Korean Circ J.
2022 Jan;52(1):34-46. 10.4070/kcj.2021.0363.
Functional Angioplasty: Definitions, Historical Overview, and Future Perspectives
- Affiliations
-
- 1Division of Cardiology, Department of Medicine, Gangneung Asan Hospital, University of Ulsan College of Medicine, Gangneung, Korea
- 2Division of Cardiology, Department of Medicine, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- KMID: 2524168
- DOI: http://doi.org/10.4070/kcj.2021.0363
Abstract
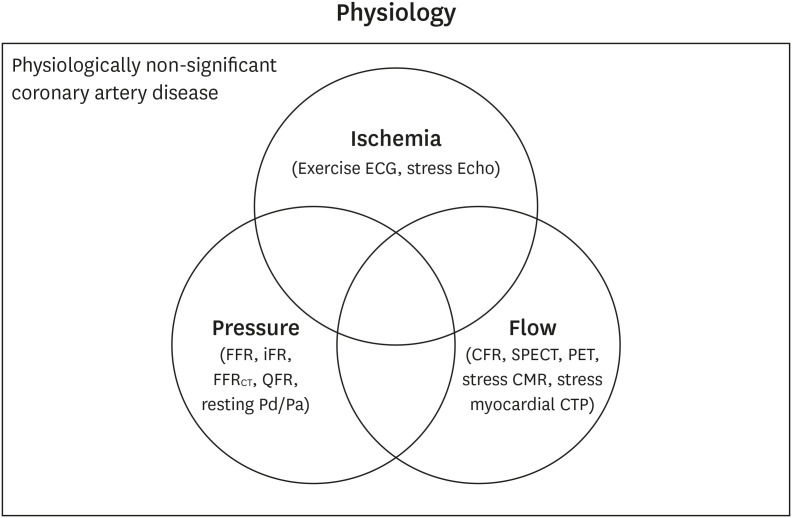
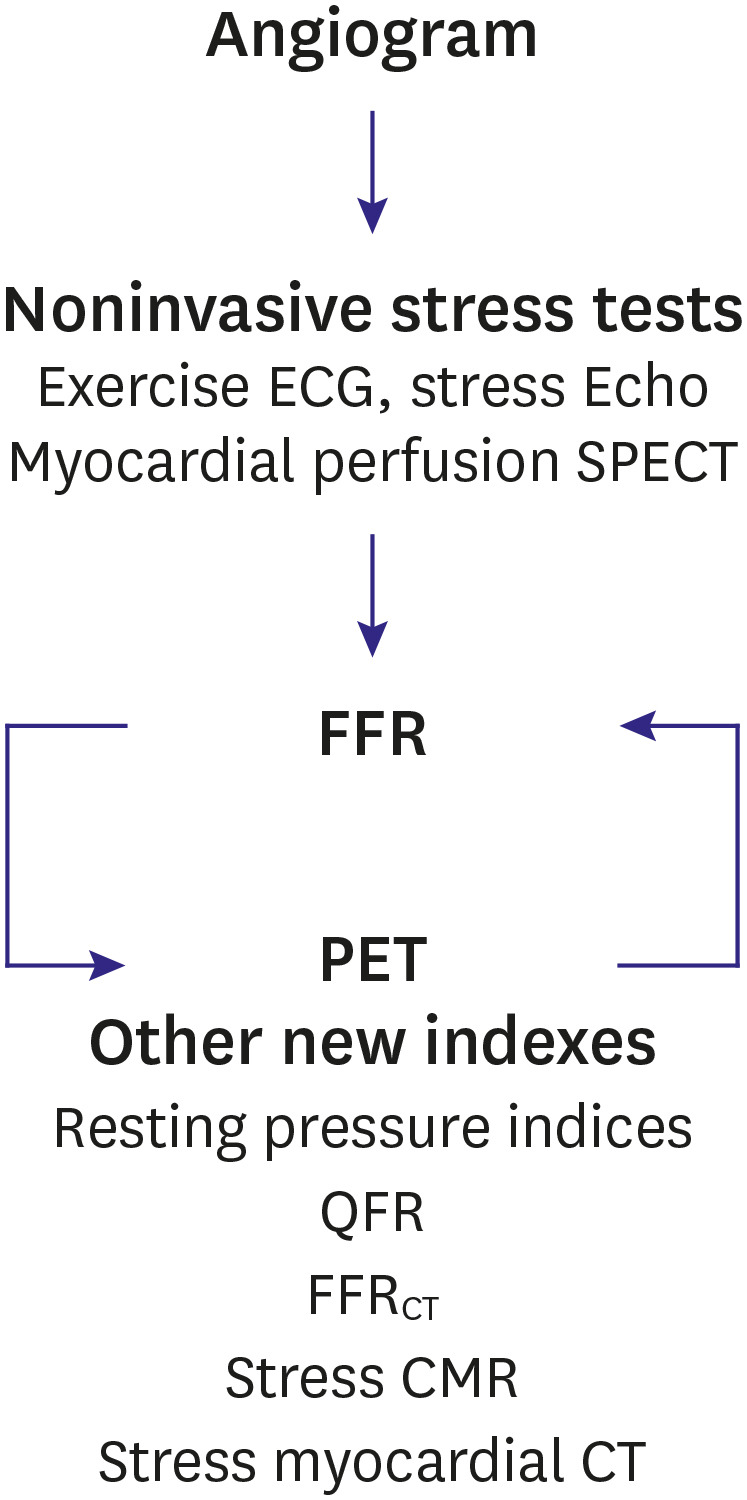
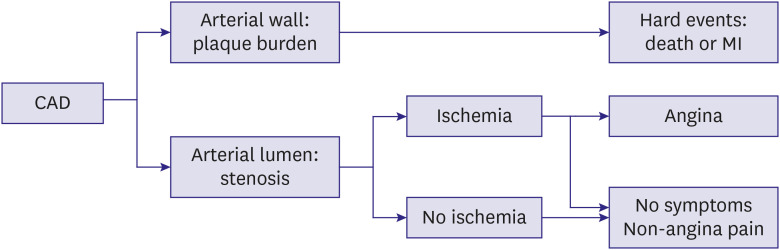
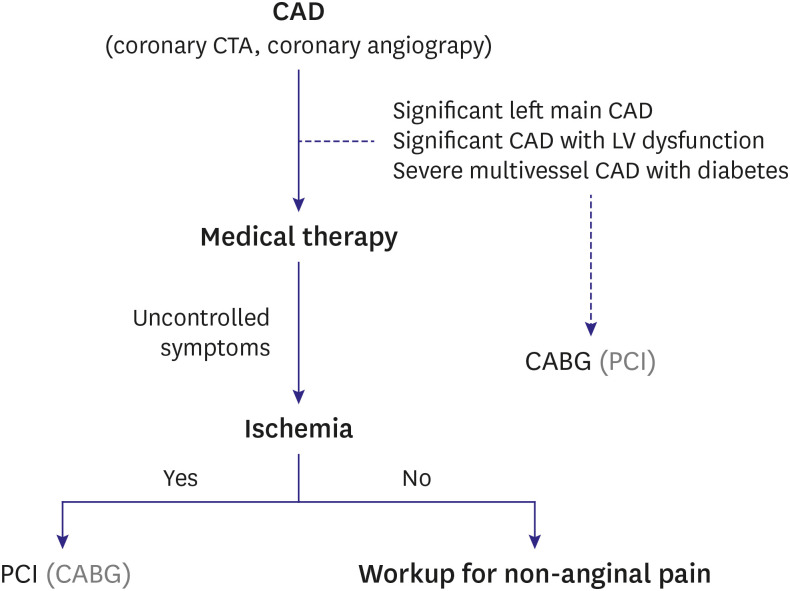
- Percutaneous coronary intervention (PCI) is used to treat obstructive coronary artery disease (CAD). The role of PCI is well defined in acute coronary syndrome, but that for stable CAD remains debatable. Although PCI generally relieves angina in patients with stable CAD, it may not change its prognosis. The extent and severity of CAD are major determinants of prognosis, and complete revascularization (CR) of all ischemia-causing lesions might improve outcomes. Several studies have shown better outcomes with CR than with incomplete revascularization, emphasizing the importance of functional angioplasty. However, different definitions of inducible myocardial ischemia have been used across studies, making their comparison difficult. Various diagnostic tools have been used to estimate the presence, extent, and severity of inducible myocardial ischemia. However, to date, there are no agreed reference standards of inducible myocardial ischemia. The hallmarks of inducible myocardial ischemia such as electrocardiographic changes and regional wall motion abnormalities may be more clinically relevant as the reference standard to define ischemia-causing lesions. In this review, we summarize studies regarding myocardial ischemia, PCI guidance, and possible explanations for similar findings across studies. Also, we provide some insights into the ideal definition of inducible myocardial ischemia and highlight the appropriate PCI strategy.
Figure
Reference
-
1. Fihn SD, Blankenship JC, Alexander KP, et al. 2014 ACC/AHA/AATS/PCNA/SCAI/STS focused update of the guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines, and the American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2014; 64:1929–1949. PMID: 25077860.2. Neumann FJ, Sousa-Uva M, Ahlsson A, et al. 2018 ESC/EACTS guidelines on myocardial revascularization. Eur Heart J. 2019; 40:87–165. PMID: 30165437.3. Bangalore S, Maron DJ, Stone GW, Hochman JS. Routine revascularization versus initial medical therapy for stable ischemic heart disease: a systematic review and meta-analysis of randomized trials. Circulation. 2020; 142:841–857. PMID: 32794407.4. Farooq V, Serruys PW, Bourantas CV, et al. Quantification of incomplete revascularization and its association with five-year mortality in the synergy between percutaneous coronary intervention with taxus and cardiac surgery (SYNTAX) trial validation of the residual SYNTAX score. Circulation. 2013; 128:141–151. PMID: 23766350.5. Ahn JM, Park DW, Lee CW, et al. Comparison of stenting versus bypass surgery according to the completeness of revascularization in severe coronary artery disease: patient-level pooled analysis of the SYNTAX, PRECOMBAT, and BEST trials. JACC Cardiovasc Interv. 2017; 10:1415–1424. PMID: 28728654.6. Kobayashi Y, Nam CW, Tonino PA, et al. The prognostic value of residual coronary stenoses after functionally complete revascularization. J Am Coll Cardiol. 2016; 67:1701–1711. PMID: 27056776.7. Choi KH, Lee JM, Koo BK, et al. Prognostic implication of functional incomplete revascularization and residual functional SYNTAX score in patients with coronary artery disease. JACC Cardiovasc Interv. 2018; 11:237–245. PMID: 29361444.8. Hearse DJ. Myocardial ischaemia: can we agree on a definition for the 21st century? Cardiovasc Res. 1994; 28:1737–1744. PMID: 7867024.9. Nesto RW, Kowalchuk GJ. The ischemic cascade: temporal sequence of hemodynamic, electrocardiographic and symptomatic expressions of ischemia. Am J Cardiol. 1987; 59:23C–30C.10. Knuuti J, Wijns W, Saraste A, et al. 2019 ESC guidelines for the diagnosis and management of chronic coronary syndromes. Eur Heart J. 2020; 41:407–477. PMID: 31504439.11. Saraiva RM, Pacheco NP, Pereira TO, et al. Left atrial structure and function predictors of new-onset atrial fibrillation in patients with chagas disease. J Am Soc Echocardiogr. 2020; 33:1363–1374.e1. PMID: 32747223.12. Nelson AJ, Ardissino M, Psaltis PJ. Current approach to the diagnosis of atherosclerotic coronary artery disease: more questions than answers. Ther Adv Chronic Dis. 2019; 10:2040622319884819. PMID: 31700595.13. Gaba P, Gersh BJ, Ali ZA, Moses JW, Stone GW. Complete versus incomplete coronary revascularization: definitions, assessment and outcomes. Nat Rev Cardiol. 2021; 18:155–168. PMID: 33067581.14. Proudfit WL, Shirey EK, Sones FM Jr. Selective cine coronary arteriography. Correlation with clinical findings in 1,000 patients. Circulation. 1966; 33:901–910. PMID: 5942973.15. Loop FD, Cosgrove DM, Lytle BW, et al. An 11 year evolution of coronary arterial surgery (1968-1978). Ann Surg. 1979; 190:444–455. PMID: 39520.16. Detre K, Hultgren H, Takaro T. Veterans administration cooperative study of surgery for coronary arterial occlusive disease. III. Methods and baseline characteristics, including experience with medical treatment. Am J Cardiol. 1977; 40:212–225. PMID: 879029.17. Varnauskas E. Twelve-year follow-up of survival in the randomized European Coronary Surgery Study. N Engl J Med. 1988; 319:332–337. PMID: 3260659.18. Coronary artery surgery study (CASS): a randomized trial of coronary artery bypass surgery. Survival data. Circulation. 1983; 68:939–950. PMID: 6137292.19. Gould KL, Lipscomb K. Effects of coronary stenoses on coronary flow reserve and resistance. Am J Cardiol. 1974; 34:48–55. PMID: 4835753.20. Uren NG, Melin JA, De Bruyne B, Wijns W, Baudhuin T, Camici PG. Relation between myocardial blood flow and the severity of coronary-artery stenosis. N Engl J Med. 1994; 330:1782–1788. PMID: 8190154.21. Mann FC, Herrick JF, Essex HE, Baldes EJ. The effect on the blood flow of decreasing the lumen of a blood vessel. Surgery. 1938; 4:249–252.22. May AG, De Weese JA, Rob CG. Hemodynamic effects of arterial stenosis. Surgery. 1963; 53:513–524. PMID: 13934072.23. Patel MR, Calhoon JH, Dehmer GJ, et al. ACC/AATS/AHA/ASE/ASNC/SCAI/SCCT/STS 2017 appropriate use criteria for coronary revascularization in patients with stable ischemic heart disease: a report of the American College of Cardiology Appropriate Use Criteria Task Force, American Association for Thoracic Surgery, American Heart Association, American Society of Echocardiography, American Society of Nuclear Cardiology, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Computed Tomography, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2017; 69:2212–2241. PMID: 28291663.24. Gould KL, Johnson NP, Bateman TM, et al. Anatomic versus physiologic assessment of coronary artery disease. Role of coronary flow reserve, fractional flow reserve, and positron emission tomography imaging in revascularization decision-making. J Am Coll Cardiol. 2013; 62:1639–1653. PMID: 23954338.25. Johnson NP, Kirkeeide RL, Gould KL. Is discordance of coronary flow reserve and fractional flow reserve due to methodology or clinically relevant coronary pathophysiology? JACC Cardiovasc Imaging. 2012; 5:193–202. PMID: 22340827.26. Lee JM, Kim CH, Koo BK, et al. Integrated myocardial perfusion imaging diagnostics improve detection of functionally significant coronary artery stenosis by 13N-ammonia positron emission tomography. Circ Cardiovasc Imaging. 2016; 9:e004768. PMID: 27609817.27. Danad I, Uusitalo V, Kero T, et al. Quantitative assessment of myocardial perfusion in the detection of significant coronary artery disease: cutoff values and diagnostic accuracy of quantitative [15O]H2O PET imaging. J Am Coll Cardiol. 2014; 64:1464–1475. PMID: 25277618.28. de Waard GA, Danad I, Petraco R, et al. Fractional flow reserve, instantaneous wave-free ratio, and resting Pd/Pa compared with [15O]H2O positron emission tomography myocardial perfusion imaging: a PACIFIC trial sub-study. Eur Heart J. 2018; 39:4072–4081. PMID: 30452609.29. Smalling RW, Kelley K, Kirkeeide RL, Fisher DJ. Regional myocardial function is not affected by severe coronary depressurization provided coronary blood flow is maintained. J Am Coll Cardiol. 1985; 5:948–955. PMID: 3973297.30. Canty JM Jr. Coronary pressure-function and steady-state pressure-flow relations during autoregulation in the unanesthetized dog. Circ Res. 1988; 63:821–836. PMID: 3168181.31. Di Gioia G, Melin JA, De Bruyne B. Coronary autoregulatory plateau in humans. J Am Coll Cardiol. 2020; 76:1270–1271. PMID: 32883422.32. Grüntzig AR, Senning A, Siegenthaler WE. Nonoperative dilatation of coronary-artery stenosis: percutaneous transluminal coronary angioplasty. N Engl J Med. 1979; 301:61–68. PMID: 449946.33. King SB 3rd, Schlumpf M. Ten-year completed follow-up of percutaneous transluminal coronary angioplasty: the early Zurich experience. J Am Coll Cardiol. 1993; 22:353–360. PMID: 8335804.34. Fischman DL, Leon MB, Baim DS, et al. A randomized comparison of coronary-stent placement and balloon angioplasty in the treatment of coronary artery disease. N Engl J Med. 1994; 331:496–501. PMID: 8041414.35. Taglieri N, Bruno AG, Ghetti G, et al. Target lesion failure with current drug-eluting stents: evidence from a comprehensive network meta-analysis. JACC Cardiovasc Interv. 2020; 13:2868–2878. PMID: 33357524.36. RITA-2 trial participants. Coronary angioplasty versus medical therapy for angina: the second Randomised Intervention Treatment of Angina (RITA-2) trial. Lancet. 1997; 350:461–468. PMID: 9274581.37. Henderson RA, Pocock SJ, Clayton TC, et al. Seven-year outcome in the RITA-2 trial: coronary angioplasty versus medical therapy. J Am Coll Cardiol. 2003; 42:1161–1170. PMID: 14522473.38. Al-Lamee R, Thompson D, Dehbi HM, et al. Percutaneous coronary intervention in stable angina (ORBITA): a double-blind, randomised controlled trial. Lancet. 2018; 391:31–40. PMID: 29103656.39. De Bruyne B, Pijls NH, Kalesan B, et al. Fractional flow reserve-guided PCI versus medical therapy in stable coronary disease. N Engl J Med. 2012; 367:991–1001. PMID: 22924638.40. Xaplanteris P, Fournier S, Pijls NH, et al. Five-year outcomes with PCI guided by fractional flow reserve. N Engl J Med. 2018; 379:250–259. PMID: 29785878.41. Maron DJ, Hochman JS, Reynolds HR, et al. Initial invasive or conservative strategy for stable coronary disease. N Engl J Med. 2020; 382:1395–1407. PMID: 32227755.42. Savage MP, Fischman DL, Rake R, et al. Efficacy of coronary stenting versus balloon angioplasty in small coronary arteries. J Am Coll Cardiol. 1998; 31:307–311. PMID: 9462572.43. Morice MC, Serruys PW, Barragan P, et al. Long-term clinical outcomes with sirolimus-eluting coronary stents: five-year results of the RAVEL trial. J Am Coll Cardiol. 2007; 50:1299–1304. PMID: 17903626.44. Morice MC, Colombo A, Meier B, et al. Sirolimus- vs paclitaxel-eluting stents in de novo coronary artery lesions: the REALITY trial: a randomized controlled trial. JAMA. 2006; 295:895–904. PMID: 16493102.45. Tonino PA, De Bruyne B, Pijls NH, et al. Fractional flow reserve versus angiography for guiding percutaneous coronary intervention. N Engl J Med. 2009; 360:213–224. PMID: 19144937.46. van Nunen LX, Zimmermann FM, Tonino PA, et al. Fractional flow reserve versus angiography for guidance of PCI in patients with multivessel coronary artery disease (FAME): 5-year follow-up of a randomised controlled trial. Lancet. 2015; 386:1853–1860. PMID: 26333474.47. Götberg M, Christiansen EH, Gudmundsdottir IJ, et al. Instantaneous wave-free ratio versus fractional flow reserve to guide PCI. N Engl J Med. 2017; 376:1813–1823. PMID: 28317438.48. Davies JE, Sen S, Dehbi HM, et al. Use of the instantaneous wave-free ratio or fractional flow reserve in PCI. N Engl J Med. 2017; 376:1824–1834. PMID: 28317458.49. Boden WE, O’Rourke RA, Teo KK, et al. Optimal medical therapy with or without PCI for stable coronary disease. N Engl J Med. 2007; 356:1503–1516. PMID: 17387127.50. Chaitman BR, Hardison RM, Adler D, et al. The bypass angioplasty revascularization investigation 2 diabetes randomized trial of different treatment strategies in type 2 diabetes mellitus with stable ischemic heart disease: impact of treatment strategy on cardiac mortality and myocardial infarction. Circulation. 2009; 120:2529–2540. PMID: 19920001.51. Bangalore S, Pencina MJ, Kleiman NS, Cohen DJ. Prognostic implications of procedural vs spontaneous myocardial infarction: results from the Evaluation of Drug Eluting Stents and Ischemic Events (EVENT) registry. Am Heart J. 2013; 166:1027–1034. PMID: 24268217.52. Prasad A, Gersh BJ, Bertrand ME, et al. Prognostic significance of periprocedural versus spontaneously occurring myocardial infarction after percutaneous coronary intervention in patients with acute coronary syndromes: an analysis from the ACUITY (Acute Catheterization and Urgent Intervention Triage Strategy) trial. J Am Coll Cardiol. 2009; 54:477–486. PMID: 19628125.53. Silvain J, Zeitouni M, Paradies V, et al. Procedural myocardial injury, infarction and mortality in patients undergoing elective PCI: a pooled analysis of patient-level data. Eur Heart J. 2021; 42:323–334. PMID: 33257958.54. Stone GW, Selker HP, Thiele H, et al. Relationship between infarct size and outcomes following primary PCI: patient-level analysis from 10 randomized trials. J Am Coll Cardiol. 2016; 67:1674–1683. PMID: 27056772.55. Brener SJ, Lytle BW, Schneider JP, Ellis SG, Topol EJ. Association between CK-MB elevation after percutaneous or surgical revascularization and three-year mortality. J Am Coll Cardiol. 2002; 40:1961–1967. PMID: 12475456.56. Novack V, Pencina M, Cohen DJ, et al. Troponin criteria for myocardial infarction after percutaneous coronary intervention. Arch Intern Med. 2012; 172:502–508. PMID: 22371874.57. Gregson J, Stone GW, Ben-Yehuda O, et al. Implications of alternative definitions of peri-procedural myocardial infarction after coronary revascularization. J Am Coll Cardiol. 2020; 76:1609–1621. PMID: 33004126.58. Warren J, Mehran R, Yu J, et al. Incidence and impact of totally occluded culprit coronary arteries in patients presenting with non-ST-segment elevation myocardial infarction. Am J Cardiol. 2015; 115:428–433. PMID: 25542393.59. Habib GB, Heibig J, Forman SA, et al. Influence of coronary collateral vessels on myocardial infarct size in humans. Results of phase I thrombolysis in myocardial infarction (TIMI) trial. Circulation. 1991; 83:739–746. PMID: 1900223.60. Billinger M, Kloos P, Eberli FR, Windecker S, Meier B, Seiler C. Physiologically assessed coronary collateral flow and adverse cardiac ischemic events: a follow-up study in 403 patients with coronary artery disease. J Am Coll Cardiol. 2002; 40:1545–1550. PMID: 12427404.61. de Marchi SF. Determinants of human coronary collaterals. Curr Cardiol Rev. 2014; 10:24–28. PMID: 23638830.62. Fox K, Ford I, Steg PG, et al. Ivabradine in stable coronary artery disease without clinical heart failure. N Engl J Med. 2014; 371:1091–1099. PMID: 25176136.63. Morrow DA, Scirica BM, Karwatowska-Prokopczuk E, et al. Effects of ranolazine on recurrent cardiovascular events in patients with non-ST-elevation acute coronary syndromes: the MERLIN-TIMI 36 randomized trial. JAMA. 2007; 297:1775–1783. PMID: 17456819.64. IONA Study Group. Effect of nicorandil on coronary events in patients with stable angina: the Impact Of Nicorandil in Angina (IONA) randomised trial. Lancet. 2002; 359:1269–1275. PMID: 11965271.65. O’Fee K, Panza JA, Brown DL. Association of inducible myocardial ischemia with long-term mortality and benefit from coronary artery bypass graft surgery in ischemic cardiomyopathy: ten-year follow-up of the STICH trial. Circulation. 2021; 143:205–207. PMID: 33428429.66. Mortensen MB, Dzaye O, Steffensen FH, et al. Impact of plaque burden versus stenosis on ischemic events in patients with coronary atherosclerosis. J Am Coll Cardiol. 2020; 76:2803–2813. PMID: 33303068.67. Bittencourt MS, Hulten E, Ghoshhajra B, et al. Prognostic value of nonobstructive and obstructive coronary artery disease detected by coronary computed tomography angiography to identify cardiovascular events. Circ Cardiovasc Imaging. 2014; 7:282–291. PMID: 24550435.68. Nakagomi A, Celermajer DS, Lumley T, Freedman SB. Angiographic severity of coronary narrowing is a surrogate marker for the extent of coronary atherosclerosis. Am J Cardiol. 1996; 78:516–519. PMID: 8806334.69. Mancini GBJ, Hartigan PM, Shaw LJ, et al. Predicting outcome in the COURAGE trial (Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation): coronary anatomy versus ischemia. JACC Cardiovasc Interv. 2014; 7:195–201. PMID: 24440015.70. Timmis A, Raharja A, Archbold RA, Mathur A. Validity of inducible ischaemia as a surrogate for adverse outcomes in stable coronary artery disease. Heart. 2018; 104:1733–1738. PMID: 29875140.71. Spertus JA, Jones PG, Maron DJ, et al. Health-status outcomes with invasive or conservative care in coronary disease. N Engl J Med. 2020; 382:1408–1419. PMID: 32227753.72. Shaw LJ, Berman DS, Maron DJ, et al. Optimal medical therapy with or without percutaneous coronary intervention to reduce ischemic burden: results from the Clinical Outcomes Utilizing Revascularization and Aggressive Drug Evaluation (COURAGE) trial nuclear substudy. Circulation. 2008; 117:1283–1291. PMID: 18268144.73. Al-Lamee RK, Shun-Shin MJ, Howard JP, et al. Dobutamine stress echocardiography ischemia as a predictor of the placebo-controlled efficacy of percutaneous coronary intervention in stable coronary artery disease: the stress echocardiography-stratified analysis of ORBITA. Circulation. 2019; 140:1971–1980. PMID: 31707827.74. Øhrn AM, Nielsen CS, Schirmer H, Stubhaug A, Wilsgaard T, Lindekleiv H. Pain tolerance in persons with recognized and unrecognized myocardial infarction: a population-based, cross-sectional study. J Am Heart Assoc. 2016; 5:e003846. PMID: 28003255.75. Deedwania PC, Carbajal EV. Silent myocardial ischemia. A clinical perspective. Arch Intern Med. 1991; 151:2373–2382. PMID: 1746993.76. Bugiardini R, Cenko E. Persisting chest pain in nonobstructive coronary artery disease. Eur Heart J Qual Care Clin Outcomes. 2016; 2:69–71. PMID: 29474630.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Current concepts in genioplasty: surgical techniques, indications, and future perspectives
- Erratum: Leptospirosis in the Republic of Korea: Historical Perspectives, Current Status and Future Challenges
- The Importance of Smoking Definitions for the Study of Adolescent Smoking Behavior
- Historical Perspectives of the Korean Society for Thoracic and Cardiovascular Surgery: Sung Nok Hong (1927–2017) Who Performed the First Coronary Artery Bypass Graft in Korea
- Historical Perspectives of the Treatment of Thyroid Disease