Korean Circ J.
2021 Jul;51(7):579-597. 10.4070/kcj.2021.0089.
Cardiac Toxicities Associated with Immune Checkpoints Inhibitors: Mechanisms, Manifestations and Management
- Affiliations
-
- 1Department of Medicine, Brookdale Hospital University and Medical Center, Brooklyn, NY, USA
- 2Department of Medicine, University of Texas Health Science Center at Houston, Houston, TX, USA
- 3Department of Cardiology, The University of Texas MD Anderson Cancer Center, Houston, TX, USA
- KMID: 2517487
- DOI: http://doi.org/10.4070/kcj.2021.0089
Abstract
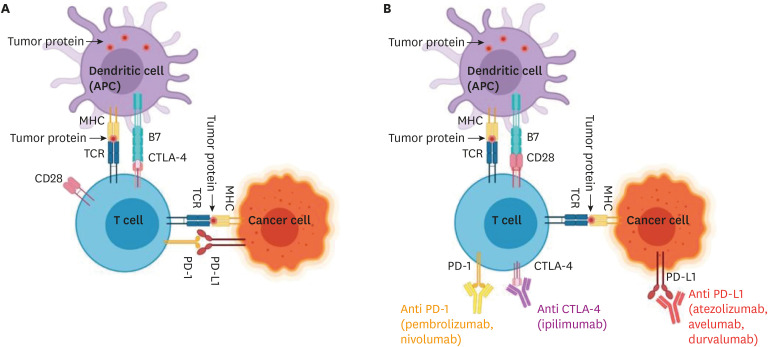
- Immune checkpoint inhibitor (ICI) associated cardiovascular adverse events (CVAE) have become more frequent with the growing use of cancer immunotherapy. CVAEs include a wide spectrum of diseases such as myocarditis, pericarditis, heart failure, arrhythmias, coronary artery disease, and hypertension. The induction of cardiovascular side effects by ICI use is hypothesized to occur due to inflammation and immune dysregulation of normal tissue in response to immunotherapy. Management of ICI-associated CVAEs mitigates an overactive immune response by utilizing steroids, immunomodulatory drugs and hemodynamic stabilization. However, few controlled studies on the cardiovascular safety of ICIs exist and treatment of their side effects are mostly from limited case series. Our review seeks to provide the most recent understanding of ICI-associated CVAEs and their management.
Keyword
Figure
Reference
-
1. Chang CF. Disease and its impact on politics, diplomacy, and the military: the case of smallpox and the Manchus (1613–1795). J Hist Med Allied Sci. 2002; 57:177–197. PMID: 11995595.
Article2. McCarthy EF. The toxins of William B. Coley and the treatment of bone and soft-tissue sarcomas. Iowa Orthop J. 2006; 26:154–158. PMID: 16789469.3. January CT, Wann LS, Alpert JS, et al. 2014 AHA/ACC/HRS guideline for the management of patients with atrial fibrillation: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on practice guidelines and the Heart Rhythm Society. Circulation. 2014; 130:2071–2104. PMID: 24682348.4. Tarrio ML, Grabie N, Bu DX, Sharpe AH, Lichtman AH. PD-1 protects against inflammation and myocyte damage in T cell-mediated myocarditis. J Immunol. 2012; 188:4876–4884. PMID: 22491251.
Article5. Tivol EA, Borriello F, Schweitzer AN, Lynch WP, Bluestone JA, Sharpe AH. Loss of CTLA-4 leads to massive lymphoproliferation and fatal multiorgan tissue destruction, revealing a critical negative regulatory role of CTLA-4. Immunity. 1995; 3:541–547. PMID: 7584144.
Article6. Nishimura H, Okazaki T, Tanaka Y, et al. Autoimmune dilated cardiomyopathy in PD-1 receptor-deficient mice. Science. 2001; 291:319–322. PMID: 11209085.
Article7. Ji C, Roy MD, Golas J, et al. Myocarditis in cynomolgus monkeys following treatment with immune checkpoint inhibitors. Clin Cancer Res. 2019; 25:4735–4748. PMID: 31085720.
Article8. Heinzerling L, Ott PA, Hodi FS, et al. Cardiotoxicity associated with CTLA4 and PD1 blocking immunotherapy. J Immunother Cancer. 2016; 4:50. PMID: 27532025.
Article9. Johnson DB, Balko JM, Compton ML, et al. Fulminant myocarditis with combination immune checkpoint blockade. N Engl J Med. 2016; 375:1749–1755. PMID: 27806233.10. Bu DX, Tarrio M, Maganto-Garcia E, et al. Impairment of the programmed cell death-1 pathway increases atherosclerotic lesion development and inflammation. Arterioscler Thromb Vasc Biol. 2011; 31:1100–1107. PMID: 21393583.
Article11. Foks AC, Kuiper J. Immune checkpoint proteins: exploring their therapeutic potential to regulate atherosclerosis. Br J Pharmacol. 2017; 174:3940–3955. PMID: 28369782.
Article12. Lee J, Zhuang Y, Wei X, et al. Contributions of PD-1/PD-L1 pathway to interactions of myeloid DCs with T cells in atherosclerosis. J Mol Cell Cardiol. 2009; 46:169–176. PMID: 19056397.
Article13. Tomiyama H, Shiina K, Matsumoto-Nakano C, et al. The contribution of inflammation to the development of hypertension mediated by increased arterial stiffness. J Am Heart Assoc. 2017; 6:e005729. PMID: 28666991.
Article14. Tadokoro T, Keshino E, Makiyama A, et al. Acute lymphocytic myocarditis with anti-PD-1 antibody nivolumab. Circ Heart Fail. 2016; 9:e003514. PMID: 27650418.
Article15. Läubli H, Balmelli C, Bossard M, Pfister O, Glatz K, Zippelius A. Acute heart failure due to autoimmune myocarditis under pembrolizumab treatment for metastatic melanoma. J Immunother Cancer. 2015; 3:11. PMID: 25901283.
Article16. Tajmir-Riahi A, Bergmann T, Schmid M, Agaimy A, Schuler G, Heinzerling L. Life-threatening autoimmune cardiomyopathy reproducibly induced in a patient by checkpoint inhibitor therapy. J Immunother. 2018; 41:35–38. PMID: 29077601.
Article17. Brahmer JR, Lacchetti C, Schneider BJ, et al. Management of immune-related adverse events in patients treated with immune checkpoint inhibitor therapy: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2018; 36:1714–1768. PMID: 29442540.18. Jain V, Bahia J, Mohebtash M, Barac A. Cardiovascular complications associated with novel cancer immunotherapies. Curr Treat Options Cardiovasc Med. 2017; 19:36. PMID: 28401456.
Article19. Mahmood SS, Fradley MG, Cohen JV, et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018; 71:1755–1764. PMID: 29567210.
Article20. Zimmer L, Goldinger SM, Hofmann L, et al. Neurological, respiratory, musculoskeletal, cardiac and ocular side-effects of anti-PD-1 therapy. Eur J Cancer. 2016; 60:210–225. PMID: 27084345.
Article21. Mahmood SS, Fradley MG, Cohen JV, et al. Myocarditis in patients treated with immune checkpoint inhibitors. J Am Coll Cardiol. 2018; 71:1755–1764. PMID: 29567210.
Article22. Salem JE, Manouchehri A, Moey M, et al. Cardiovascular toxicities associated with immune checkpoint inhibitors: an observational, retrospective, pharmacovigilance study. Lancet Oncol. 2018; 19:1579–1589. PMID: 30442497.
Article23. Moslehi JJ, Salem JE, Sosman JA, Lebrun-Vignes B, Johnson DB. Increased reporting of fatal immune checkpoint inhibitor-associated myocarditis. Lancet. 2018; 391:933.
Article24. Amiri-Kordestani L, Moslehi J, Cheng J, et al. Cardiovascular adverse events in immune checkpoint inhibitor clinical trials: a U.S. Food and Drug Administration pooled analysis. J Clin Oncol. 2018; 36(15 Suppl):3009.
Article25. Palaskas N, Morgan J, Daigle T, et al. Targeted cancer therapies with pericardial effusions requiring pericardiocentesis focusing on immune checkpoint inhibitors. Am J Cardiol. 2019; 123:1351–1357. PMID: 30765065.
Article26. Kolla BC, Patel MR. Recurrent pleural effusions and cardiac tamponade as possible manifestations of pseudoprogression associated with nivolumab therapy - a report of two cases. J Immunother Cancer. 2016; 4:80. PMID: 27895919.
Article27. Khan A, Zhang H, Chen C, et al. Impact of pericardial effusion for patients receiving immune checkpoint inhibitors. J Clin Oncol. 2019; 37(15 Suppl):e14121.
Article28. Caforio AL, Pankuweit S, Arbustini E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013; 34:2636–2648. PMID: 23824828.
Article29. Escudier M, Cautela J, Malissen N, et al. Clinical features, management, and outcomes of immune checkpoint inhibitor-related cardiotoxicity. Circulation. 2017; 136:2085–2087. PMID: 29158217.
Article30. Mirabel M, Karapetiantz P, Marijon E, et al. The risk of sudden cardiac death or ventricular arrhythmias on immune checkpoint inhibitors. Eur Heart J. 2020; 41(Suppl 2):ehaa946.3488.
Article31. Cautela J, Rouby F, Salem JE, et al. Acute coronary syndrome with immune checkpoint inhibitors: a proof-of-concept case and pharmacovigilance analysis of a life-threatening adverse event. Can J Cardiol. 2020; 36:476–481. PMID: 32144037.
Article32. Otsu K, Tajiri K, Sakai S, Ieda M. Vasospastic angina following immune checkpoint blockade. Eur Heart J. 2020; 41:1702. PMID: 31697341.
Article33. Nykl R, Fischer O, Vykoupil K, Taborsky M. A unique reason for coronary spasm causing temporary ST elevation myocardial infarction (inferior STEMI) - systemic inflammatory response syndrome after use of pembrolizumab. Arch Med Sci Atheroscler Dis. 2017; 2:e100–2. PMID: 29379889.
Article34. Lyon AR, Yousaf N, Battisti NM, Moslehi J, Larkin J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol. 2018; 19:e447–58. PMID: 30191849.
Article35. Hui R, Garon EB, Goldman JW, et al. Pembrolizumab as first-line therapy for patients with PD-L1-positive advanced non-small cell lung cancer: a phase 1 trial. Ann Oncol. 2017; 28:874–881. PMID: 28168303.
Article36. Garon EB, Hellmann MD, Rizvi NA, et al. Five-year overall survival for patients with advanced non‒small-cell lung cancer treated with pembrolizumab: results from the phase I KEYNOTE-001 study. J Clin Oncol. 2019; 37:2518–2527. PMID: 31154919.
Article37. Mok TS, Wu YL, Kudaba I, et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019; 393:1819–1830. PMID: 30955977.38. Herbst RS, Baas P, Kim DW, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet. 2016; 387:1540–1550. PMID: 26712084.
Article39. Reck M, Rodríguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. 2016; 375:1823–1833. PMID: 27718847.
Article40. Chen J, Hu X, Li Q, et al. Effectiveness and safety of toripalimab, camrelizumab, and sintilimab in a real-world cohort of hepatitis B virus associated hepatocellular carcinoma patients. Ann Transl Med. 2020; 8:1187. PMID: 33241036.
Article41. de Azevedo SJ, de Melo AC, Roberts L, Caro I, Xue C, Wainstein A. First-line atezolizumab monotherapy in patients with advanced BRAFV600 wild-type melanoma. Pigment Cell Melanoma Res. 2021.42. Lee MS, Ryoo BY, Hsu CH, et al. Atezolizumab with or without bevacizumab in unresectable hepatocellular carcinoma (GO30140): an open-label, multicentre, phase 1b study. Lancet Oncol. 2020; 21:808–820. PMID: 32502443.
Article43. Massin M, Crochelet AS, Jacquemart C. Acute myocarditis with very high troponin but no ventricular dysfunction. Clin Pediatr (Phila). 2017; 56:582–583. PMID: 27356633.
Article44. Bando S, Soeki T, Matsuura T, et al. Plasma brain natriuretic peptide levels are elevated in patients with cancer. PLoS One. 2017; 12:e0178607. PMID: 28570595.
Article45. Hughes M, Lilleker JB, Herrick AL, Chinoy H. Cardiac troponin testing in idiopathic inflammatory myopathies and systemic sclerosis-spectrum disorders: biomarkers to distinguish between primary cardiac involvement and low-grade skeletal muscle disease activity. Ann Rheum Dis. 2015; 74:795–798. PMID: 25732174.
Article46. Alvarez-Cardona Jose A, Zhang Kathleen W, Mitchell Joshua D, Zaha Vlad G, Fisch Michael J, Lenihan Daniel J. Cardiac biomarkers during cancer therapy. JACC CardioOncol. 2020; 2:791–794.47. Kindermann I, Kindermann M, Kandolf R, et al. Predictors of outcome in patients with suspected myocarditis. Circulation. 2008; 118:639–648. PMID: 18645053.
Article48. Awadalla M, Mahmood SS, Groarke JD, et al. Global longitudinal strain and cardiac events in patients with immune checkpoint inhibitor-related myocarditis. J Am Coll Cardiol. 2020; 75:467–478. PMID: 32029128.49. Ferreira VM, Schulz-Menger J, Holmvang G, et al. Cardiovascular magnetic resonance in nonischemic myocardial inflammation: expert recommendations. J Am Coll Cardiol. 2018; 72:3158–3176. PMID: 30545455.50. Zhang L, Awadalla M, Mahmood SS, et al. Cardiovascular magnetic resonance in immune checkpoint inhibitor-associated myocarditis. Eur Heart J. 2020; 41:1733–1743. PMID: 32112560.51. Chen W, Jeudy J. Assessment of myocarditis: cardiac MR, PET/CT, or PET/MR? Curr Cardiol Rep. 2019; 21:76. PMID: 31243587.
Article52. Hauck AJ, Kearney DL, Edwards WD. Evaluation of postmortem endomyocardial biopsy specimens from 38 patients with lymphocytic myocarditis: implications for role of sampling error. Mayo Clin Proc. 1989; 64:1235–1245. PMID: 2593714.
Article53. Wu LA, Lapeyre AC 3rd, Cooper LT. Current role of endomyocardial biopsy in the management of dilated cardiomyopathy and myocarditis. Mayo Clin Proc. 2001; 76:1030–1038. PMID: 11605687.
Article54. Baughman KL. Diagnosis of myocarditis: death of Dallas criteria. Circulation. 2006; 113:593–595. PMID: 16449736.55. Agrawal N, Khunger A, Vachhani P, et al. Cardiac toxicity associated with immune checkpoint inhibitors: case series and review of the literature. Case Rep Oncol. 2019; 12:260–276. PMID: 31011325.
Article56. Palaskas N, Lopez-Mattei J, Durand JB, Iliescu C, Deswal A. Immune checkpoint inhibitor myocarditis: pathophysiological characteristics, diagnosis, and treatment. J Am Heart Assoc. 2020; 9:e013757. PMID: 31960755.
Article57. Saade A, Mansuet-Lupo A, Arrondeau J, et al. Pericardial effusion under nivolumab: case-reports and review of the literature. J Immunother Cancer. 2019; 7:266. PMID: 31627742.
Article58. Frigeri M, Meyer P, Banfi C, et al. Immune checkpoint inhibitor-associated myocarditis: a new challenge for cardiologists. Can J Cardiol. 2018; 34:92.e1–92.e3.
Article59. Tay RY, Blackley E, McLean C, et al. Successful use of equine anti-thymocyte globulin (ATGAM) for fulminant myocarditis secondary to nivolumab therapy. Br J Cancer. 2017; 117:921–924. PMID: 28797029.
Article60. Jain V, Mohebtash M, Rodrigo ME, Ruiz G, Atkins MB, Barac A. Autoimmune myocarditis caused by immune checkpoint inhibitors treated with antithymocyte globulin. J Immunother. 2018; 41:332–335. PMID: 29965858.
Article61. Wang DY, Okoye GD, Neilan TG, Johnson DB, Moslehi JJ. Cardiovascular toxicities associated with cancer immunotherapies. Curr Cardiol Rep. 2017; 19:21. PMID: 28220466.
Article62. Caforio AL, Pankuweit S, Arbustini E, et al. Current state of knowledge on aetiology, diagnosis, management, and therapy of myocarditis: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2013; 34:2636–2648. PMID: 23824828.
Article63. Balanescu DV, Donisan T, Palaskas N, et al. Immunomodulatory treatment of immune checkpoint inhibitor-induced myocarditis: pathway toward precision-based therapy. Cardiovasc Pathol. 2020; 47:107211. PMID: 32268262.
Article64. Yamaguchi S, Morimoto R, Okumura T, et al. Late-onset fulminant myocarditis with immune checkpoint inhibitor nivolumab. Can J Cardiol. 2018; 34:812.e1–812.e3.
Article65. Xing Q, Zhang ZW, Lin QH, et al. Myositis-myasthenia gravis overlap syndrome complicated with myasthenia crisis and myocarditis associated with anti-programmed cell death-1 (sintilimab) therapy for lung adenocarcinoma. Ann Transl Med. 2020; 8:250. PMID: 32309397.
Article66. Yanase T, Moritoki Y, Kondo H, Ueyama D, Akita H, Yasui T. Myocarditis and myasthenia gravis by combined nivolumab and ipilimumab immunotherapy for renal cell carcinoma: a case report of successful management. Urol Case Rep. 2020; 34:101508. PMID: 33318935.
Article67. Fazel M, Jedlowski PM. Severe myositis, myocarditis, and myasthenia gravis with elevated anti-striated muscle antibody following single dose of ipilimumab-nivolumab therapy in a patient with metastatic melanoma. Case Reports Immunol. 2019; 2019:2539493. PMID: 31183226.
Article68. Yogasundaram H, Alhumaid W, Chen JW, et al. Plasma exchange for immune checkpoint inhibitor-induced myocarditis. CJC Open. 2020; 3:379–382. PMID: 33778457.
Article69. Compton F, He L, Sarode R, et al. Immune checkpoint inhibitor toxicity: a new indication for therapeutic plasma exchange? J Clin Apher. 2021; jca.21890.
Article70. Schiopu SR, Käsmann L, Schönermarck U, et al. Pembrolizumab-induced myocarditis in a patient with malignant mesothelioma: plasma exchange as a successful emerging therapy-case report. Transl Lung Cancer Res. 2021; 10:1039–1046. PMID: 33718042.
Article71. Kimura T, Fukushima S, Miyashita A, et al. Myasthenic crisis and polymyositis induced by one dose of nivolumab. Cancer Sci. 2016; 107:1055–1058. PMID: 27420474.
Article72. Rota E, Varese P, Agosti S, et al. Concomitant myasthenia gravis, myositis, myocarditis and polyneuropathy, induced by immune-checkpoint inhibitors: a life-threatening continuum of neuromuscular and cardiac toxicity. eNeurologicalSci. 2018; 14:4–5. PMID: 30533536.
Article73. Reddy N, Moudgil R, Lopez-Mattei JC, et al. Progressive and reversible conduction disease with checkpoint inhibitors. Can J Cardiol. 2017; 33:1335.e13–1335.e15.
Article74. Hu JR, Florido R, Lipson EJ, et al. Cardiovascular toxicities associated with immune checkpoint inhibitors. Cardiovasc Res. 2019; 115:854–868. PMID: 30715219.
Article75. Kwon HJ, Coté TR, Cuffe MS, Kramer JM, Braun MM. Case reports of heart failure after therapy with a tumor necrosis factor antagonist. Ann Intern Med. 2003; 138:807–811. PMID: 12755552.
Article76. Chung ES, Packer M, Lo KH, Fasanmade AA, Willerson JT. Anti-TNF Therapy Against Congestive Heart Failure Investigators. Randomized, double-blind, placebo-controlled, pilot trial of infliximab, a chimeric monoclonal antibody to tumor necrosis factor-alpha, in patients with moderate-to-severe heart failure: results of the anti-TNF Therapy Against Congestive Heart Failure (ATTACH) trial. Circulation. 2003; 107:3133–3140. PMID: 12796126.77. Jeyakumar N, Etchegaray M, Henry J, et al. The terrible triad of checkpoint inhibition: a case report of myasthenia gravis, myocarditis, and myositis induced by cemiplimab in a patient with metastatic cutaneous squamous cell carcinoma. Case Reports Immunol. 2020; 2020:5126717. PMID: 32695533.
Article78. Esfahani K, Buhlaiga N, Thébault P, Lapointe R, Johnson NA, Miller WH Jr. Alemtuzumab for immune-related myocarditis due to PD-1 therapy. N Engl J Med. 2019; 380:2375–2376. PMID: 31189042.
Article79. Deftereos SN, Georgonikou D. Effectiveness of rituximab in treating immune-checkpoint-inhibitor-induced immune-related adverse events: results of a systematic review. Ann Oncol. 2021; 32:282–283. PMID: 33309745.
Article80. Yancy CW, Jessup M, Bozkurt B, et al. 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017; 136:e137–61. PMID: 28455343.
Article81. Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC Guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2014; 64:e139–228. PMID: 25260718.82. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation. 2004; 110:588–636. PMID: 15289388.83. Al-Khatib SM, Stevenson WG, Ackerman MJ, et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm. 2018; 15:e190–252. PMID: 29097320.84. Behling J, Kaes J, Münzel T, Grabbe S, Loquai C. New-onset third-degree atrioventricular block because of autoimmune-induced myositis under treatment with anti-programmed cell death-1 (nivolumab) for metastatic melanoma. Melanoma Res. 2017; 27:155–158. PMID: 27977496.
Article85. Ingelfinger JR, Schwartz RS. Immunosuppression--the promise of specificity. N Engl J Med. 2005; 353:836–839. PMID: 16120865.86. Wei SC, Meijers WC, Axelrod ML, et al. A genetic mouse model recapitulates immune checkpoint inhibitor-associated myocarditis and supports a mechanism-based therapeutic intervention. Cancer Discov. 2021; 11:614–625. PMID: 33257470.
Article87. Liu S, Chan J, Brinc D, et al. Immune checkpoint inhibitor-associated myocarditis with persistent troponin elevation despite abatacept and prolonged immunosuppression. JACC CardioOncol. 2020; 2:800–804.
Article88. Salem JE, Allenbach Y, Vozy A, et al. Abatacept for severe immune checkpoint inhibitor-associated myocarditis. N Engl J Med. 2019; 380:2377–2379. PMID: 31189043.
Article89. Blair HA, Deeks ED. Abatacept: a review in rheumatoid arthritis. Drugs. 2017; 77:1221–1233. PMID: 28608166.
Article90. Simon TA, Boers M, Hochberg M, et al. Comparative risk of malignancies and infections in patients with rheumatoid arthritis initiating abatacept versus other biologics: a multi-database real-world study. Arthritis Res Ther. 2019; 21:228. PMID: 31703717.
Article91. Chen DY, Huang WK, Chien-Chia Wu V, et al. Cardiovascular toxicity of immune checkpoint inhibitors in cancer patients: a review when cardiology meets immuno-oncology. J Formos Med Assoc. 2020; 119:1461–1475. PMID: 31444018.
Article92. Zhou YW, Zhu YJ, Wang MN, et al. Immune checkpoint inhibitor-associated cardiotoxicity: current understanding on its mechanism, diagnosis and management. Front Pharmacol. 2019; 10:1350. PMID: 31849640.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immune-related Adverse Events: Overview and Management Strategies for the Use of Immune Checkpoint Inhibitors
- Management of the adverse effects of cancer immunotherapy with a focus on the gastrointestinal and hepatic systems
- Characteristics of Cardiac Toxicity in Antidepressant Overdose and Its Treatment
- Immune checkpoint blockade therapy for bladder cancer treatment
- Management of adverse events in cancer treatment with immune checkpoint inhibitors