Efficacy and safety of vedolizumab in ulcerative colitis in patients from Asian countries in the GEMINI 1 study
- Affiliations
-
- 1Duke-NUS Medical School, Gleneagles Medical Centre, Singapore
- 2University Malaya Medical Centre, Kuala Lumpur, Malaysia
- 3Kyung Hee University School of Medicine, Seoul, Korea
- 4M. S. Ramaiah Medical College and Hospital, Bangalore, India
- 5Kaohsiung Medical University Hospital, Kaohsiung Medical University, Kaohsiung, Taiwan
- 6Takeda Pharmaceutical International AG, Singapore
- 7Takeda Pharmaceutical International AG, Zurich, Switzerland
- KMID: 2512103
- DOI: http://doi.org/10.5217/ir.2019.09159
Abstract
- Background/Aims
The efficacy and safety of vedolizumab in moderate to severely active ulcerative colitis (UC) have been demonstrated in the GEMINI 1 study (NCT00783718). This post-hoc exploratory analysis sought to establish the efficacy and safety of vedolizumab in a subgroup of patients from Asian countries with UC from GEMINI 1.
Methods
Efficacy outcomes of interest were clinical response, clinical remission and mucosal healing at week 6 (induction phase); and clinical remission, durable clinical response, durable clinical remission, mucosal healing and glucocorticoid-free remission at week 52 (maintenance phase). Differences in outcome rates between vedolizumab and placebo in Asian countries (Hong Kong, India, Malaysia, Singapore, South Korea, and Taiwan) were assessed using descriptive analyses, and efficacy and safety compared between Asian and non-Asian countries.
Results
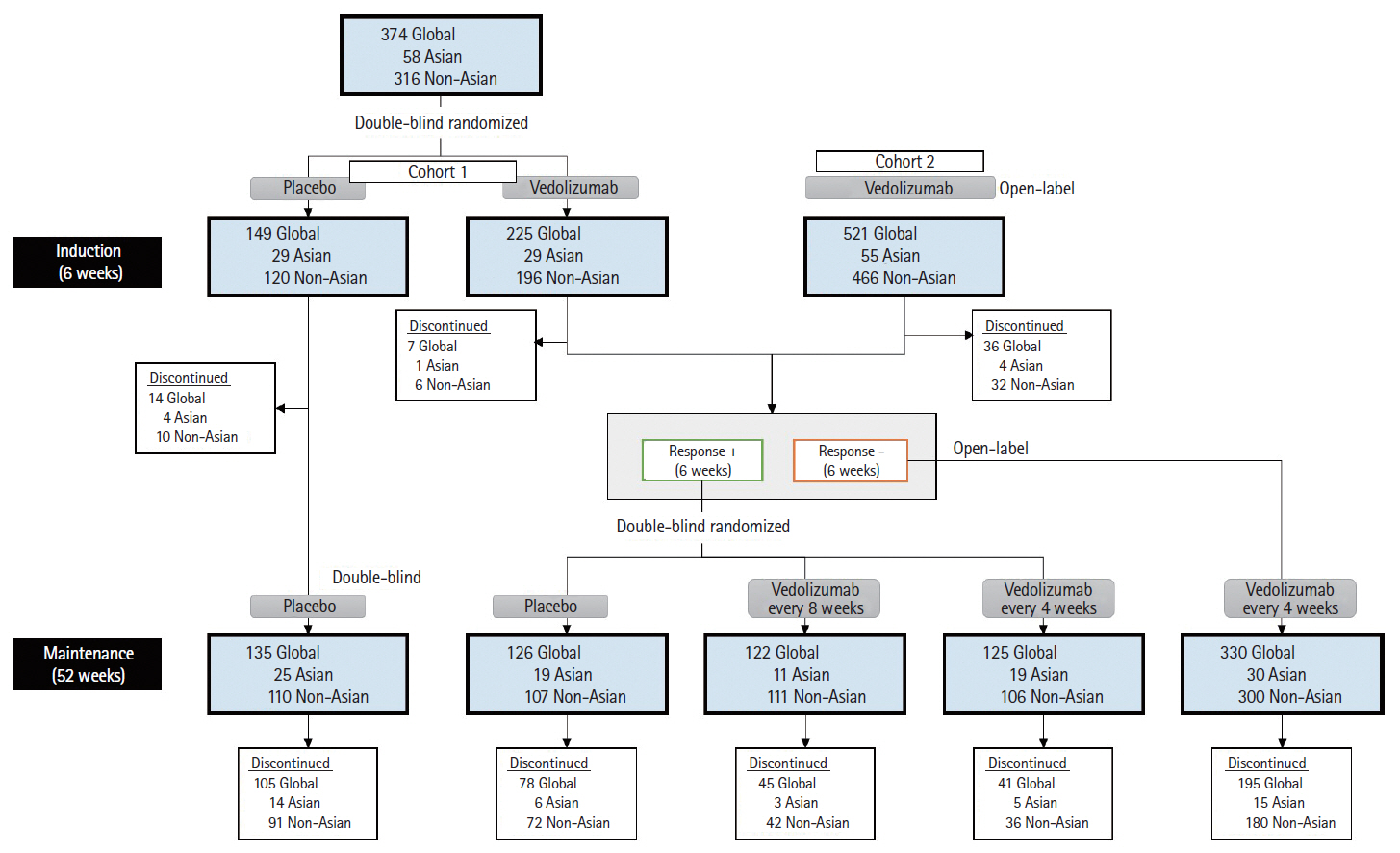
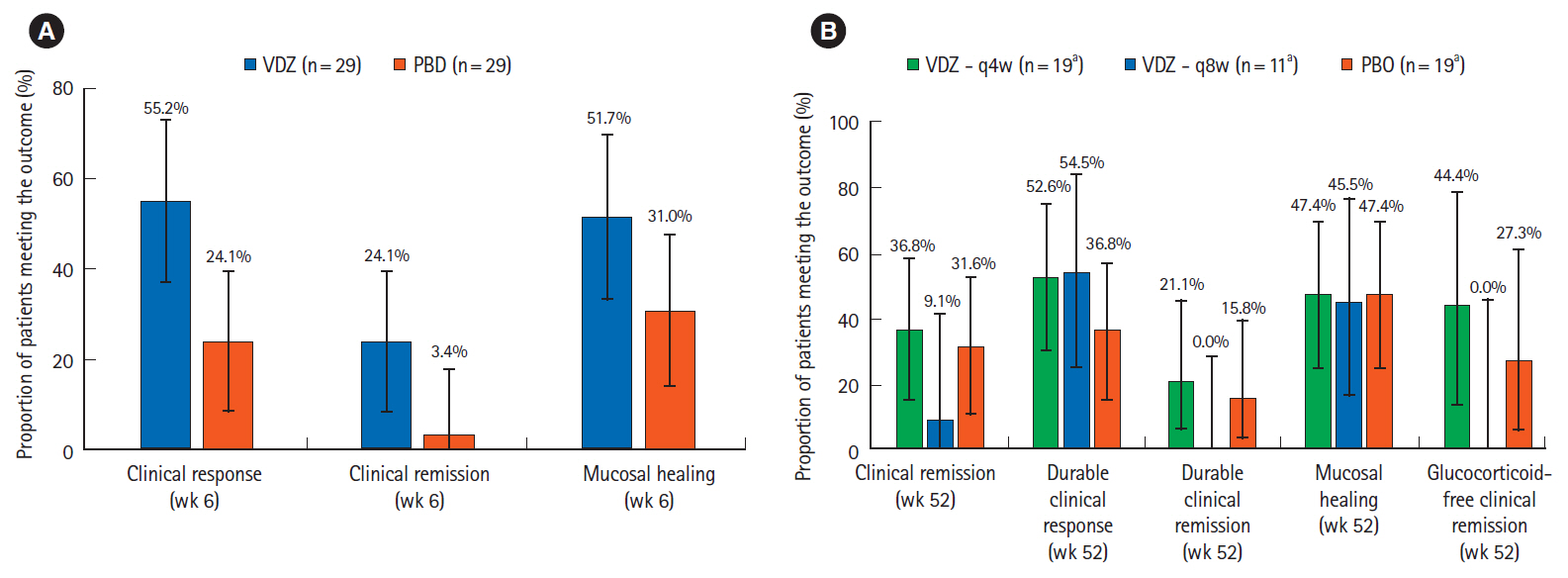
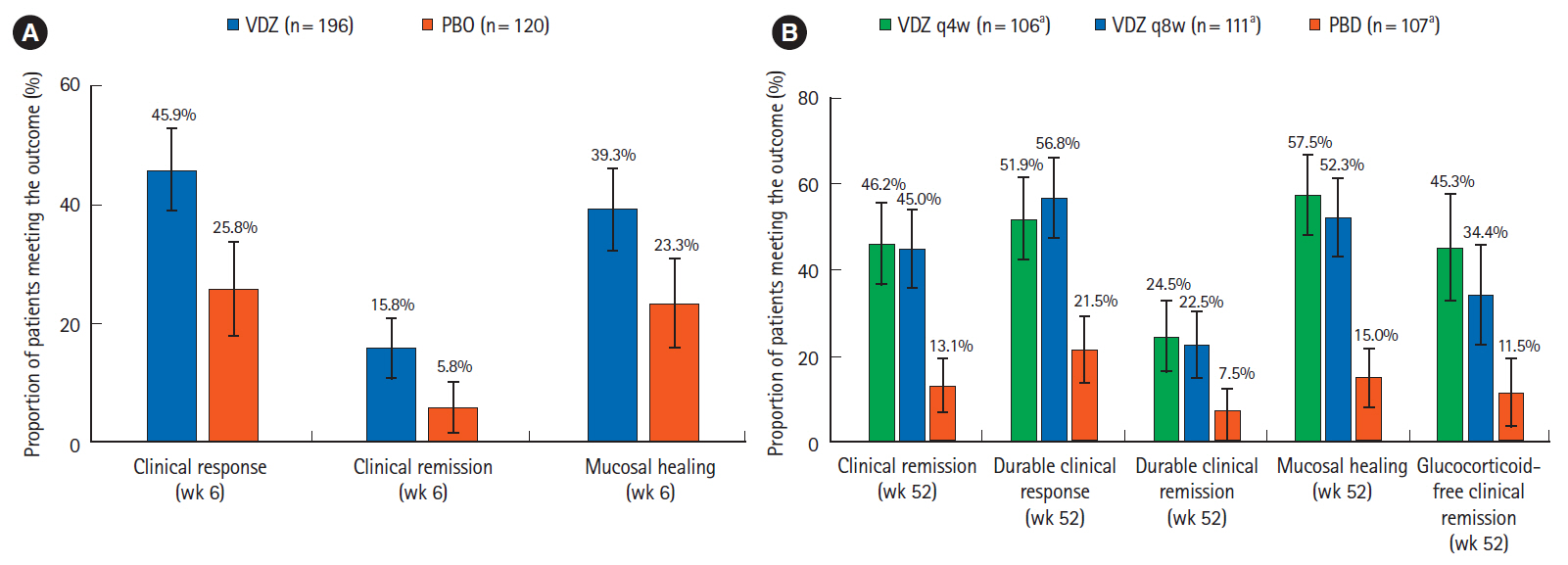
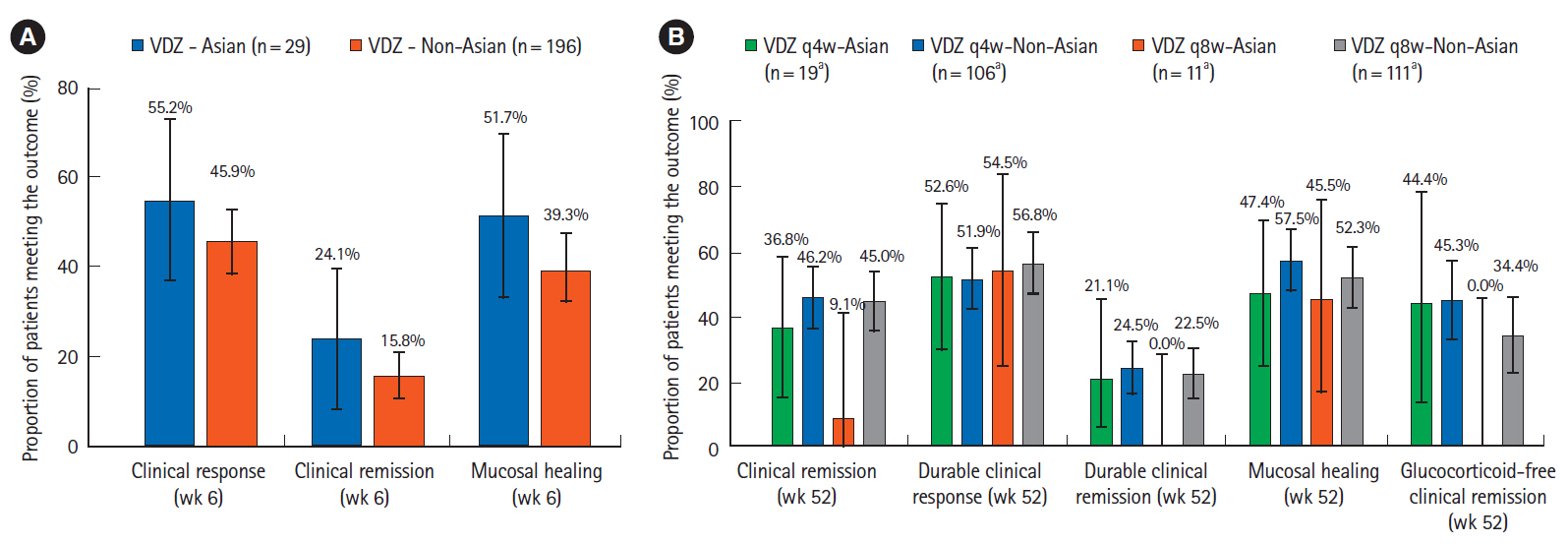
During induction, in Asian countries (n = 58), clinical response rates at week 6 with vedolizumab and placebo were 55.2% and 24.1%, respectively (difference 31.0%; 95% confidence interval: 7.2%–54.9%). In non-Asian countries (n = 316), response rates at week 6 with vedolizumab and placebo were 45.9% and 25.8%, respectively. During maintenance, in Asian countries, clinical remission rates at 52 weeks with vedolizumab administered every 8 weeks, vedolizumab administered every 4 weeks and placebo were 9.1%, 36.8%, and 31.6%, respectively; corresponding rates for mucosal healing were 45.5%, 47.4%, and 47.4%, respectively. Vedolizumab was well-tolerated; adverse event frequency was comparable in Asian and non-Asian countries.
Conclusions
In patients from Asian countries, the efficacy and safety of vedolizumab in treatment of UC were broadly consistent with that in the overall study population.
Figure
Cited by 4 articles
-
The Risk of Tuberculosis in Patients With Inflammatory Bowel Disease Treated With Vedolizumab or Ustekinumab in Korea
Myeong Geun Choi, Byong Duk Ye, Suk-Kyun Yang, Tae Sun Shim, Kyung-Wook Jo, Sang Hyoung Park
J Korean Med Sci. 2022;37(14):e107. doi: 10.3346/jkms.2022.37.e107.Vedolizumab does not increase perioperative surgical complications in patients with inflammatory bowel disease, cohort study
Vitaliy Y. Poylin, Jose Cataneo Serrato, Jonathan Pastrana Del Valle, Joseph D. Feuerstein
Intest Res. 2022;20(1):72-77. doi: 10.5217/ir.2020.00117.Natural history of inflammatory bowel disease: a comparison between the East and the West
Eun Mi Song, Suk-Kyun Yang
Intest Res. 2022;20(4):418-430. doi: 10.5217/ir.2021.00104.The role and prospect of tofacitinib in patients with ulcerative colitis
Jun Lee
Intest Res. 2023;21(1):168-169. doi: 10.5217/ir.2022.00098.
Reference
-
1. National Institute for Health and Care Excellence. Ulcerative colitis: management (NICE Guideline CG166) [Internet]. c2013 [cited 2018 Apr 10]. https://www.nice.org.uk/guidance/cg166.2. Feuerstein JD, Cheifetz AS. Ulcerative colitis: epidemiology, diagnosis, and management. Mayo Clin Proc. 2014; 89:1553–1563.3. Prideaux L, Kamm MA, De Cruz PP, Chan FK, Ng SC. Inflammatory bowel disease in Asia: a systematic review. J Gastroenterol Hepatol. 2012; 27:1266–1280.
Article4. Ng SC, Kaplan GG, Tang W, et al. Population density and risk of inflammatory bowel disease: a prospective population-based study in 13 countries or regions in Asia-Pacific. Am J Gastroenterol. 2019; 114:107–115.
Article5. Yang SK, Yun S, Kim JH, et al. Epidemiology of inflammatory bowel disease in the Songpa-Kangdong district, Seoul, Korea, 1986-2005: a KASID study. Inflamm Bowel Dis. 2008; 14:542–549.
Article6. Wong U, Cross RK. Primary and secondary nonresponse to infliximab: mechanisms and countermeasures. Expert Opin Drug Metab Toxicol. 2017; 13:1039–1046.
Article7. Ford AC, Peyrin-Biroulet L. Opportunistic infections with anti-tumor necrosis factor-α therapy in inflammatory bowel disease: meta-analysis of randomized controlled trials. Am J Gastroenterol. 2013; 108:1268–1276.
Article8. World Health Organization. Global tuberculosis report 2017 [Internet]. c2017 [cited 2018 Apr 10]. http://www.who.int/tb/publications/global_report/en/.9. Ungar B, Kopylov U. Advances in the development of new biologics in inflammatory bowel disease. Ann Gastroenterol. 2016; 29:243–248.
Article10. Feagan BG, Rutgeerts P, Sands BE, et al. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013; 369:699–710.
Article11. Schreiber S, Dignass A, Peyrin-Biroulet L, et al. Systematic review with meta-analysis: real-world effectiveness and safety of vedolizumab in patients with inflammatory bowel disease. J Gastroenterol. 2018; 53:1048–1064.
Article12. Ng SC, Hilmi IN, Blake A, et al. Low frequency of opportunistic infections in patients receiving vedolizumab in clinical trials and post-marketing setting. Inflamm Bowel Dis. 2018; 24:2431–2441.
Article13. Cheon JH. Understanding the complications of anti-tumor necrosis factor therapy in East Asian patients with inflammatory bowel disease. J Gastroenterol Hepatol. 2017; 32:769–777.
Article14. Leung WK, Ng SC, Chow DK, et al. Use of biologics for inflammatory bowel disease in Hong Kong: consensus statement. Hong Kong Med J. 2013; 19:61–68.15. Leung WK. Optimization of inflammatory bowel disease cohort studies in Asia. Intest Res. 2015; 13:208–212.
Article16. Feagan BG, Rubin DT, Danese S, et al. Efficacy of vedolizumab induction and maintenance therapy in patients with ulcerative colitis, regardless of prior exposure to tumor necrosis factor antagonists. Clin Gastroenterol Hepatol. 2017; 15:229–239.
Article17. Colombel JF, Keir ME, Scherl A, et al. Discrepancies between patient-reported outcomes, and endoscopic and histological appearance in UC. Gut. 2017; 66:2063–2068.
Article18. Torres J, Billioud V, Sachar DB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis as a progressive disease: the forgotten evidence. Inflamm Bowel Dis. 2012; 18:1356–1363.
Article19. Motoya S, Watanabe K, Ogata H, et al. Vedolizumab in Japanese patients with ulcerative colitis: a phase 3, randomized, double-blind, placebo-controlled study. PLoS One. 2019; 14:e0212989.
Article20. Gan AT, Chan WP, Ling KL, et al. P634 Real-world data on the efficacy and safety of vedolizumab therapy in patients with inflammatory bowel disease: a retrospective nation-wide cohort study in Singapore. J Crohns Colitis. 2019; 13(Suppl 1):S434–S435.
Article21. Kim J, Ham NS, Oh EH, et al. P277 Real life effectiveness and safety of vedolizumab induction and maintenance therapy for Korean IBD patients in whom anti-TNF treatment failed: a prospective cohort study. J Crohns Colitis. 2019; 13(Suppl 1):S237.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Efficacy and safety of vedolizumab in Crohn’s disease in patients from Asian countries in the GEMINI 2 study
- Combined eosinophilic gastroenteritis and ulcerative colitis successfully treated by vedolizumab: a case report
- Population pharmacokinetics of vedolizumab in Asian and non-Asian patients with ulcerative colitis and Crohn’s disease
- Efficacy and safety of a new vedolizumab subcutaneous formulation in Japanese patients with moderately to severely active ulcerative colitis
- Treatment of inflammatory bowel diseases: focusing on biologic agents and new therapies