J Korean Neurosurg Soc.
2022 Mar;65(2):276-286. 10.3340/jkns.2021.0145.
Clinical Outcomes after Spinal Cord Stimulation According to Pain Characteristics
- Affiliations
-
- 1Department of Neurosurgery, Soonchunhyang University Bucheon Hospital, Soonchunhyang University, Bucheon, Korea
- 2Department of Neurosurgery, Incheon St. Mary’s Hospital, The Catholic University of Korea, Incheon, Korea
- 3Department of Psychiatry, Soonchunhyang University Bucheon Hospital, Soonchunhyang University, Bucheon, Korea
- KMID: 2527184
- DOI: http://doi.org/10.3340/jkns.2021.0145
Abstract
Objective
: Spinal cord stimulation (SCS) is an effective treatment for chronic neuropathic pain. However, its clinical efficacy in regard to specific types of pain has not been well studied. The primary objective of this study was to retrospectively analyze the clinical outcomes of paddle-type SCS according to the type of neuropathic pain.
Methods
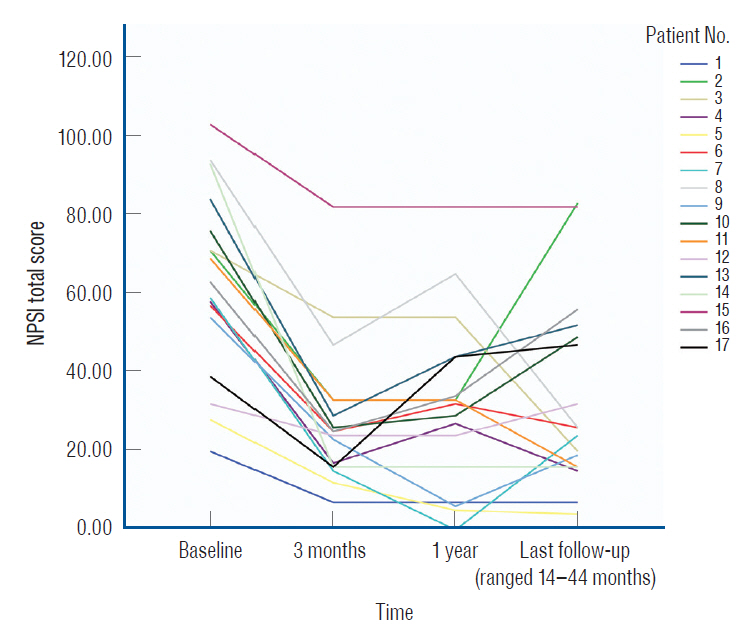
: Seventeen patients who underwent paddle-lead SCS at our hospital were examined. Clinical outcomes were evaluated pre- and postoperatively (3 months, 1 year, and last follow-up) using the Neuropathic Pain Symptom Inventory (NPSI). The NPSI categorizes pain as superficial, deep, paroxysmal, evoked, or dysesthesia and assess the duration of the pain (pain time score). Changes in NPSI scores were compared with change in Visual analogue scale (VAS) scores.
Results
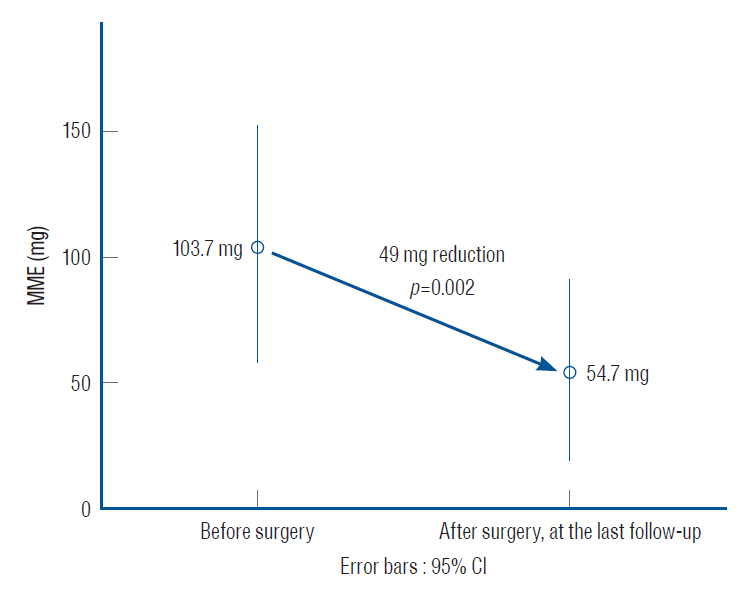
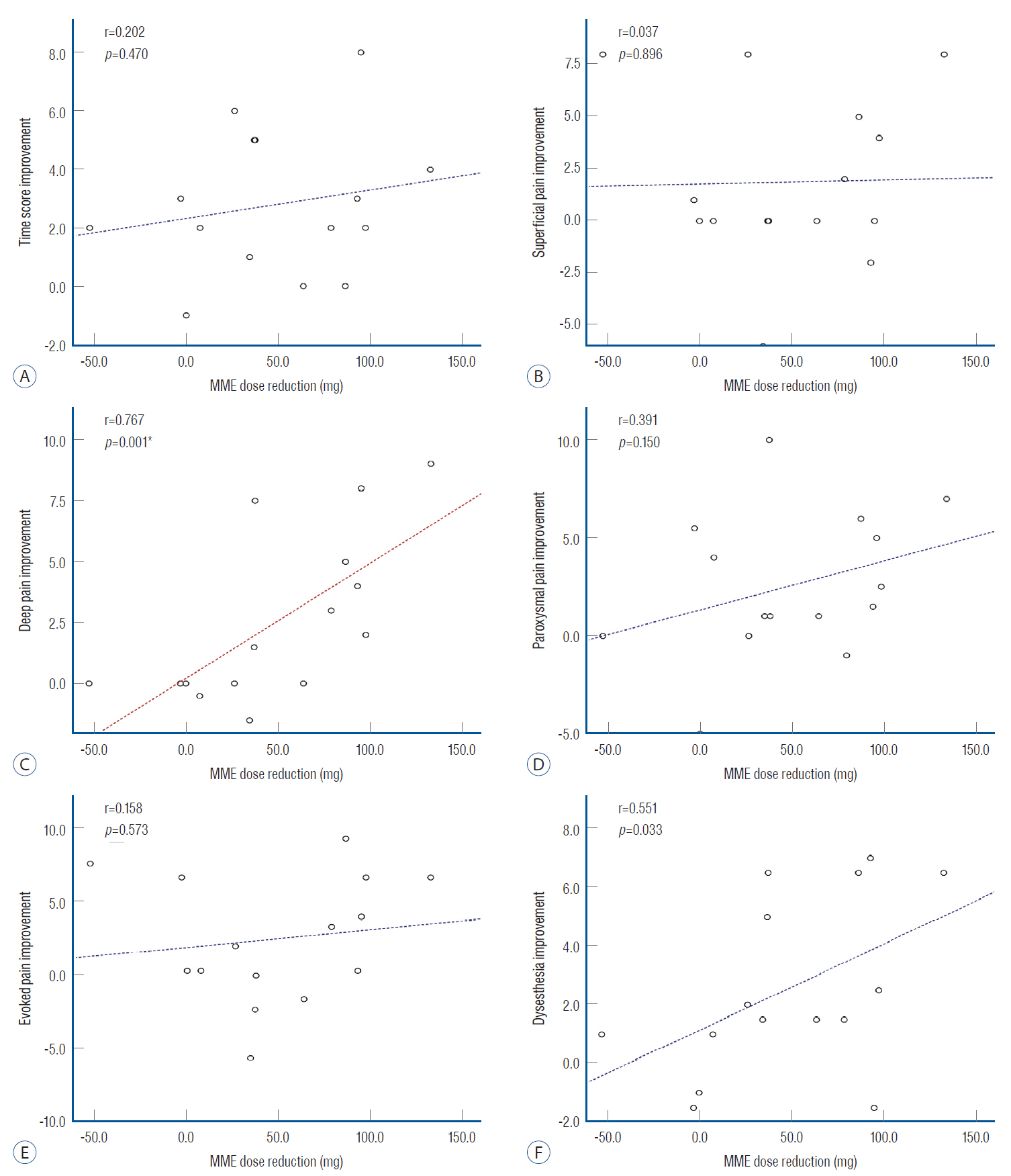
: After SCS, the pain time score improved by 45% (independent t-test, p=0.0002) and the deep pain score improved by 58% (independent t-test, p=0.001). Improvements in the pain time score significantly correlated with improvements in the VAS score (r=0.667, p=0.003, Spearman correlation). Additionally, the morphine milligram equivalent value was markedly lower after vs. before surgery (~49 mg, pared t-test, p=0.002). No preoperative value was associated with clinical outcome.
Conclusion
: The NPSI is a useful tool for evaluating the therapeutic effects of SCS. Chronic use of a paddle-type spinal cord stimulation improved the deep pain and the pain time scores.
Figure
Reference
-
References
1. Aiudi CM, Dunn RY, Burns SM, Roth SA, Opalacz A, Zhang Y, et al. Loss of efficacy to spinal cord stimulator therapy: clinical evidence and possible causes. Pain Physician. 20:E1073–E1080. 2017.2. Apkarian AV, Baliki MN, Geha PY. Towards a theory of chronic pain. Prog Neurobiol. 87:81–97. 2009.
Article3. Baron R, Binder A, Wasner G. Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol. 9:807–819. 2010.
Article4. Basbaum AI, Bautista DM, Scherrer G, Julius D. Cellular and molecular mechanisms of pain. Cell. 139:267–284. 2009.
Article5. Bouhassira D, Attal N, Fermanian J, Alchaar H, Gautron M, Masquelier E, et al. Development and validation of the neuropathic pain symptom inventory. Pain. 108:248–257. 2004.
Article6. Bouhassira D, Wilhelm S, Schacht A, Perrot S, Kosek E, Cruccu G, et al. Neuropathic pain phenotyping as a predictor of treatment response in painful diabetic neuropathy: data from the randomized, double-blind, COMBO-DN study. Pain. 155:2171–2179. 2014.
Article7. Bushnell MC, Ceko M, Low LA. Cognitive and emotional control of pain and its disruption in chronic pain. Nat Rev Neurosci. 14:502–511. 2013.
Article8. Cameron T. Safety and efficacy of spinal cord stimulation for the treatment of chronic pain: a 20-year literature review. J Neurosurg. 100(3 Suppl Spine):254–267. 2004.
Article9. Cohen SP, Mao J. Neuropathic pain: mechanisms and their clinical implications. BMJ. 348:g2323. 2014.
Article10. Crawford B, Bouhassira D, Wong A, Dukes E. Conceptual adequacy of the neuropathic pain symptom inventory in six countries. Health Qual Life Outcomes. 6:62. 2008.
Article11. Deer TR, Skaribas IM, Haider N, Salmon J, Kim C, Nelson C, et al. Effectiveness of cervical spinal cord stimulation for the management of chronic pain. Neuromodulation. 17:265–271. discussion 271. 2014.
Article12. Frey ME, Manchikanti L, Benyamin RM, Schultz DM, Smith HS, Cohen SP. Spinal cord stimulation for patients with failed back surgery syndrome: a systematic review. Pain Physician. 12:379–397. 2009.13. Gee L, Smith HC, Ghulam-Jelani Z, Khan H, Prusik J, Feustel PJ, et al. Spinal cord stimulation for the treatment of chronic pain reduces opioid use and results in superior clinical outcomes when used without opioids. Neurosurgery. 84:217–226. 2019.
Article14. Grider JS, Manchikanti L, Carayannopoulos A, Sharma ML, Balog CC, Harned ME, et al. Effectiveness of spinal cord stimulation in chronic spinal pain: a systematic review. Pain Physician. 19:E33–E54. 2016.15. Holsheimef J, Barolat G. Spinal geometry and paresthesia coverage in spinal cord stimulation. Neuromodulation. 1:129–136. 1998.
Article16. Ikemoto T, Hayashi K, Shiro Y, Arai YC, Marcuzzi A, Costa D, et al. A systematic review of cross-cultural validation of the pain catastrophizing scale. Eur J Pain. 24:1228–1241. 2020.
Article17. Kemler MA, De Vet HC, Barendse GA, Van Den Wildenberg FA, Van Kleef M. The effect of spinal cord stimulation in patients with chronic reflex sympathetic dystrophy: two years’ follow-up of the randomized controlled trial. Ann Neurol. 55:13–18. 2004.
Article18. Kemler MA, de Vet HC, Barendse GA, van den Wildenberg FA, van Kleef M. Spinal cord stimulation for chronic reflex sympathetic dystrophy--five-year follow-up. N Engl J Med. 354:2394–2396. 2006.
Article19. Kumar K, Hunter G, Demeria D. Spinal cord stimulation in treatment of chronic benign pain: challenges in treatment planning and present status, a 22-year experience. Neurosurgery. 58:481–496. discussion 481-496. 2006.
Article20. Kumar K, Taylor RS, Jacques L, Eldabe S, Meglio M, Molet J, et al. Spinal cord stimulation versus conventional medical management for neuropathic pain: a multicentre randomised controlled trial in patients with failed back surgery syndrome. Pain. 132:179–188. 2007.
Article21. Kumar K, Taylor RS, Jacques L, Eldabe S, Meglio M, Molet J, et al. The effects of spinal cord stimulation in neuropathic pain are sustained: a 24-month follow-up of the prospective randomized controlled multicenter trial of the effectiveness of spinal cord stimulation. Neurosurgery. 63:762–770. discussion 770. 2008.
Article22. Kumar K, Rizvi S. Cost-effectiveness of spinal cord stimulation therapy in management of chronic pain. Pain Med. 14:1631–1649. 2013.
Article23. Kupers RC, Van den Oever R, Van Houdenhove B, Vanmechelcn W, Hepp B, Nuttin B, et al. Spinal cord stimulation in Belgium: a nationwide survey on the incidence, indications and therapeutic efficacy by the health insurer. Pain. 56:211–216. 1994.
Article24. Light AR. Normal anatomy and physiology of the spinal cord dorsal horn. Appl Neurophysiol. 51:78–88. 1988.
Article25. Main CJ. Pain assessment in context: a state of the science review of the McGill pain questionnaire 40 years on. Pain. 157:1387–1399. 2016.
Article26. Meyerson BA, Linderoth B. Mechanisms of spinal cord stimulation in neuropathic pain. Neurol Res. 22:285–292. 2000.
Article27. Nielsen S, Degenhardt L, Hoban B, Gisev N. A synthesis of oral morphine equivalents (OME) for opioid utilisation studies. Pharmacoepidemiol Drug Saf. 25:733–737. 2016.
Article28. Nissen M, Ikäheimo TM, Huttunen J, Leinonen V, von Und Zu Fraunberg M. Long-term outcome of spinal cord stimulation in failed back surgery syndrome: 20 years of experience with 224 consecutive patients. Neurosurgery. 84:1011–1018. 2019.
Article29. North RB, Kidd DH, Farrokhi F, Piantadosi SA. Spinal cord stimulation versus repeated lumbosacral spine surgery for chronic pain: a randomized, controlled trial. Neurosurgery. 56:98–106. discussion 106-107. 2005.
Article30. North RB, Kidd DH, Petrucci L, Dorsi MJ. Spinal cord stimulation electrode design: a prospective, randomized, controlled trial comparing percutaneous with laminectomy electrodes: part II-clinical outcomes. Neurosurgery. 57:990–996. discussion 990-996. 2005.
Article31. North RB, Kidd DH, Wimberly RL, Edwin D. Prognostic value of psychological testing in patients undergoing spinal cord stimulation: a prospective study. Neurosurgery. 39:301–310. discussion 310-311. 1996.
Article32. Olson KA, Bedder MD, Anderson VC, Burchiel KJ, Villanueva MR. Psychological variables associated with outcome of spinal cord stimulation trials. Neuromodulation. 1:6–13. 1998.
Article33. Ossipov MH, Dussor GO, Porreca F. Central modulation of pain. J Clin Invest. 120:3779–3787. 2010.
Article34. Padua L, Briani C, Jann S, Nobile-Orazio E, Pazzaglia C, Morini A, et al. Validation of the Italian version of the neuropathic pain symptom inventory in peripheral nervous system diseases. Neurol Sci. 30:99–106. 2009.
Article35. Patel SK, Gozal YM, Saleh MS, Gibson JL, Karsy M, Mandybur GT. Spinal cord stimulation failure: evaluation of factors underlying hardware explantation. J Neurosurg Spine. 32:23–30. 2020.
Article36. Rudd RA, Seth P, David F, Scholl L. Increases in Drug and opioid-involved overdose deaths - United States, 2010-2015. M M W R M o r b Mortal Wkly Rep. 65:1445–1452. 2016.
Article37. Sankarasubramanian V, Harte SE, Chiravuri S, Harris RE, Brummett CM, Patil PG, et al. Objective measures to characterize the physiological effects of spinal cord stimulation in neuropathic pain: a literature review. Neuromodulation. 22:127–148. 2019.
Article38. Schu S, Slotty PJ, Bara G, von Knop M, Edgar D, Vesper J. A prospective, randomised, double-blind, placebo-controlled study to examine the effectiveness of burst spinal cord stimulation patterns for the treatment of failed back surgery syndrome. Neuromodulation. 17:443–450. 2014.
Article39. Simopoulos T, Sharma S, Wootton RJ, Orhurhu V, Aner M, Gill JS. Discontinuation of chronic opiate therapy after successful spinal cord stimulation is highly dependent upon the daily opioid dose. Pain Pract. 19:794–799. 2019.
Article40. Solaro C, Cella M, Signori A, Martinelli V, Radaelli M, Centonze D, et al. Identifying neuropathic pain in patients with multiple sclerosis: a cross-sectional multicenter study using highly specific criteria. J Neurol. 265:828–835. 2018.
Article41. Sommer C, Richter H, Rogausch JP, Frettlöh J, Lungenhausen M, Maier C. A modified score to identify and discriminate neuropathic pain: a study on the German version of the Neuropathic Pain Symptom Inventory (NPSI). BMC Neurol. 11:104. 2011.
Article42. Strang J, Volkow ND, Degenhardt L, Hickman M, Johnson K, Koob GF, et al. Opioid use disorder. Nat Rev Dis Primers. 6:3. 2020.
Article43. Wiech K. Deconstructing the sensation of pain: the influence of cognitive processes on pain perception. Science. 354:584–587. 2016.
Article44. Wolter T. Spinal cord stimulation for neuropathic pain: current perspectives. J Pain Res. 7:651–663. 2014.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Spinal cord stimulation for a patient with neuropathic pain related to congenital syringomyelia
- Spinal Cord Stimulation in Pain Management: A Review
- Spontaneous Lead Breakage in Implanted Spinal Cord Stimulation Systems
- Limitations of spinal cord stimulation for pain management
- Spinal cord stimulation for neuropathic pain following idiopathic transverse myelitis: A case report