Obstet Gynecol Sci.
2022 Jan;65(1):37-45. 10.5468/ogs.21121.
A comparative study of smica in various body fluids of diagnosed cervical cancer patients and healthy women
- Affiliations
-
- 1Department of Obstetric and Gynecology, Dr. DY Patil Medical College, Hospital & Research Center, Kolhapur, India
- 2Department of Stem Cells and Regenerative Medicine, DY Patil Education Society, Deemed University, Kolhapur, India
- 3Department of Pathology, Shri Siddhivinayak Ganpati Cancer Hospital, Miraj, India
- 4Department of Preventive Oncology, Shri Siddhivinayak Ganpati Cancer Hospital, Miraj, India
- 5Department of Obstetrics and Gynaecology, Somshekhar Hospital, Miraj, India
- KMID: 2524683
- DOI: http://doi.org/10.5468/ogs.21121
Abstract
Objective
Cervical cancer (CC) is a major public health problem in women, and its early detection can help reduce morbidity and mortality. The objective of this study was to compare serum levels of soluble major histocompatibility complex class I-related chain A (sMICA) levels in various body fluids between women diagnosed with CC and healthy women.
Methods
A case-control study was conducted at a tertiary care hospital and a cancer center in Kolhapur, India. Overall, 150 individuals (100 CC patients and 50 healthy women) participated after providing informed written consent. Demographic data, histopathology history, parity, and tumor, node, and metastasis (TNM) staging data were collected. Pap smears, saliva, blood, and urine samples were collected. Pap smears were examined microscopically, and sMICA levels in all samples were determined by enzyme-linked immunoassay (ELISA).
Results
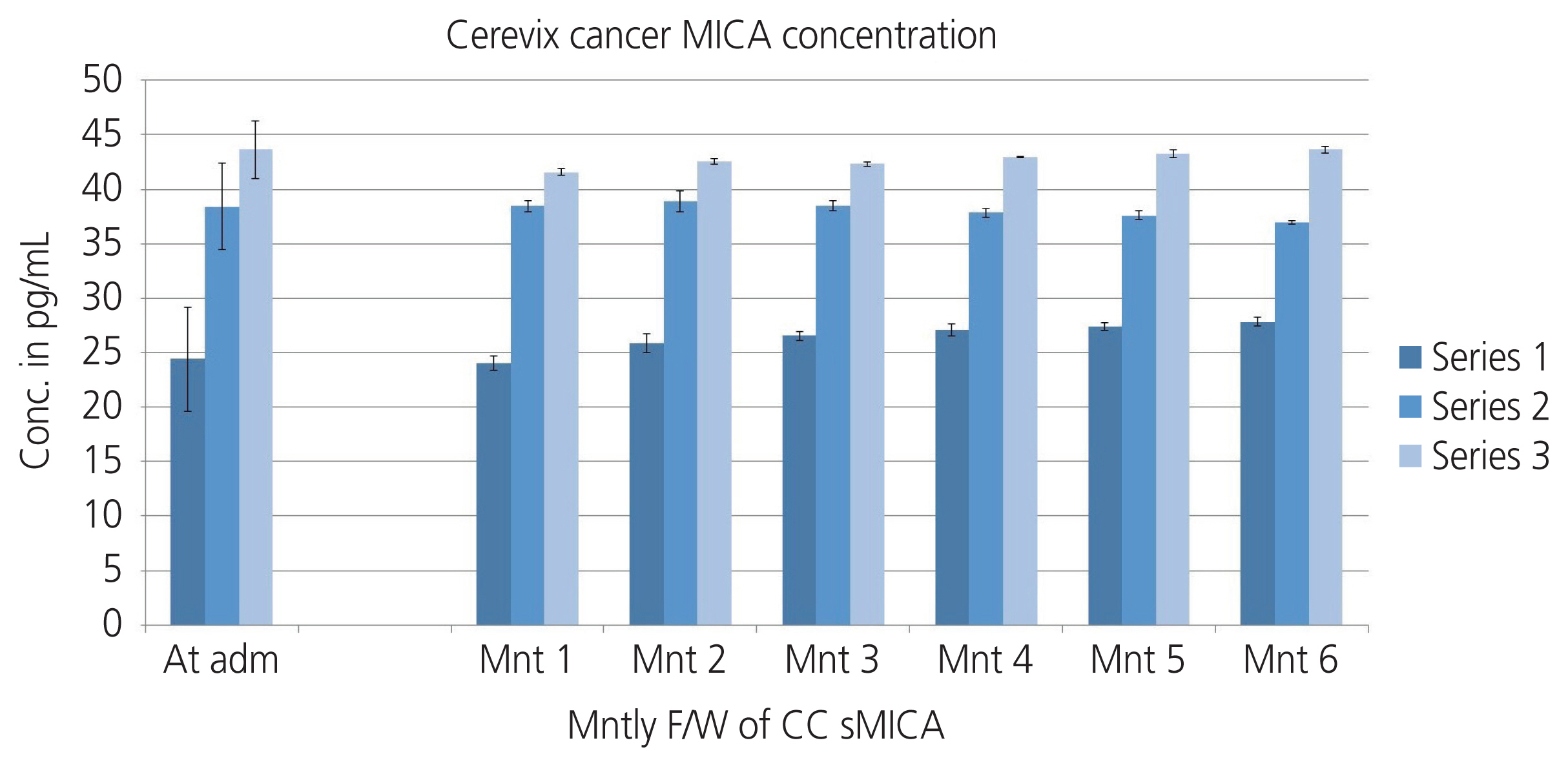
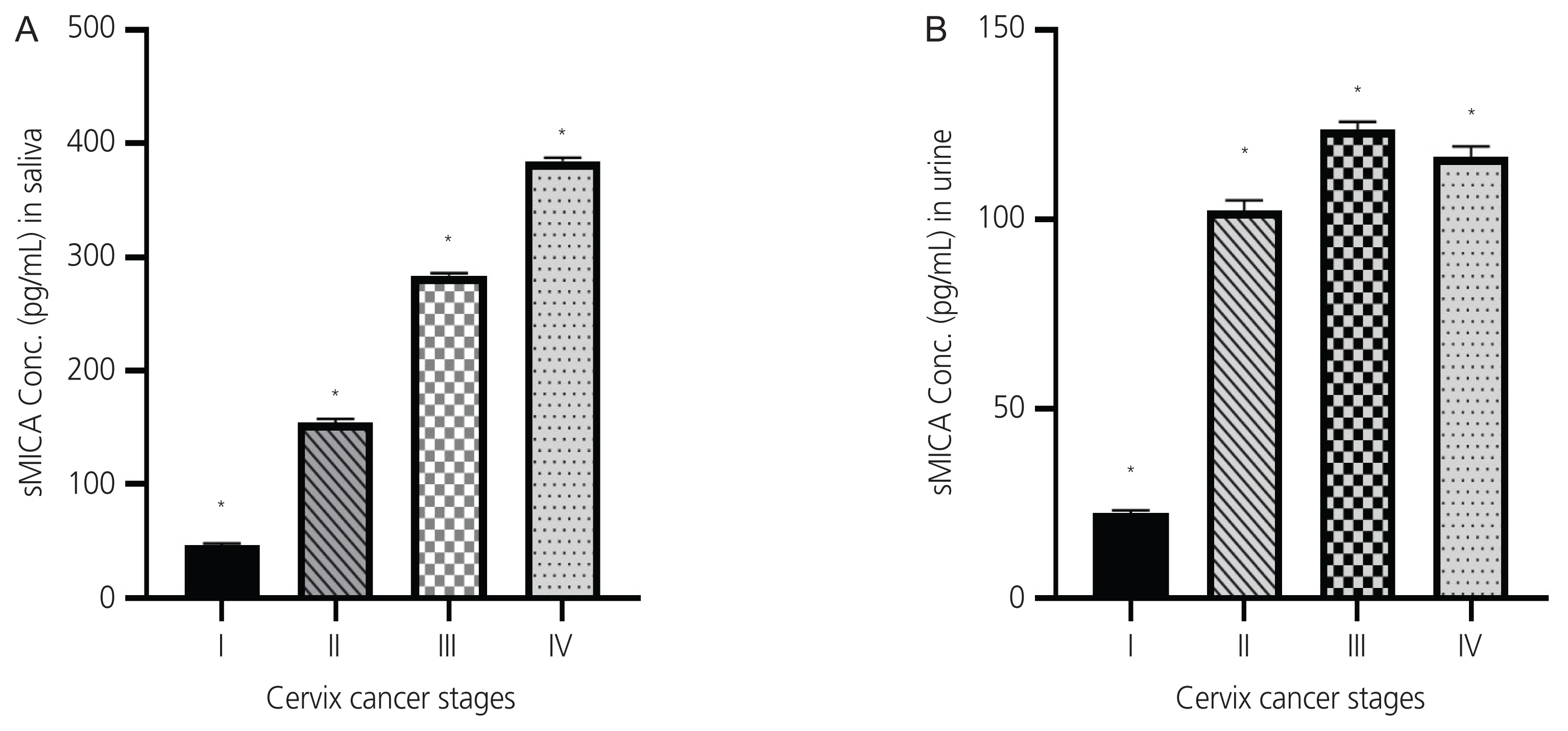
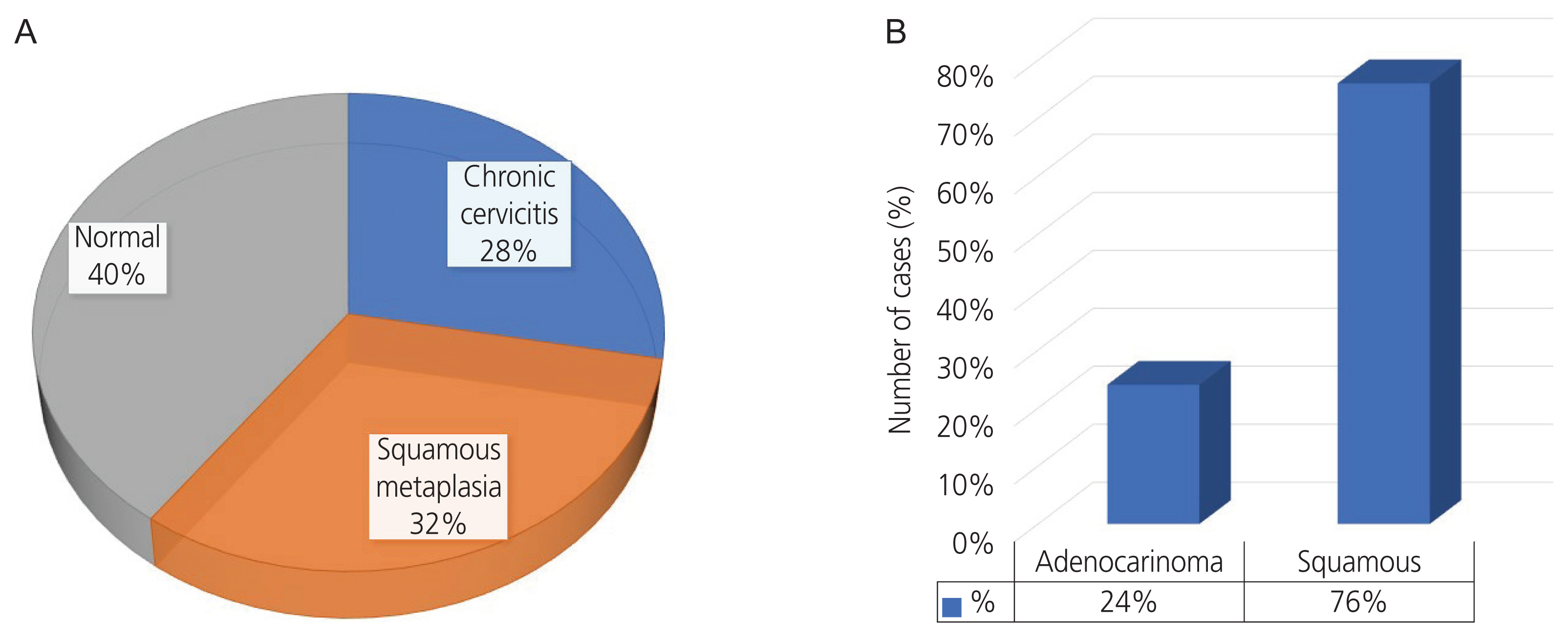
The mean age of women with cervical cancer was 49.86±8.18 years. Squamous cell carcinoma (70%) was the most common histological variant in CC patients. Serum soluble sMICA levels differed significantly with parity and TNM staging (P<0.05). Mean levels of sMICA were significantly different in samples (CC cases vs. healthy patients; saliva: 166.721±108.718 vs. 0.039±0.005 pg/mL; urine: 82.921±45.580 vs. 0.010±0.005 pg/mL; serum: 35.756±10.799 vs. 0.039±0.005 pg/mL, P<0.001).
Conclusion
Levels of sMICA in body fluids can be considered as a diagnostic or prognostic tool to determine disease progression or tumor regression.
Figure
Reference
-
References
1. Srivastava AN, Misra JS, Srivastava S, Das BC, Gupta S. Cervical cancer screening in rural India: status & current concepts. Indian J Med Res. 2018; 148:687–96.2. Arbyn M, Weiderpass E, Bruni L, de Sanjosé S, Saraiya M, Ferlay J, et al. Estimates of incidence and mortality of cervical cancer in 2018: a worldwide analysis. Lancet Glob Health. 2020; 8:e191–203.
Article3. Bobdey S, Sathwara J, Jain A, Balasubramaniam G. Burden of cervical cancer and role of screening in India. Indian J Med Paediatr Oncol. 2016; 37:278–85.
Article4. Parish SJ, Nappi RE, Krychman ML, Kellogg-Spadt S, Simon JA, Goldstein JA, et al. Impact of vulvovaginal health on postmenopausal women: a review of surveys on symptoms of vulvovaginal atrophy. Int J Womens Health. 2013; 5:437–47.
Article5. Dalal PK, Agarwal M. Postmenopausal syndrome. Indian J Psychiatry. 2015; 57(Suppl 2):S222–32.
Article6. Catarino R, Petignat P, Dongui G, Vassilakos P. Cervical cancer screening in developing countries at a crossroad: emerging technologies and policy choices. World J Clin Oncol. 2015; 6:281–90.
Article7. WHO guidelines WHO guidelines for screening and treatment of precancerous lesions for cervical cancer prevention. Geneva: World Health Organization;2013.8. World Health Organization. Comprehensive cervical cancer control: a guide to essential practice. 2nd ed. Geneva: World Health Organization;2014.9. Mishra GA, Pimple SA, Shastri SS. An overview of prevention and early detection of cervical cancers. Indian J Med Paediatr Oncol. 2011; 32:125–32.
Article10. Baranwal AK, Mehra NK. Major histocompatibility complex class i chain-related a (MICA) molecules: relevance in solid organ transplantation. Front Immunol. 2017; 8:182.
Article11. Zhao Y, Chen N, Yu Y, Zhou L, Niu C, Liu Y, et al. Prognostic value of MICA/B in cancers: a systematic review and meta-analysis. Oncotarget. 2017; 8:96384–95.
Article12. Samuels S, Ferns DM, Meijer D, van Straalen JP, Buist MR, Zijlmans HJ, et al. High levels of soluble MICA are significantly related to increased disease-free and disease-specific survival in patients with cervical adenocarcinoma. Tissue Antigens. 2015; 85:476–83.
Article13. Safaeian M, Solomon D, Castle PE. Cervical cancer prevention--cervical screening: science in evolution. Obstet Gynecol Clin North Am. 2007; 34:739–60. ix
Article14. Sreedevi A, Javed R, Dinesh A. Epidemiology of cervical cancer with special focus on India. Int J Womens Health. 2015; 7:405–14.15. Mhaske M, Jawadekar SJ, Saundale SG. Study of association of some risk factors & cervical dysplasia/cancer among rural women. Natl J Community Med. 2011; 2:209–12.16. Wang W, Zhang F, Hu K, Hou X. Image-guided, intensity-modulated radiation therapy in definitive radiotherapy for 1433 patients with cervical cancer. Gynecol Oncol. 2018; 151:444–8.
Article17. Kashyap N, Krishnan N, Kaur S, Ghai S. Risk factors of cervical cancer: a case-control study. Asia Pac J Oncol Nurs. 2019; 6:308–14.
Article18. Vinh-Hung V, Bourgain C, Vlastos G, Cserni G, De Ridder M, Storme G, et al. Prognostic value of histopathology and trends in cervical cancer: a SEER population study. BMC Cancer. 2007; 7:164.
Article19. Schmiedel D, Mandelboim O. NKG2D ligands-critical targets for cancer immune escape and therapy. Front Immunol. 2018; 9:2040.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Mutation Analysis of K-ras Codon 12 in Malignant Effusion
- Epidemiologic characteristics of cervical cancer in Korean women
- Comparison of Quality of Life and Sexuality between Cervical Cancer Survivors and Healthy Women
- Relationship between Mothers' Diagnosis of Cervical Cancer and Attitudes toward Preventing Cervical Cancer in Their Pubertal Daughters
- Effect of Pap smear screening on cervical cancer stage at diagnosis: results from the Korean National Cancer Screening Program