Intest Res.
2021 Oct;19(4):430-437. 10.5217/ir.2020.00067.
Clinical characteristics of inflammatory bowel disease patients with immunoglobulin A nephropathy
- Affiliations
-
- 1Department of Endoscopy, Hiroshima University Hospital, Hiroshima, Japan
- 2Department of Gastroenterology and Metabolism, Hiroshima University Hospital, Hiroshima, Japan
- 3Department of Nephrology, Hiroshima University Hospital, Hiroshima, Japan
- 4Institute of Physical and Chemical Research (RIKEN) Center for Integrative Medical Sciences, Yokohama, Japan
- KMID: 2521606
- DOI: http://doi.org/10.5217/ir.2020.00067
Abstract
- Background/Aims
Inflammatory bowel disease (IBD) is a chronic inflammation of the gastrointestinal tract. Some patients with this condition have been reported to present with immunoglobulin A nephropathy (IgAN), a renal complication that can cause end-stage renal failure, but the frequency of this comorbidity has not been described. Thus, the aim of this study was to investigate the frequency of IgAN in patients with IBD.
Methods
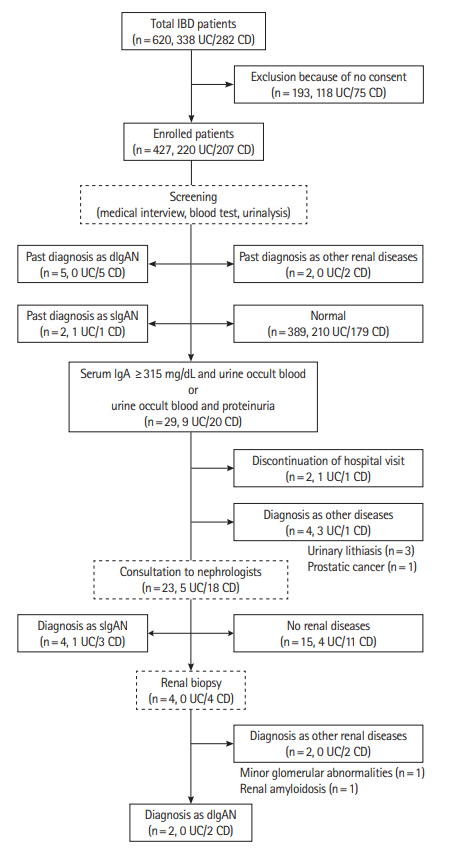
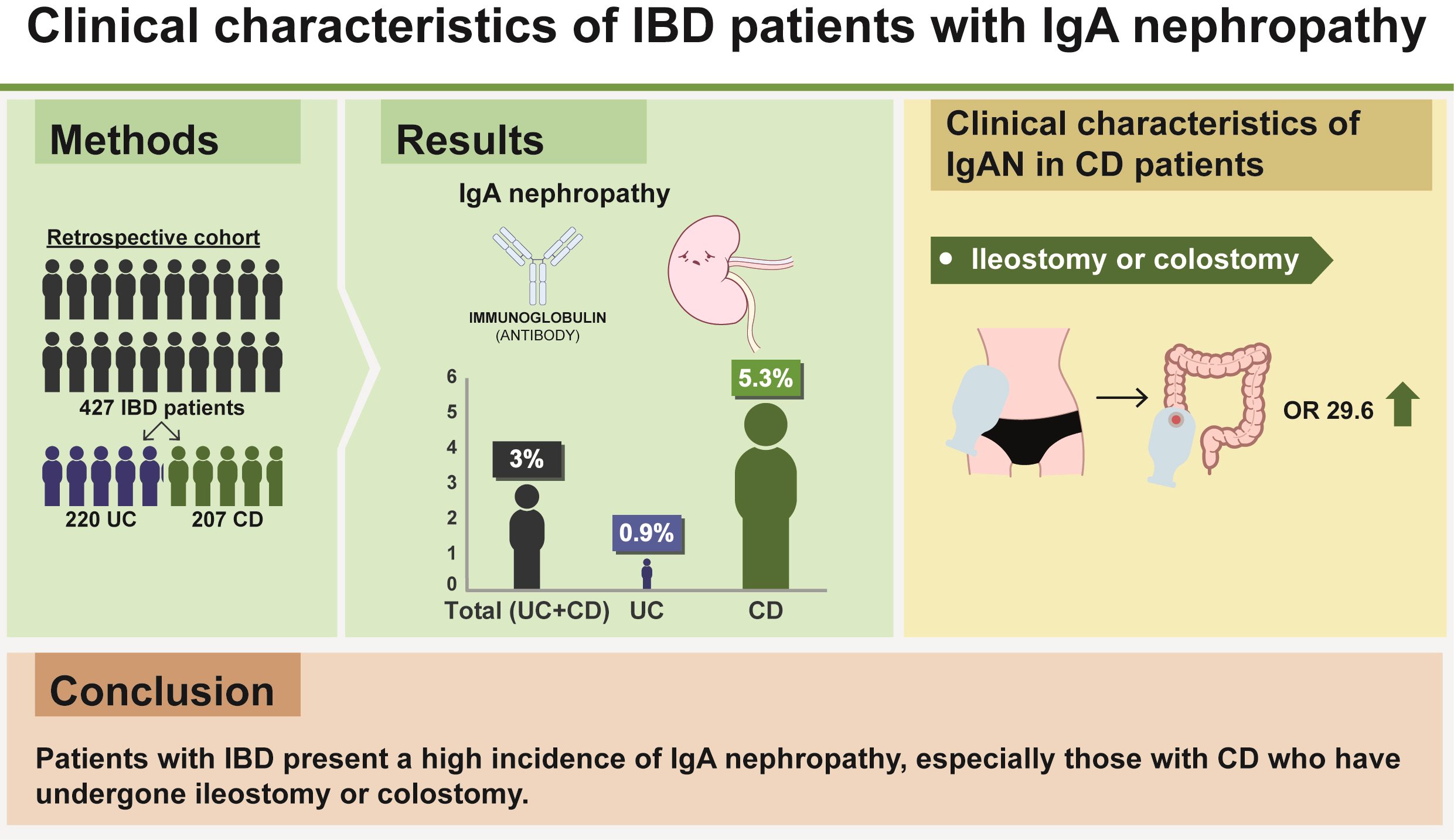
This study included 620 patients with IBD (338 with ulcerative colitis [UC] and 282 with Crohn’s disease [CD]) from the Hiroshima University Hospital outpatient department. IgAN cases were identified from medical interviews, blood examinations (serum immunoglobulin A), and urinalyses (occult blood, proteinuria). Definitive IgAN cases were diagnosed by renal biopsies, while those detected through the clinical course and test results, but not clinically recommended for renal biopsy, were defined as suspected IgAN.
Results
We analyzed 427 cases meeting the inclusion criteria (220 with UC and 207 with CD). The incidence of IgAN across all patients with IBD was 3.0%. The frequency of IgAN was significantly higher in patients with CD (11/207, 5.3%) than in those with UC (2/220, 0.9%) (P< 0.01). Moreover, a significant correlation was found between CD patients with ileostomy or colostomy and a diagnosis of IgAN.
Conclusions
Patients with IBD present a high incidence of IgAN, especially those with CD who have undergone ileostomy or colostomy.
Keyword
Figure
Reference
-
1. Kaser A, Zeissig S, Blumberg RS. Inflammatory bowel disease. Annu Rev Immunol. 2010; 28:573–621.2. Danese S, Fiocchi C. Ulcerative colitis. N Engl J Med. 2011; 365:1713–1725.
Article3. Maloy KJ, Powrie F. Intestinal homeostasis and its breakdown in inflammatory bowel disease. Nature. 2011; 474:298–306.4. Gregory B, Ho VC. Cutaneous manifestations of gastrointestinal disorders. Part II. J Am Acad Dermatol. 1992; 26:371–383.5. Gravallese EM, Kantrowitz FG. Arthritic manifestations of inflammatory bowel disease. Am J Gastroenterol. 1988; 83:703–709.6. Orchard TR, Thiyagaraja S, Welsh KI, Wordsworth BP, Hill Gaston JS, Jewell DP. Clinical phenotype is related to HLA genotype in the peripheral arthropathies of inflammatory bowel disease. Gastroenterology. 2000; 118:274–278.
Article7. Ardizzone S, Puttini PS, Cassinotti A, Porro GB. Extraintestinal manifestations of inflammatory bowel disease. Dig Liver Dis. 2008; 40–Suppl 2:S253-S259.8. Loftus EV Jr, Harewood GC, Loftus CG, et al. PSC-IBD: a unique form of inflammatory bowel disease associated with primary sclerosing cholangitis. Gut. 2005; 54:91–96.9. Pardi DS, Tremaine WJ, Sandborn WJ, McCarthy JT. Renal and urologic complications of inflammatory bowel disease. Am J Gastroenterol. 1998; 93:504–514.
Article10. Donadio JV, Grande JP. IgA nephropathy. N Engl J Med. 2002; 347:738–748.
Article11. Takemura T, Okada M, Yagi K, Kuwajima H, Yanagida H. An adolescent with IgA nephropathy and Crohn disease: pathogenetic implications. Pediatr Nephrol. 2002; 17:863–866.
Article12. Ueno Y, Tanaka S, Onitake T, et al. Infliximab treatment for Crohn’s disease in a patient with IgA nephropathy. Clin J Gastroenterol. 2009; 2:380–383.13. Forshaw MJ, Guirguis O, Hennigan TW. IgA nephropathy in association with Crohn’s disease. Int J Colorectal Dis. 2005; 20:463–465.
Article14. Marek-Bukowiec K, Konieczny A, Ratajczyk K, Witkiewicz W. Candidate urine peptide biomarkers for IgA nephropathy: where are we now? Dis Markers. 2018; 2018:5205831.
Article15. O’Shaughnessy MM, Hogan SL, Thompson BD, Coppo R, Fogo AB, Jennette JC. Glomerular disease frequencies by race, sex and region: results from the International Kidney Biopsy Survey. Nephrol Dial Transplant. 2018; 33:661–669.
Article16. Satsangi J, Silverberg MS, Vermeire S, Colombel JF. The Montreal classification of inflammatory bowel disease: controversies, consensus, and implications. Gut. 2006; 55:749–753.
Article17. Rachmilewitz D. Coated mesalazine (5-aminosalicylic acid) versus sulphasalazine in the treatment of active ulcerative colitis: a randomized trial. BMJ. 1989; 298:82–86.
Article18. Best WR, Becktel JM, Singleton JW, Kern F Jr. Development of a Crohn’s disease activity index. national cooperative Crohn’s disease study. Gastroenterology. 1976; 70:439–444.19. Utsunomiya Y, Koda T, Kado T, et al. Incidence of pediatric IgA nephropathy. Pediatr Nephrol. 2003; 18:511–515.
Article20. McGrogan A, Franssen CF, de Vries CS. The incidence of primary glomerulonephritis worldwide: a systematic review of the literature. Nephrol Dial Transplant. 2011; 26:414–430.21. Ambruzs JM, Walker PD, Larsen CP. The histopathologic spectrum of kidney biopsies in patients with inflammatory bowel disease. Clin J Am Soc Nephrol. 2014; 9:265–270.22. Wyatt RJ, Julian BA. IgA nephropathy. N Engl J Med. 2013; 368:2402–2414.
Article23. Coppo R. The intestine-renal connection in IgA nephropathy. Nephrol Dial Transplant. 2015; 30:360–366.
Article24. Cesta MF. Normal structure, function, and histology of mucosa-associated lymphoid tissue. Toxicol Pathol. 2006; 34:599–608.
Article25. Kiryluk K, Novak J. The genetics and immunobiology of IgA nephropathy. J Clin Invest. 2014; 124:2325–2332.
Article26. Nagy J, Scott H, Brandtzaeg P. Antibodies to dietary antigens in IgA nephropathy. Clin Nephrol. 1988; 29:275–279.27. Kamata T, Nogaki F, Fagarasan S, et al. Increased frequency of surface IgA-positive plasma cells in the intestinal lamina propria and decreased IgA excretion in hyper IgA (HIGA) mice, a murine model of IgA nephropathy with hyperserum IgA. J Immunol. 2000; 165:1387–1394.
Article28. Kiryluk K, Novak J, Gharavi AG. Pathogenesis of immunoglobulin A nephropathy: recent insight from genetic studies. Annu Rev Med. 2013; 64:339–356.
Article29. Wehkamp J, Salzman NH, Porter E, et al. Reduced Paneth cell alpha-defensins in ileal Crohn’s disease. Version 2. Proc Natl Acad Sci U S A. 2005; 102:18129–18134.30. Hayashi R, Tsuchiya K, Fukushima K, et al. Reduced human α-defensin 6 in noninflamed jejunal tissue of patients with Crohn’s disease. Inflamm Bowel Dis. 2016; 22:1119–1128.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Inflammatory Bowel Disease in Pediatric Age
- Lymphocyte Subpopulations in Patients with Minimal Change Disease and IgA Nephropathy
- Inflammatory Bowel Disease in Korea: Epidemiological, Genomic, Clinical, and Therapeutic Characteristics
- A Case of Familial IgA Nephropathy
- Biological Therapy for Inflammatory Bowel Disease in Children