Graduate School of Human Life & Science, Osaka City University, Osaka 558-8585, Japan
- KMID: 2527807
- DOI: http://doi.org/10.4162/nrp.2022.16.2.147
Abstract
- BACKGROUND/OBJECTIVES
Patients with chronic kidney disease (CKD) have a high concentration of uremic toxins in their blood and often experience muscle atrophy. Indoxyl sulfate (IS) is a uremic toxin produced by tryptophan metabolism. Although an elevated IS level may induce muscle dysfunction, the effect of IS on physiological concentration has not been elucidated. Additionally, the effects of ursolic acid (UA) on muscle hypertrophy have been reported in healthy models; however, it is unclear whether UA ameliorates muscle dysfunction associated with chronic diseases, such as CKD. Thus, this study aimed to investigate whether UA can improve the IS-induced impairment of mitochondrial biogenesis.
MATERIALS/METHODS
C2C12 cells were incubated with or without IS (0.1 mM) and UA (1 or 2 µM) to elucidate the physiological effect of UA on CKD-related mitochondrial dysfunction and its related mechanisms using real-time reverse transcription-polymerase chain reaction, western blotting and enzyme-linked immunosorbent assay.
RESULTS
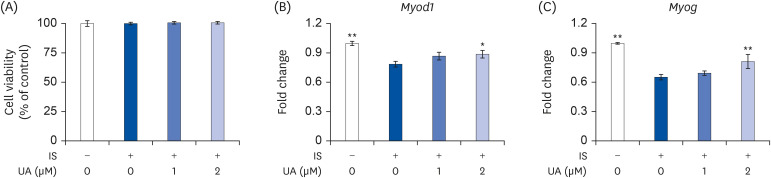
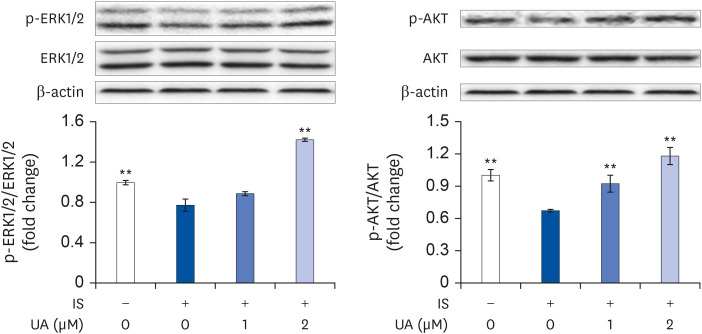
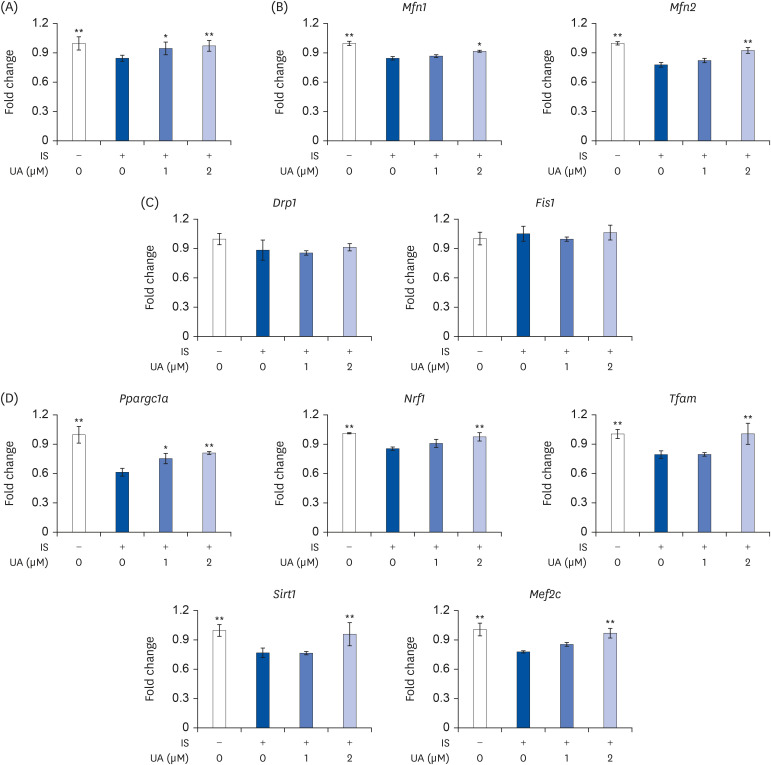
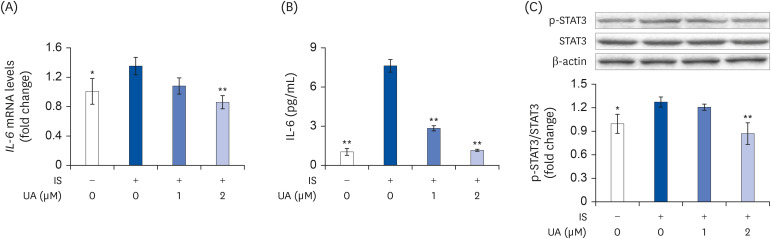
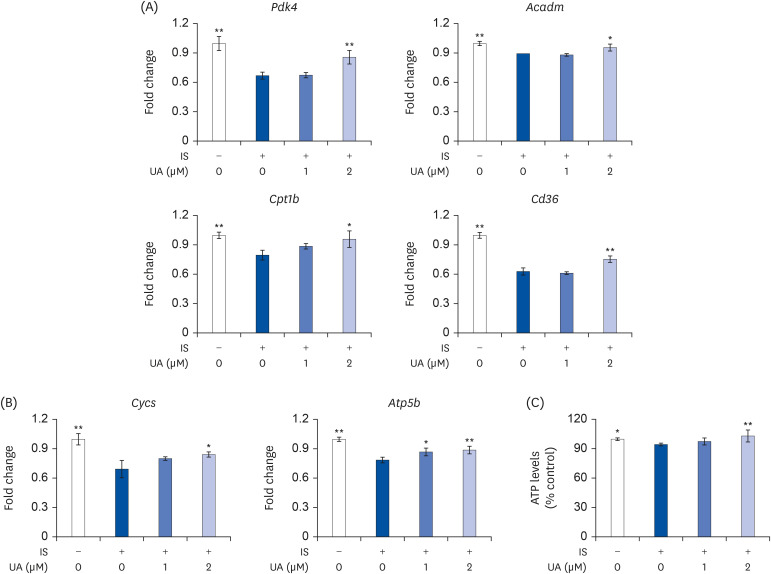
IS suppressed the expression of differentiation marker genes without decreasing cell viability. IS decreased the mitochondrial DNA copy number and ATP levels by downregulating the genes pertaining to mitochondrial biogenesis (Ppargc1a, Nrf1, Tfam, Sirt1, and Mef2c), fusion (Mfn1 and Mfn2), oxidative phosphorylation (Cycs and Atp5b), and fatty acid oxidation (Pdk4, Acadm, Cpt1b, and Cd36). Furthermore, IS increased the intracellular mRNA and secretory protein levels of interleukin (IL)-6. Finally, UA ameliorated the IS-induced impairment in C2C12 cells.
CONCLUSIONS
Our results indicated that UA improves the IS-induced impairment of mitochondrial biogenesis by affecting differentiation, ATP levels, and IL-6 secretion in C2C12 cells. Therefore, UA could be a novel therapeutic agent for CKD-induced muscle dysfunction.
Figure
Reference
-
1. Wang XH, Mitch WE. Mechanisms of muscle wasting in chronic kidney disease. Nat Rev Nephrol. 2014; 10:504–516. PMID: 24981816.2. Carrero JJ, Johansen KL, Lindholm B, Stenvinkel P, Cuppari L, Avesani CM. Screening for muscle wasting and dysfunction in patients with chronic kidney disease. Kidney Int. 2016; 90:53–66. PMID: 27157695.3. Hirai K, Ookawara S, Morishita Y. Sarcopenia and physical inactivity in patients with chronic kidney disease. Nephrourol Mon. 2016; 8:e37443. PMID: 27570755.4. Kim JK, Choi SR, Choi MJ, Kim SG, Lee YK, Noh JW, Kim HJ, Song YR. Prevalence of and factors associated with sarcopenia in elderly patients with end-stage renal disease. Clin Nutr. 2014; 33:64–68. PMID: 23631844.5. Souza VA, Oliveira D, Mansur HN, Fernandes NM, Bastos MG. Sarcopenia in chronic kidney disease. J Bras Nefrol. 2015; 37:98–105. PMID: 25923756.6. Beasley JM, Shikany JM, Thomson CA. The role of dietary protein intake in the prevention of sarcopenia of aging. Nutr Clin Pract. 2013; 28:684–690. PMID: 24163319.7. Mandayam S, Mitch WE. Dietary protein restriction benefits patients with chronic kidney disease. Nephrology (Carlton). 2006; 11:53–57. PMID: 16509933.8. Joyner MJ, Coyle EF. Endurance exercise performance: the physiology of champions. J Physiol. 2008; 586:35–44. PMID: 17901124.9. Tadaishi M, Miura S, Kai Y, Kano Y, Oishi Y, Ezaki O. Skeletal muscle-specific expression of PGC-1α-b, an exercise-responsive isoform, increases exercise capacity and peak oxygen uptake. PLoS One. 2011; 6:e28290. PMID: 22174785.10. Kosmadakis GC, Bevington A, Smith AC, Clapp EL, Viana JL, Bishop NC, Feehally J. Physical exercise in patients with severe kidney disease. Nephron Clin Pract. 2010; 115:c7–16. PMID: 20173344.11. Gamboa JL, Billings FT 4th, Bojanowski MT, Gilliam LA, Yu C, Roshanravan B, Roberts LJ 2nd, Himmelfarb J, Ikizler TA, Brown NJ. Mitochondrial dysfunction and oxidative stress in patients with chronic kidney disease. Physiol Rep. 2016; 4:e12780. PMID: 27162261.12. Rao M, Li L, Demello C, Guo D, Jaber BL, Pereira BJ, Balakrishnan VS. HEMO Study Group. Mitochondrial DNA injury and mortality in hemodialysis patients. J Am Soc Nephrol. 2009; 20:189–196. PMID: 18684894.13. Fujii H, Nakai K, Fukagawa M. Role of oxidative stress and indoxyl sulfate in progression of cardiovascular disease in chronic kidney disease. Ther Apher Dial. 2011; 15:125–128. PMID: 21426501.14. Lim CF, Bernard BF, de Jong M, Docter R, Krenning EP, Hennemann G. A furan fatty acid and indoxyl sulfate are the putative inhibitors of thyroxine hepatocyte transport in uremia. J Clin Endocrinol Metab. 1993; 76:318–324. PMID: 8432774.15. Enoki Y, Watanabe H, Arake R, Sugimoto R, Imafuku T, Tominaga Y, Ishima Y, Kotani S, Nakajima M, Tanaka M, et al. Indoxyl sulfate potentiates skeletal muscle atrophy by inducing the oxidative stress-mediated expression of myostatin and atrogin-1. Sci Rep. 2016; 6:32084. PMID: 27549031.16. Jäger S, Trojan H, Kopp T, Laszczyk MN, Scheffler A. Pentacyclic triterpene distribution in various plants - rich sources for a new group of multi-potent plant extracts. Molecules. 2009; 14:2016–2031. PMID: 19513002.17. Lee J, Yee ST, Kim JJ, Choi MS, Kwon EY, Seo KI, Lee MK. Ursolic acid ameliorates thymic atrophy and hyperglycemia in streptozotocin-nicotinamide-induced diabetic mice. Chem Biol Interact. 2010; 188:635–642. PMID: 20869956.18. Tsai SJ, Yin MC. Antioxidative and anti-inflammatory protection of oleanolic acid and ursolic acid in PC12 cells. J Food Sci. 2008; 73:H174–H178. PMID: 18803714.19. Leng S, Hao Y, Du D, Xie S, Hong L, Gu H, Zhu X, Zhang J, Fan D, Kung HF. Ursolic acid promotes cancer cell death by inducing Atg5-dependent autophagy. Int J Cancer. 2013; 133:2781–2790. PMID: 23737395.20. Kunkel SD, Suneja M, Ebert SM, Bongers KS, Fox DK, Malmberg SE, Alipour F, Shields RK, Adams CM. mRNA expression signatures of human skeletal muscle atrophy identify a natural compound that increases muscle mass. Cell Metab. 2011; 13:627–638. PMID: 21641545.21. Bang HS, Seo DY, Chung YM, Oh KM, Park JJ, Arturo F, Jeong SH, Kim N, Han J. Ursolic Acid-induced elevation of serum irisin augments muscle strength during resistance training in men. Korean J Physiol Pharmacol. 2014; 18:441–446. PMID: 25352765.22. Zhang SZ, Lipsky MM, Trump BF, Hsu IC. Neutral red (NR) assay for cell viability and xenobiotic-induced cytotoxicity in primary cultures of human and rat hepatocytes. Cell Biol Toxicol. 1990; 6:219–234. PMID: 2113829.23. Duranton F, Cohen G, De Smet R, Rodriguez M, Jankowski J, Vanholder R, Argiles A. European Uremic Toxin Work Group. Normal and pathologic concentrations of uremic toxins. J Am Soc Nephrol. 2012; 23:1258–1270. PMID: 22626821.24. Rodrigues GGC, Dellê H, Brito RBO, Cardoso VO, Fernandes KPS, Mesquita-Ferrari RA, Cunha RS, Stinghen AEM, Dalboni MA, Barreto FC. Indoxyl sulfate contributes to uremic sarcopenia by inducing apoptosis in myoblasts. Arch Med Res. 2020; 51:21–29. PMID: 32086105.25. Bentzinger CF, Wang YX, Rudnicki MA. Building muscle: molecular regulation of myogenesis. Cold Spring Harb Perspect Biol. 2012; 4:a008342. PMID: 22300977.26. Alessi DR, Andjelkovic M, Caudwell B, Cron P, Morrice N, Cohen P, Hemmings BA. Mechanism of activation of protein kinase B by insulin and IGF-1. EMBO J. 1996; 15:6541–6551. PMID: 8978681.27. Ahn NG. The MAP kinase cascade. Discovery of a new signal transduction pathway. Mol Cell Biochem. 1993; 127-128:201–209. PMID: 7935352.28. Westermann B. Bioenergetic role of mitochondrial fusion and fission. Biochim Biophys Acta. 2012; 1817:1833–1838. PMID: 22409868.29. Ihle JN. Cytokine receptor signalling. Nature. 1995; 377:591–594. PMID: 7566171.30. Kuppusamy P, Soundharrajan I, Kim DH, Hwang I, Choi KC. 4-Hydroxy-3-methoxy cinnamic acid accelerate myoblasts differentiation on C2C12 mouse skeletal muscle cells via AKT and ERK 1/2 activation. Phytomedicine. 2019; 60:152873. PMID: 30879871.31. Li X, Zhu Y, Zhang H, Ma G, Wu G, Xiang A, Shi X, Yang GS, Sun S. MicroRNA-106a-5p inhibited C2C12 myogenesis via targeting PIK3R1 and modulating the PI3K/AKT signaling. Genes (Basel). 2018; 9:333.32. Kim JC, Kang YS, Noh EB, Seo BW, Seo DY, Park GD, Kim SH. Concurrent treatment with ursolic acid and low-intensity treadmill exercise improves muscle atrophy and related outcomes in rats. Korean J Physiol Pharmacol. 2018; 22:427–436. PMID: 29962857.33. Wagatsuma A, Sakuma K. Mitochondria as a potential regulator of myogenesis. Sci World J. 2013; 2013:593267.34. Avin KG, Chen NX, Organ JM, Zarse C, O'Neill K, Conway RG, Konrad RJ, Bacallao RL, Allen MR, Moe SM. Skeletal muscle regeneration and oxidative stress are altered in chronic kidney disease. PLoS One. 2016; 11:e0159411. PMID: 27486747.35. Dai W, Jiang L. Dysregulated mitochondrial dynamics and metabolism in obesity, diabetes, and cancer. Front Endocrinol (Lausanne). 2019; 10:570. PMID: 31551926.36. Vásquez-Trincado C, García-Carvajal I, Pennanen C, Parra V, Hill JA, Rothermel BA, Lavandero S. Mitochondrial dynamics, mitophagy and cardiovascular disease. J Physiol. 2016; 594:509–525. PMID: 26537557.37. Chen H, Vermulst M, Wang YE, Chomyn A, Prolla TA, McCaffery JM, Chan DC. Mitochondrial fusion is required for mtDNA stability in skeletal muscle and tolerance of mtDNA mutations. Cell. 2010; 141:280–289. PMID: 20403324.38. Ishihara N, Nomura M, Jofuku A, Kato H, Suzuki SO, Masuda K, Otera H, Nakanishi Y, Nonaka I, Goto Y, et al. Mitochondrial fission factor Drp1 is essential for embryonic development and synapse formation in mice. Nat Cell Biol. 2009; 11:958–966. PMID: 19578372.39. Rugarli EI, Langer T. Mitochondrial quality control: a matter of life and death for neurons. EMBO J. 2012; 31:1336–1349. PMID: 22354038.40. Wu H, Kanatous SB, Thurmond FA, Gallardo T, Isotani E, Bassel-Duby R, Williams RS. Regulation of mitochondrial biogenesis in skeletal muscle by CaMK. Science. 2002; 296:349–352. PMID: 11951046.41. Wang D, Wei L, Yang Y, Liu H. Dietary supplementation with ketoacids protects against CKD-induced oxidative damage and mitochondrial dysfunction in skeletal muscle of 5/6 nephrectomised rats. Skelet Muscle. 2018; 8:18. PMID: 29855350.42. Su. Z, Klein JD, Du J, Franch HA, Zhang L, Hassounah F, Hudson MB, Wang XH. Chronic kidney disease induces autophagy leading to dysfunction of mitochondria in skeletal muscle. Am J Physiol Renal Physiol. 2017; 312:F1128–F1140. PMID: 28381463.43. Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, Troy A, Cinti S, Lowell B, Scarpulla RC, et al. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell. 1999; 98:115–124. PMID: 10412986.44. Picca A, Lezza AM. Regulation of mitochondrial biogenesis through TFAM-mitochondrial DNA interactions: useful insights from aging and calorie restriction studies. Mitochondrion. 2015; 25:67–75. PMID: 26437364.45. Kim JC, Park GD, Kim SH. Inhibition of oxidative stress by antioxidant supplementation does not limit muscle mitochondrial biogenesis or endurance capacity in rats. J Nutr Sci Vitaminol (Tokyo). 2017; 63:277–283. PMID: 29225311.46. Tamaki M, Miyashita K, Wakino S, Mitsuishi M, Hayashi K, Itoh H. Chronic kidney disease reduces muscle mitochondria and exercise endurance and its exacerbation by dietary protein through inactivation of pyruvate dehydrogenase. Kidney Int. 2014; 85:1330–1339. PMID: 24284514.47. White JP, Puppa MJ, Sato S, Gao S, Price RL, Baynes JW, Kostek MC, Matesic LE, Carson JA. IL-6 regulation on skeletal muscle mitochondrial remodeling during cancer cachexia in the ApcMin/+ mouse. Skelet Muscle. 2012; 2:14. PMID: 22769563.48. Bonetto A, Aydogdu T, Jin X, Zhang Z, Zhan R, Puzis L, Koniaris LG, Zimmers TA. JAK/STAT3 pathway inhibition blocks skeletal muscle wasting downstream of IL-6 and in experimental cancer cachexia. Am J Physiol Endocrinol Metab. 2012; 303:E410–E421. PMID: 22669242.49. Siengdee P, Trakooljul N, Murani E, Schwerin M, Wimmers K, Ponsuksili S. MicroRNAs regulate cellular ATP levels by targeting mitochondrial energy metabolism genes during C2C12 myoblast differentiation. PLoS One. 2015; 10:e0127850. PMID: 26010876.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Rg3 Improves Mitochondrial Function and the Expression of Key Genes Involved in Mitochondrial Biogenesis in C2C12 Myotubes
- Ursolic acid: A promising therapeutic agent for metabolic dysfunction-associated steatotic liver disease via inhibition of SPP1-induced Th17 cell differentiation: Editorial on “Ursolic acid targets secreted phosphoprotein 1 to regulate Th17 cells against metabolic dysfunction-associated steatotic liver disease”
- The Role of Mitochondrial Biogenesis Dysfunction in Diabetic Cardiomyopathy
- Glucagon-Like Peptide-1 Increases Mitochondrial Biogenesis and Function in INS-1 Rat Insulinoma Cells
- Inhibition of indoxyl sulfate-induced intrarenal renin-angiotensin system activation: targeting the aryl hydrocarbon receptor