Nutr Res Pract.
2021 Jun;15(3):279-293. 10.4162/nrp.2021.15.3.279.
The protective effects of steamed ginger on adipogenesis in 3T3-L1 cells and adiposity in diet-induced obese mice
- Affiliations
-
- 1Department of Food Science and Nutrition, Pusan National University, Busan 46264, Korea
- 2Department of Food Science and Human Nutrition and Obesity Research Center, Jeonbuk National University, Jeonju 54896, Korea
- KMID: 2516068
- DOI: http://doi.org/10.4162/nrp.2021.15.3.279
Abstract
- BACKGROUND/OBJECTIVES
The steamed ginger has been shown to have antioxidative effects and a protective effect against obesity. In the present study, we investigated the effects of ethanolic extract of steamed ginger (SGE) on adipogenesis in 3T3-L1 preadipocytes and diet-induced obesity (DIO) mouse model.
MATERIALS/METHODS
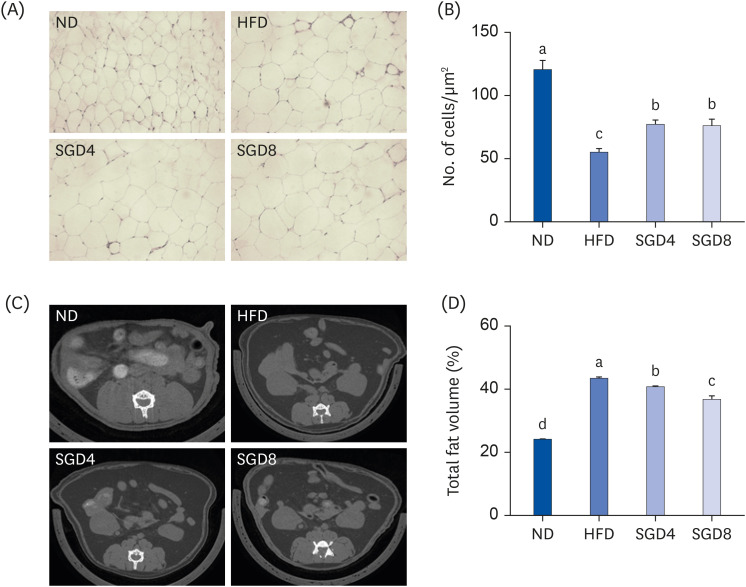
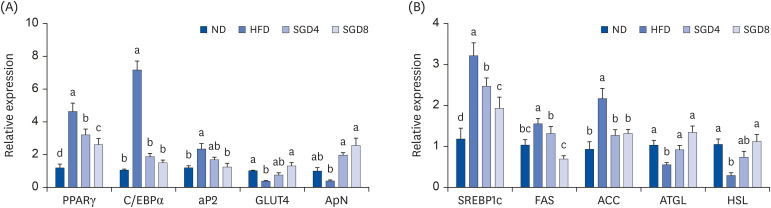
The protective effects of SGE on adipogenesis were examined in 3T3-L1 adipocytes by measuring lipid accumulations and genes involved in adipogenesis. Male C57BL/6J mice were fed a normal diet (ND, 10% fat w/w), a high-fat diet (HFD, 60% fat w/w), and HFD supplemented with either 40 mg/kg or 80 mg/kg of SGE for 12 weeks. Serum chemistry was measured, and the expression of genes involved in lipid metabolism was determined in the adipose tissue. Histological analysis and micro-computed tomography were performed to identify lipid accumulations in epididymal fat pads.
RESULTS
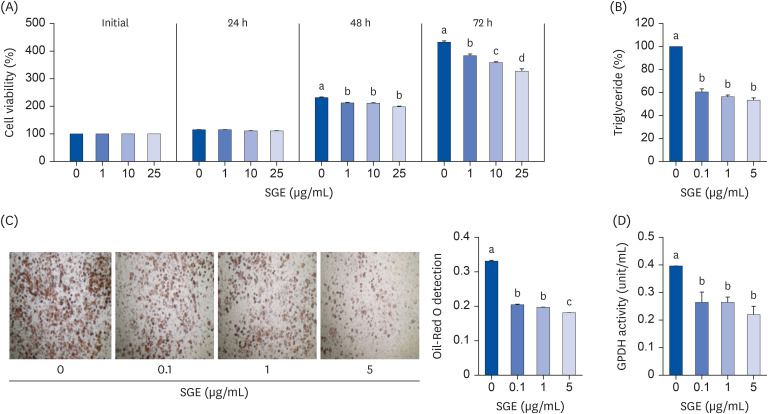
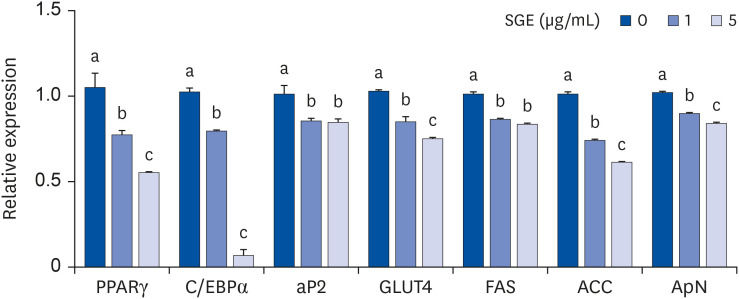
In 3T3-L1 cells, SGE significantly decreased lipid accumulation, with concomitant decreases in the expression of adipogenesis-related genes. SGE significantly attenuated the increase in body, liver, and epididymal adipose tissue weights by HFD. Serum total cholesterol and triglyceride levels were significantly lower in SGE fed groups compared to HFD. In adipose tissue, SGE significantly decreased adipocyte size than that of HFD and altered adipogenesis-related genes.
CONCLUSIONS
In conclusion, steamed ginger exerted anti-obesity effects by regulating genes involved in adipogenesis and lipogenesis in 3T3-L1 cell and epididymal adipose tissue of DIO mice.
Keyword
Figure
Reference
-
1. Botchlett R, Woo SL, Liu M, Pei Y, Guo X, Li H, Wu C. Nutritional approaches for managing obesity-associated metabolic diseases. J Endocrinol. 2017; 233:R145–71. PMID: 28400405.
Article2. Jo J, Gavrilova O, Pack S, Jou W, Mullen S, Sumner AE, Cushman SW, Periwal V. Hypertrophy and/or hyperplasia: dynamics of adipose tissue growth. PLOS Comput Biol. 2009; 5:e1000324. PMID: 19325873.
Article3. Choe SS, Huh JY, Hwang IJ, Kim JI, Kim JB. Adipose tissue remodeling: its role in energy metabolism and metabolic disorders. Front Endocrinol (Lausanne). 2016; 7:30. PMID: 27148161.
Article4. Nishimura S, Manabe I, Nagasaki M, Hosoya Y, Yamashita H, Fujita H, Ohsugi M, Tobe K, Kadowaki T, Nagai R, Sugiura S. Adipogenesis in obesity requires close interplay between differentiating adipocytes, stromal cells, and blood vessels. Diabetes. 2007; 56:1517–1526. PMID: 17389330.
Article5. Haczeyni F, Bell-Anderson KS, Farrell GC. Causes and mechanisms of adipocyte enlargement and adipose expansion. Obes Rev. 2018; 19:406–420. PMID: 29243339.
Article6. Tandon P, Wafer R, Minchin JE. Adipose morphology and metabolic disease. J Exp Biol. 2018; 221(Suppl 1):221.
Article7. Roberts R, Hodson L, Dennis AL, Neville MJ, Humphreys SM, Harnden KE, Micklem KJ, Frayn KN. Markers of de novo lipogenesis in adipose tissue: associations with small adipocytes and insulin sensitivity in humans. Diabetologia. 2009; 52:882–890. PMID: 19252892.8. Ali AT, Hochfeld WE, Myburgh R, Pepper MS. Adipocyte and adipogenesis. Eur J Cell Biol. 2013; 92:229–236. PMID: 23876739.
Article9. Mota de Sá P, Richard AJ, Hang H, Stephens JM. Transcriptional regulation of adipogenesis. Compr Physiol. 2017; 7:635–674. PMID: 28333384.10. Siersbaek R, Nielsen R, Mandrup S. PPARgamma in adipocyte differentiation and metabolism--novel insights from genome-wide studies. FEBS Lett. 2010; 584:3242–3249. PMID: 20542036.11. Musri MM, Párrizas M. Epigenetic regulation of adipogenesis. Curr Opin Clin Nutr Metab Care. 2012; 15:342–349. PMID: 22617562.
Article12. Liu Y, Sun M, Yao H, Liu Y, Gao R. Herbal medicine for the treatment of obesity: an overview of scientific evidence from 2007 to 2017. Evid Based Complement Alternat Med. 2017; 2017:8943059. PMID: 29234439.
Article13. Choi JY, Kim YJ, Cho SJ, Kwon EY, Ryu R, Choi MS. Metabolic effect of an oriental herbal medicine on obesity and its comorbidities with transcriptional responses in diet-induced obese mice. Int J Mol Sci. 2017; 18:18.
Article14. Ebrahimzadeh Attari V, Malek Mahdavi A, Javadivala Z, Mahluji S, Zununi Vahed S, Ostadrahimi A. A systematic review of the anti-obesity and weight lowering effect of ginger (Zingiber officinale Roscoe) and its mechanisms of action. Phytother Res. 2018; 32:577–585. PMID: 29193411.
Article15. Wang J, Ke W, Bao R, Hu X, Chen F. Beneficial effects of ginger Zingiber officinale Roscoe on obesity and metabolic syndrome: a review. Ann N Y Acad Sci. 2017; 1398:83–98. PMID: 28505392.16. Ali BH, Blunden G, Tanira MO, Nemmar A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): a review of recent research. Food Chem Toxicol. 2008; 46:409–420. PMID: 17950516.
Article17. Lau AJ, Seo BH, Woo SO, Koh HL. High-performance liquid chromatographic method with quantitative comparisons of whole chromatograms of raw and steamed Panax notoginseng. J Chromatogr A. 2004; 1057:141–149. PMID: 15584233.
Article18. Toh DF, New LS, Koh HL, Chan EC. Ultra-high performance liquid chromatography/time-of-flight mass spectrometry (UHPLC/TOFMS) for time-dependent profiling of raw and steamed Panax notoginseng. J Pharm Biomed Anal. 2010; 52:43–50. PMID: 20079593.
Article19. Toh DF, Patel DN, Chan EC, Teo A, Neo SY, Koh HL. Anti-proliferative effects of raw and steamed extracts of Panax notoginseng and its ginsenoside constituents on human liver cancer cells. Chin Med. 2011; 6:4. PMID: 21255464.
Article20. Yoo KY, Lee CH, Li H, Park JH, Choi JH, Hwang IK, Kang IJ, Won MH. Ethyl acetate extracts of raw and steamed Codonopsis lanceolata protects against ischemic damage potentially by maintaining SOD1 and BDNF levels. Int J Neurosci. 2011; 121:503–509. PMID: 21671837.21. Ho SC, Su MS. Evaluating the anti-neuroinflammatory capacity of raw and steamed garlic as well as five organosulfur compounds. Molecules. 2014; 19:17697–17714. PMID: 25365295.
Article22. Noda Y, Asada C, Sasaki C, Hashimoto S, Nakamura Y. Extraction method for increasing antioxidant activity of raw garlic using steam explosion. Biochem Eng J. 2013; 73:1–4.
Article23. Cheng XL, Liu Q, Peng YB, Qi LW, Li P. Steamed ginger (Zingiber officinale): changed chemical profile and increased anticancer potential. Food Chem. 2011; 129:1785–1792.
Article24. Iwasaki Y, Morita A, Iwasawa T, Kobata K, Sekiwa Y, Morimitsu Y, Kubota K, Watanabe T. A nonpungent component of steamed ginger--[10]-shogaol--increases adrenaline secretion via the activation of TRPV1. Nutr Neurosci. 2006; 9:169–178. PMID: 17176640.
Article25. Kim HJ, Kim B, Mun EG, Jeong SY, Cha YS. The antioxidant activity of steamed ginger and its protective effects on obesity induced by high-fat diet in C57BL/6J mice. Nutr Res Pract. 2018; 12:503–511. PMID: 30515278.
Article26. Park SH, Jung SJ, Choi EK, Ha KC, Baek HI, Park YK, Han KH, Jeong SY, Oh JH, Cha YS, Park BH, Chae SW. The effects of steamed ginger ethanolic extract on weight and body fat loss: a randomized, double-blind, placebo-controlled clinical trial. Food Sci Biotechnol. 2019; 29:265–273. PMID: 32064135.
Article27. Thomas SS, Kim M, Lee SJ, Cha YS. Antiobesity effects of purple perilla (Perilla frutescens var. acuta) on adipocyte differentiation and mice fed a high-fat diet. J Food Sci. 2018; 83:2384–2393. PMID: 30070698.28. Kim M, Song SB, Cha YS. Effects of black adzuki bean (Vigna angularis, Geomguseul) extract on body composition and hypothalamic neuropeptide expression in rats fed a high-fat diet. Food Nutr Res. 2015; 59:27719. PMID: 26493717.
Article29. Kim M, Park JE, Song SB, Cha YS. Effects of black adzuki bean (Vigna angularis) extract on proliferation and differentiation of 3T3-L1 preadipocytes into mature adipocytes. Nutrients. 2015; 7:277–292. PMID: 25569623.
Article30. Campos CF, Duarte MS, Guimarães SE, Verardo LL, Wei S, Du M, Jiang Z, Bergen WG, Hausman GJ, Fernyhough-Culver M, Albrecht E, Dodson MV. Review: animal model and the current understanding of molecule dynamics of adipogenesis. Animal. 2016; 10:927–932. PMID: 26776309.
Article31. Li HX, Xiao L, Wang C, Gao JL, Zhai YG. Review: epigenetic regulation of adipocyte differentiation and adipogenesis. J Zhejiang Univ Sci B. 2010; 11:784–791. PMID: 20872986.32. Suk S, Kwon GT, Lee E, Jang WJ, Yang H, Kim JH, Thimmegowda NR, Chung MY, Kwon JY, Yang S, Kim JK, Park JH, Lee KW. Gingerenone A, a polyphenol present in ginger, suppresses obesity and adipose tissue inflammation in high-fat diet-fed mice. Mol Nutr Food Res. 2017; 61:1700139.
Article33. Tzeng TF, Liu IM. 6-gingerol prevents adipogenesis and the accumulation of cytoplasmic lipid droplets in 3T3-L1 cells. Phytomedicine. 2013; 20:481–487. PMID: 23369342.
Article34. Tzeng TF, Chang CJ, Liu IM. 6-gingerol inhibits rosiglitazone-induced adipogenesis in 3T3-L1 adipocytes. Phytother Res. 2014; 28:187–192. PMID: 23519881.
Article35. Li C, Zhou L. Inhibitory effect 6-gingerol on adipogenesis through activation of the Wnt/β-catenin signaling pathway in 3T3-L1 adipocytes. Toxicol In Vitro. 2015; 30:394–401. PMID: 26498061.
Article36. Suk S, Seo SG, Yu JG, Yang H, Jeong E, Jang YJ, Yaghmoor SS, Ahmed Y, Yousef JM, Abualnaja KO, Al-Malki AL, Kumosani TA, Lee CY, Lee HJ, Lee KW. A bioactive constituent of ginger, 6-shogaol, prevents adipogenesis and stimulates lipolysis in 3T3-L1 adipocytes. J Food Biochem. 2016; 40:84–90.
Article37. Wei CK, Tsai YH, Korinek M, Hung PH, El-Shazly M, Cheng YB, Wu YC, Hsieh TJ, Chang FR. 6-paradol and 6-shogaol, the pungent compounds of ginger, promote glucose utilization in adipocytes and myotubes, and 6-paradol reduces blood glucose in high-fat diet-fed mice. Int J Mol Sci. 2017; 18:18.
Article38. Ahn EK, Oh JS. Inhibitory effect of galanolactone isolated from Zingiber officinale Roscoe extract on adipogenesis in 3T3-L1 cells. J Korean Soc Appl Biol Chem. 2012; 55:63–68.
Article39. Kuri-Harcuch W, Velez-delValle C, Vazquez-Sandoval A, Hernández-Mosqueira C, Fernandez-Sanchez V. A cellular perspective of adipogenesis transcriptional regulation. J Cell Physiol. 2019; 234:1111–1129. PMID: 30146705.
Article40. Ntambi JM, Young-Cheul K. Adipocyte differentiation and gene expression. J Nutr. 2000; 130:3122S–3126S. PMID: 11110885.
Article41. Farmer SR. Transcriptional control of adipocyte formation. Cell Metab. 2006; 4:263–273. PMID: 17011499.
Article42. Lee JE, Ge K. Transcriptional and epigenetic regulation of PPARγ expression during adipogenesis. Cell Biosci. 2014; 4:29. PMID: 24904744.
Article43. Kersten S. Mechanisms of nutritional and hormonal regulation of lipogenesis. EMBO Rep. 2001; 2:282–286. PMID: 11306547.
Article44. Semwal RB, Semwal DK, Combrinck S, Viljoen AM. Gingerols and shogaols: important nutraceutical principles from ginger. Phytochemistry. 2015; 117:554–568. PMID: 26228533.
Article45. Misawa K, Hashizume K, Yamamoto M, Minegishi Y, Hase T, Shimotoyodome A. Ginger extract prevents high-fat diet-induced obesity in mice via activation of the peroxisome proliferator-activated receptor δ pathway. J Nutr Biochem. 2015; 26:1058–1067. PMID: 26101135.
Article46. Fuhrman B, Rosenblat M, Hayek T, Coleman R, Aviram M. Ginger extract consumption reduces plasma cholesterol, inhibits LDL oxidation and attenuates development of atherosclerosis in atherosclerotic, apolipoprotein E-deficient mice. J Nutr. 2000; 130:1124–1131. PMID: 10801908.
Article47. Goyal RK, Kadnur SV. Beneficial effects of Zingiber officinale on goldthioglucose induced obesity. Fitoterapia. 2006; 77:160–163. PMID: 16513292.
Article48. Nammi S, Sreemantula S, Roufogalis BD. Protective effects of ethanolic extract of Zingiber officinale rhizome on the development of metabolic syndrome in high-fat diet-fed rats. Basic Clin Pharmacol Toxicol. 2009; 104:366–373. PMID: 19413656.49. Li Y, Tran VH, Kota BP, Nammi S, Duke CC, Roufogalis BD. Preventative effect of Zingiber officinale on insulin resistance in a high-fat high-carbohydrate diet-fed rat model and its mechanism of action. Basic Clin Pharmacol Toxicol. 2014; 115:209–215. PMID: 24428842.50. Bhandari U, Kanojia R, Pillai KK. Effect of ethanolic extract of Zingiber officinale on dyslipidaemia in diabetic rats. J Ethnopharmacol. 2005; 97:227–230. PMID: 15707757.
Article51. Bhandari U, Sharma JN, Zafar R. The protective action of ethanolic ginger (Zingiber officinale) extract in cholesterol fed rabbits. J Ethnopharmacol. 1998; 61:167–171. PMID: 9683348.
Article52. ElRokh SM, Yassin NA, El-Shenawy SM, Ibrahim BM. Antihypercholesterolaemic effect of ginger rhizome (Zingiber officinale) in rats. Inflammopharmacology. 2010; 18:309–315. PMID: 20730603.
Article53. Matsuda A, Wang Z, Takahashi S, Tokuda T, Miura N, Hasegawa J. Upregulation of mRNA of retinoid binding protein and fatty acid binding protein by cholesterol enriched-diet and effect of ginger on lipid metabolism. Life Sci. 2009; 84:903–907. PMID: 19379761.
Article54. Beattie JH, Nicol F, Gordon MJ, Reid MD, Cantlay L, Horgan GW, Kwun IS, Ahn JY, Ha TY. Ginger phytochemicals mitigate the obesogenic effects of a high-fat diet in mice: a proteomic and biomarker network analysis. Mol Nutr Food Res. 2011; 55(Suppl 2):S203–13. PMID: 21954187.
Article55. Son MJ, Miura Y, Yagasaki K. Mechanisms for antidiabetic effect of gingerol in cultured cells and obese diabetic model mice. Cytotechnology. 2015; 67:641–652. PMID: 24794903.
Article56. Saravanan G, Ponmurugan P, Deepa MA, Senthilkumar B. Anti-obesity action of gingerol: effect on lipid profile, insulin, leptin, amylase and lipase in male obese rats induced by a high-fat diet. J Sci Food Agric. 2014; 94:2972–2977. PMID: 24615565.
Article57. Tzeng TF, Liou SS, Chang CJ, Liu IM. [6]-gingerol dampens hepatic steatosis and inflammation in experimental nonalcoholic steatohepatitis. Phytomedicine. 2015; 22:452–461. PMID: 25925967.
Article58. Lei L, Liu Y, Wang X, Jiao R, Ma KY, Li YM, Wang L, Man SW, Sang S, Huang Y, Chen ZY. Plasma cholesterol-lowering activity of gingerol- and shogaol-enriched extract is mediated by increasing sterol excretion. J Agric Food Chem. 2014; 62:10515–10521. PMID: 25290252.
Article59. Wang J, Gao H, Ke D, Zuo G, Yang Y, Yamahara J, Li Y. Improvement of liquid fructose-induced adipose tissue insulin resistance by ginger treatment in rats is associated with suppression of adipose macrophage-related proinflammatory cytokines. Evid Based Complement Alternat Med. 2013; 2013:590376. PMID: 23533500.
Article60. de Las Heras N, Valero-Muñoz M, Martín-Fernández B, Ballesteros S, López-Farré A, Ruiz-Roso B, Lahera V. Molecular factors involved in the hypolipidemic- and insulin-sensitizing effects of a ginger (Zingiber officinale Roscoe) extract in rats fed a high-fat diet. Appl Physiol Nutr Metab. 2017; 42:209–215. PMID: 28125276.61. Al-Amin ZM, Thomson M, Al-Qattan KK, Peltonen-Shalaby R, Ali M. Anti-diabetic and hypolipidaemic properties of ginger (Zingiber officinale) in streptozotocin-induced diabetic rats. Br J Nutr. 2006; 96:660–666. PMID: 17010224.62. Abdulrazaq NB, Cho MM, Win NN, Zaman R, Rahman MT. Beneficial effects of ginger (Zingiber officinale) on carbohydrate metabolism in streptozotocin-induced diabetic rats. Br J Nutr. 2012; 108:1194–1201. PMID: 22152092.63. Jafri SA, Abass S, Qasim M. Hypoglycemic effect of ginger (Zingiber officinale) in alloxan induced diabetic rats (Rattus norvagicus). Pak Vet J. 2011; 31:160–162.64. Wang QA, Tao C, Gupta RK, Scherer PE. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat Med. 2013; 19:1338–1344. PMID: 23995282.
Article65. Kim S, Lee MS, Jung S, Son HY, Park S, Kang B, Kim SY, Kim IH, Kim CT, Kim Y. Ginger extract ameliorates obesity and inflammation via regulating microRNA-21/132 expression and AMPK activation in white adipose tissue. Nutrients. 2018; 10:10.
Article66. Frühbeck G, Méndez-Giménez L, Fernández-Formoso JA, Fernández S, Rodríguez A. Regulation of adipocyte lipolysis. Nutr Res Rev. 2014; 27:63–93. PMID: 24872083.
Article67. Wang H, Eckel RH. Lipoprotein lipase: from gene to obesity. Am J Physiol Endocrinol Metab. 2009; 297:E271–E288. PMID: 19318514.
Article68. Zechner R, Zimmermann R, Eichmann TO, Kohlwein SD, Haemmerle G, Lass A, Madeo F. FAT SIGNALS--lipases and lipolysis in lipid metabolism and signaling. Cell Metab. 2012; 15:279–291. PMID: 22405066.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Arctiin inhibits adipogenesis in 3T3-L1 cells and decreases adiposity and body weight in mice fed a high-fat diet
- Capsanthin Inhibits both Adipogenesis in 3T3-L1 Preadipocytes and Weight Gain in High-Fat Diet-Induced Obese Mice
- The Inhibitory Effect of Testosterone on PPARγ-induced Adipogenesis
- Expression of eotaxin in 3T3-L1 adipocytes and the effects of weight loss in high-fat diet induced obese mice
- NF-kappaB is involved in the TNF-alpha induced inhibition of the differentiation of 3T3-L1 cells by reducing PPARg expression