J Liver Cancer.
2020 Sep;20(2):148-153. 10.17998/jlc.20.2.148.
Long-term Survival of a Patient with a Large Hepatocellular Carcinoma with Main Portal Vein Tumor Thrombosis and Spontaneous Tumor Rupture
- Affiliations
-
- 1Department of Internal Medicine, Kyung Hee University School of Medicine, Seoul, Korea
- KMID: 2508026
- DOI: http://doi.org/10.17998/jlc.20.2.148
Abstract
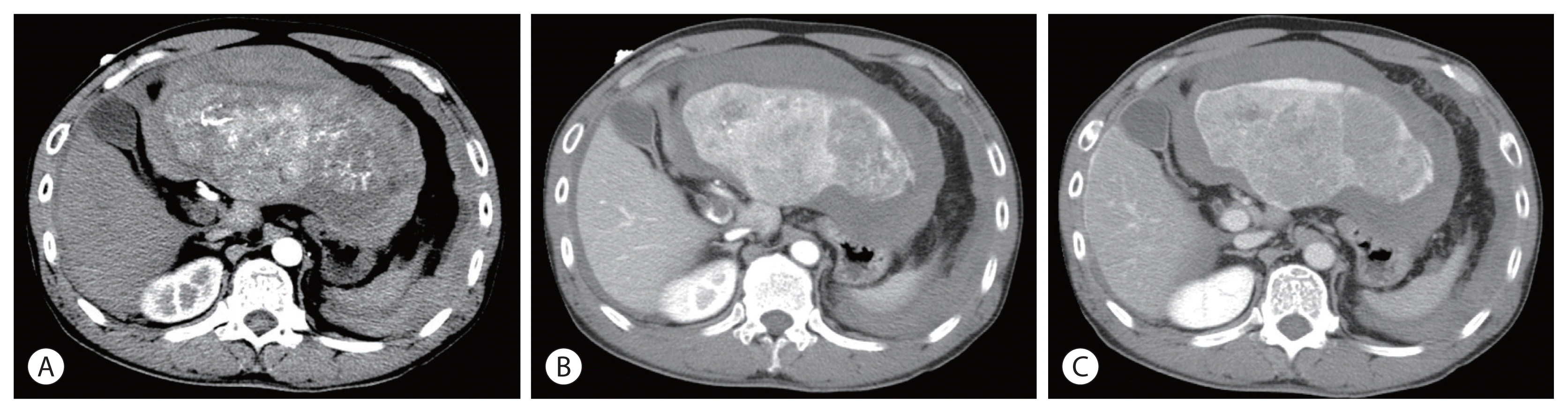
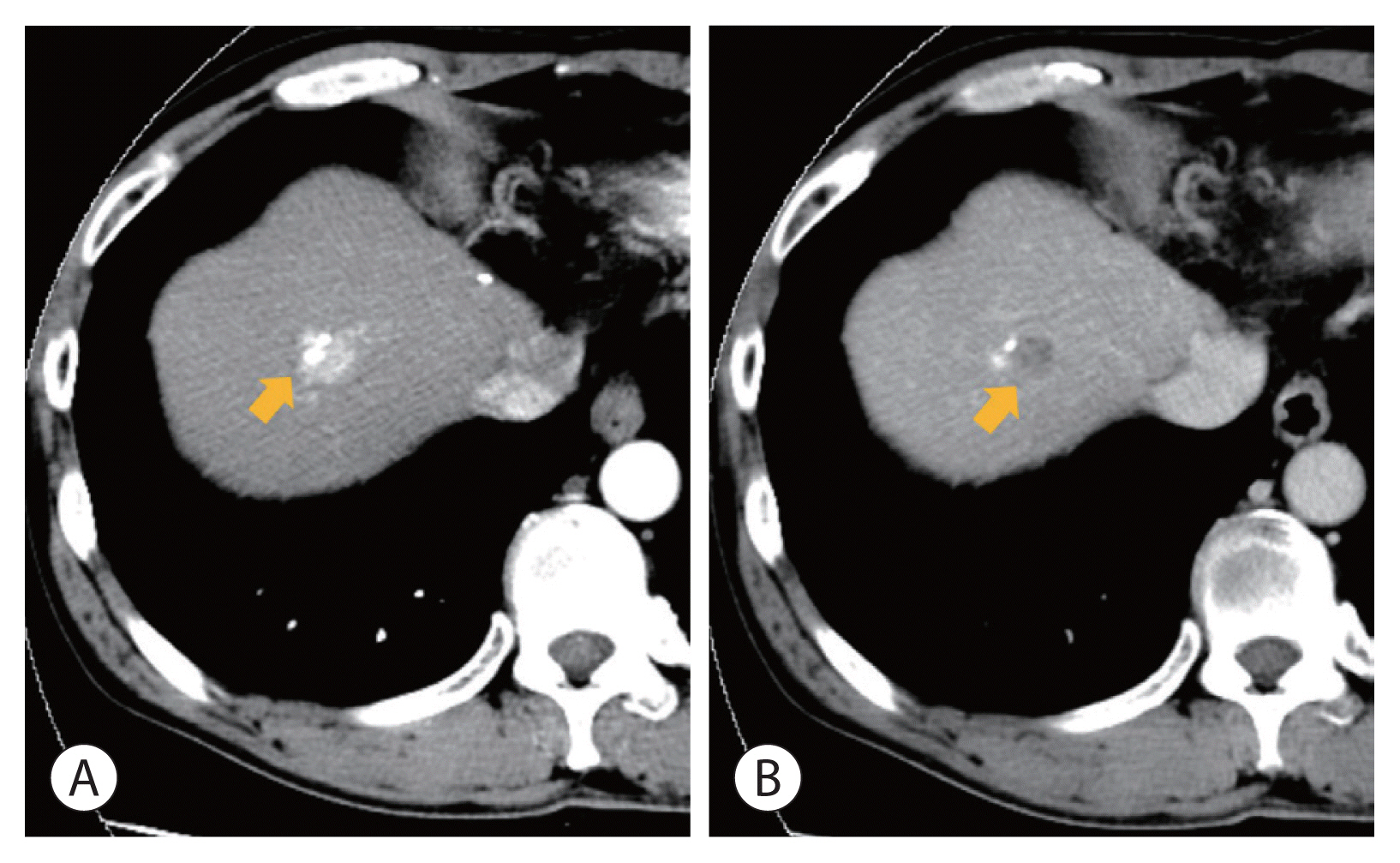
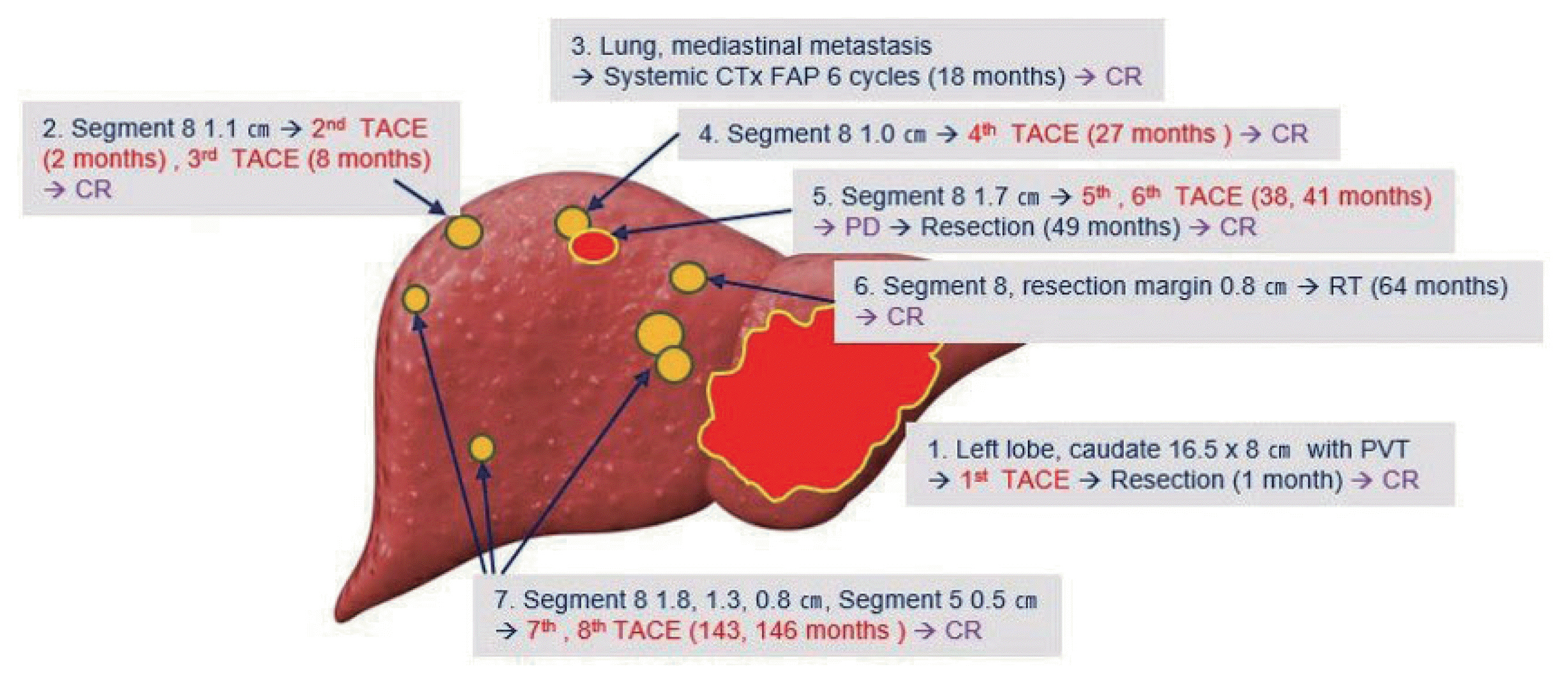
- Optimal treatments for patients with advanced hepatocellular carcinoma (HCC) are still limited and their prognosis remains dismal. Yet, there have been rare cases that have shed light on longer survival in these patients assisted by various treatments. This paper aims to present an extraordinary case of far advanced HCC that had been properly managed in spite of continuous recurrence. A patient visited the hospital with a ruptured large HCC with main portal vein tumor thrombosis but survived longer than 14 years owing to active and prompt interventions.
Figure
Reference
-
1. Jung KW, Won YJ, Kong HJ, Lee ES. Cancer statistics in Korea: incidence, mortality, survival, and prevalence in 2016. Cancer Res Treat. 2019; 51:417–430.2. Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008; 359:378–390.3. Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018; 391:1163–1173.4. Yoon SM, Ryoo BY, Lee SJ, Kim JH, Shin JH, An JH, et al. Efficacy and safety of transarterial chemoembolization plus external beam radiotherapy vs sorafenib in hepatocellular carcinoma with macroscopic vascular invasion: a randomized clinical trial. JAMA Oncol. 2018; 4:661–669.5. He M, Li Q, Zou R, Shen J, Fang W, Tan G, et al. Sorafenib plus hepatic arterial infusion of oxaliplatin, fluorouracil, and leucovorin vs sorafenib alone for hepatocellular carcinoma with portal vein invasion: a randomized clinical trial. JAMA Oncol. 2019; 5:953–960.6. Fernandes ESM, Rodrigues PD, Álvares-da-Silva MR, Scaffaro LA, Farenzena M, Teixeira UF, et al. Treatment strategies for locally advanced hepatocellular carcinoma. Transl Gastroenterol Hepatol. 2019; 4:12.7. Yoshida H, Mamada Y, Taniai N, Uchida E. Spontaneous ruptured hepatocellular carcinoma. Hepatol Res. 2016; 46:13–21.8. Aoki T, Kokudo N, Matsuyama Y, Izumi N, Ichida T, Kudo M, et al. Prognostic impact of spontaneous tumor rupture in patients with hepatocellular carcinoma: an analysis of 1160 cases from a nationwide survey. Ann Surg. 2014; 259:532–542.9. Kokudo T, Hasegawa K, Matsuyama Y, Takayama T, Izumi N, Kadoya M, et al. Survival benefit of liver resection for hepatocellular carcinoma associated with portal vein invasion. J Hepatol. 2016; 65:938–943.10. Lee JM, Jang BK, Lee YJ, Choi WY, Choi SM, Chung WJ, et al. Survival outcomes of hepatic resection compared with transarterial chemoembolization or sorafenib for hepatocellular carcinoma with portal vein tumor thrombosis. Clin Mol Hepatol. 2016; 22:160–167.11. Korean Liver Cancer Association; National Cancer Center. 2018 Korean Liver Cancer Association-National Cancer Center Korea practice guideline for the management of hepatocellular carcinoma. Gut Liver. 2019; 13:227–299.12. Li L, Zhao W, Wang M, Hu J, Wang E, Zhao Y, et al. Transarterial chemoembolization plus sorafenib for the management of unresectable hepatocellular carcinoma: a systematic review and meta-analysis. BMC Gastroenterol. 2018; 18:138.13. Choi GH, Shim JH, Kim MJ, Ryu MH, Ryoo BY, Kang YK, et al. Sorafenib alone versus sorafenib combined with transarterial chemoembolization for advanced-stage hepatocellular carcinoma: results of propensity score analyses. Radiology. 2013; 269:603–611.14. Chen J, Lu S, Zhang Y, Xu L, Chen J, Wang J, et al. Sorafenib monotherapy versus sorafenib combined with regional therapies for hepatocellular carcinoma patients with pulmonary oligometastases: a propensity score-matched analysis. J Cancer. 2018; 9:1745–1753.15. Jeon MY, Kim HS, Lim TS, Han DH, Kim BK, Park JY, et al. Refractoriness to transarterial chemoembolization in patients with recurrent hepatocellular carcinoma after curative resection. PLoS One. 2019; 14:e0214613.16. Galle PR, Tovoli F, Foerster F, Wörns MA, Cucchetti A, Bolondi L. The treatment of intermediate stage tumours beyond TACE: From surgery to systemic therapy. J Hepatol. 2017; 67:173–183.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Characteristics and Prognosis after Resection for Ruptured Hepatocellular Carcinoma
- Reappraisal of transarterial radioembolization for liver-confined hepatocellular carcinoma with portal vein tumor thrombosis: Editorial on “Transarterial radioembolization versus tyrosine kinase inhibitor in hepatocellular carcinoma with portal vein thrombosis”
- Clinical features of portal vein thrombosis in hepatocellular carcinoma
- Liver transplantation for hepatocellular carcinoma with portal vein tumor thrombosis
- Sonographic features of portal vein thrombosis