Nutr Res Pract.
2020 Oct;14(5):453-462. 10.4162/nrp.2020.14.5.453.
Immunomodulatory effects of fermented Platycodon grandiflorum extract through NF-κB signaling in RAW 264.7 cells
- Affiliations
-
- 1Department of Food and Nutrition, College of BioNano Technology, Gachon University, Seongnam 13120, Korea
- 2Institute for Aging and Clinical Nutrition Research, Gachon University, Seongnam 13120, Korea
- KMID: 2506883
- DOI: http://doi.org/10.4162/nrp.2020.14.5.453
Abstract
- BACKGROUND/OBJECTIVES
Platycodon grandiflorum (PG), an oriental herbal medicine, has been known to improve liver function, and has both anti-inflammatory and antimicrobial properties. However, little is known about the immune-enhancing effects of PG and its mechanism. In this study, we aimed to investigate whether fermented PG extract (FPGE), which has increased platycodin D content, activates the immune response in a murine macrophage cell line, RAW 264.7.
MATERIALS/METHODS
Cell viability was determined by Cell Counting Kit-8 assay and the nitric oxide (NO) levels were measured using Griess reagent. Cytokine messenger RNA levels of were monitored by quantitative reverse transcription polymerase chain reaction. To investigate the molecular mechanisms underlying immunomodulatory actions of FPGE in RAW 264.7 cells, we have conducted luciferase reporter gene assay and western blotting.
RESULTS
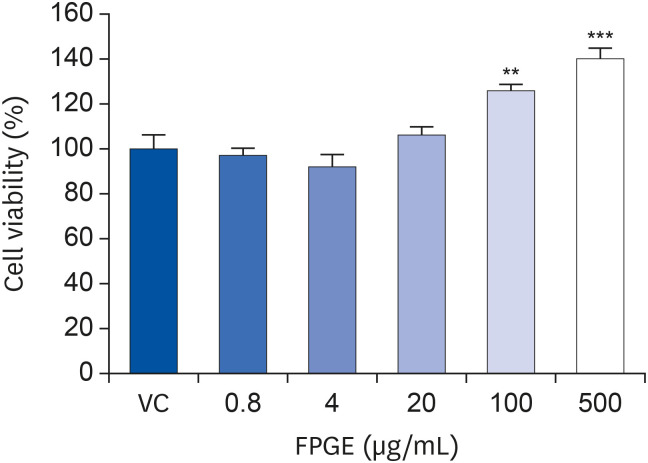
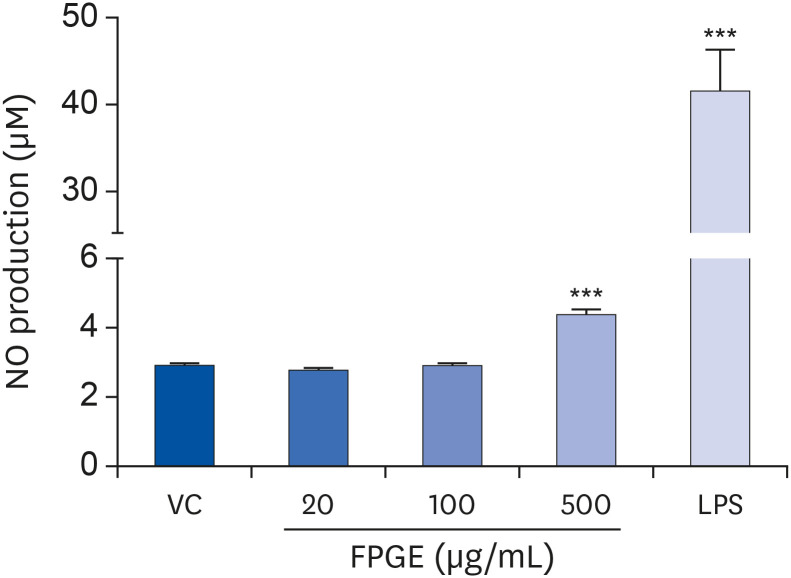
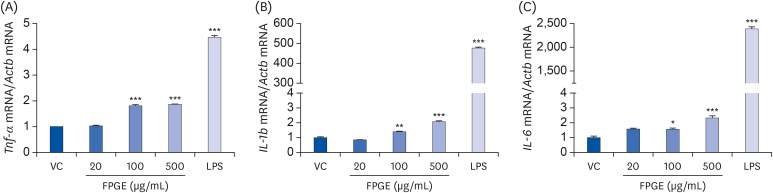
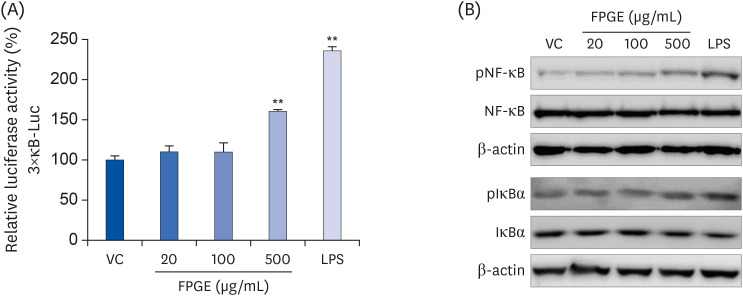
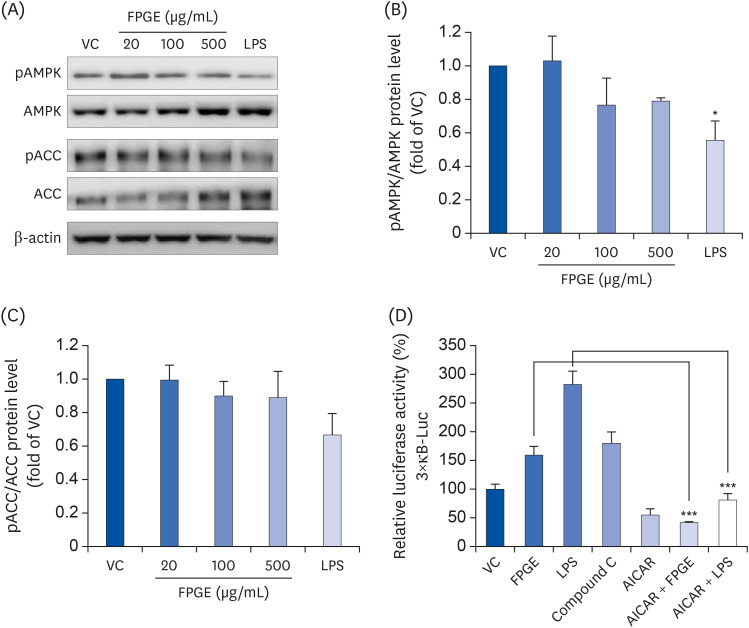
We found that FPGE treatment induced macrophage cell proliferation in a dosedependent manner. FPGE also modulated the expression of NO and pro-inflammatory cytokines, such as tumor necrosis factor-α, interleukin (IL)-1β, and IL-6. The activation and phosphorylation levels of nuclear factor kappa B (NF-κB) were increased by FPGE treatment. Moreover, 5-aminoimidazole-4-carboxamide ribonucleotide, an activator of AMP-activated kinase (AMPK), significantly reduced both lipopolysaccharides- and FPGE-induced NF-κB reporter gene activity.
CONCLUSIONS
Taken together, our findings suggest that FPGE may be a novel immuneenhancing agent acting via AMPK-NF-κB signaling pathway.
Figure
Reference
-
1. Liu RH. Health-promoting components of fruits and vegetables in the diet. Adv Nutr. 2013; 4:384S–392S. PMID: 23674808.
Article2. Park SY, Park JH, Kim HS, Lee CY, Lee HJ, Kang KS, Kim CE. Systems-level mechanisms of action of Panax ginseng: a network pharmacological approach. J Ginseng Res. 2018; 42:98–106. PMID: 29348728.3. Azab A, Nassar A, Azab AN. Anti-inflammatory activity of natural products. Molecules. 2016; 21:1321.
Article4. Kim GT, Tran NK, Choi EH, Song YJ, Song JH, Shim SM, Park TS. Immunomodulatory efficacy of standardized Annona muricata (Graviola) leaf extract via activation of mitogen-activated protein kinase pathways in RAW 264.7 macrophages. Evid Based Complement Alternat Med. 2016; 2016:2905127. PMID: 28096884.5. Pan G, Xie Z, Huang S, Tai Y, Cai Q, Jiang W, Sun J, Yuan Y. Immune-enhancing effects of polysaccharides extracted from Lilium lancifolium Thunb. Int Immunopharmacol. 2017; 52:119–126. PMID: 28898768.6. Zhang Q, Lenardo MJ, Baltimore D. 30 years of NF-κB: a blossoming of relevance to human pathobiology. Cell. 2017; 168:37–57. PMID: 28086098.
Article7. Hesketh M, Sahin KB, West ZE, Murray RZ. Macrophage phenotypes regulate scar formation and chronic wound healing. Int J Mol Sci. 2017; 18:1545.
Article8. Tugal D, Liao X, Jain MK. Transcriptional control of macrophage polarization. Arterioscler Thromb Vasc Biol. 2013; 33:1135–1144. PMID: 23640482.
Article9. Yang Z, Kahn BB, Shi H, Xue BZ. Macrophage alpha1 AMP-activated protein kinase (alpha1AMPK) antagonizes fatty acid-induced inflammation through SIRT1. J Biol Chem. 2010; 285:19051–19059. PMID: 20421294.10. Day EA, Ford RJ, Steinberg GR. AMPK as a therapeutic target for treating metabolic diseases. Trends Endocrinol Metab. 2017; 28:545–560. PMID: 28647324.
Article11. Sag D, Carling D, Stout RD, Suttles J. Adenosine 5′-monophosphate-activated protein kinase promotes macrophage polarization to an anti-inflammatory functional phenotype. J Immunol. 2008; 181:8633–8641. PMID: 19050283.
Article12. Chung JW, Noh EJ, Zhao HL, Sim JS, Ha YW, Shin EM, Lee EB, Cheong CS, Kim YS. Anti-inflammatory activity of prosapogenin methyl ester of platycodin D via nuclear factor-kappaB pathway inhibition. Biol Pharm Bull. 2008; 31:2114–2120. PMID: 18981583.
Article13. Nyakudya E, Jeong JH, Lee NK, Jeong YS. Platycosides from the roots of Platycodon grandiflorum and their health benefits. Prev Nutr Food Sci. 2014; 19:59–68. PMID: 25054103.14. Leng J, Wang Z, Fu CL, Zhang J, Ren S, Hu JN, Jiang S, Wang YP, Chen C, Li W. NF-κB and AMPK/PI3K/Akt signaling pathways are involved in the protective effects of Platycodon grandiflorum saponins against acetaminophen-induced acute hepatotoxicity in mice. Phytother Res. 2018; 32:2235–2246. PMID: 30039882.15. Kim JI, Jeon SG, Kim KA, Kim JJ, Song EJ, Jeon Y, Kim E, Lee KB, Kwak JH, Moon M. Platycodon grandiflorus root extract improves learning and memory by enhancing synaptogenesis in mice hippocampus. Nutrients. 2017; 9:794.16. Wang C, Schuller Levis GB, Lee EB, Levis WR, Lee DW, Kim BS, Park SY, Park E. Platycodin D and D3 isolated from the root of Platycodon grandiflorum modulate the production of nitric oxide and secretion of TNF-alpha in activated RAW 264.7 cells. Int Immunopharmacol. 2004; 4:1039–1049. PMID: 15222978.17. Zhao X, Wang Y, Yan P, Cheng G, Wang C, Geng N, Wang X, Liu J. Effects of polysaccharides from Platycodon grandiflorum on immunity-enhancing activity in vitro. Molecules. 2017; 22:1918.18. Song HS, Lee S, Han EH, Yu HJ, Lim MK. Screening for substances with synergistic anti-inflammatory effects in combination with fermented Platycodon grandiflorum extracts in LPS-stimulated RAW264.7 cells. J Korean Soc Food Sci Nutr. 2019; 48:282–289.19. Chun JN, Park S, Lee S, Kim JK, Park EJ, Kang M, Kim HK, Park JK, So I, Jeon JH. Schisandrol B and schisandrin B inhibit TGFβ1-mediated NF-κB activation via a Smad-independent mechanism. Oncotarget. 2017; 9:3121–3130. PMID: 29423034.
Article20. Bogdan C. Nitric oxide and the immune response. Nat Immunol. 2001; 2:907–916. PMID: 11577346.
Article21. Lee YS, Cho IJ, Kim JW, Lee SK, Ku SK, Lee HJ. Evaluation of in vitro anti-oxidant and anti-inflammatory activities of Korean and Chinese Lonicera caerulea . Nutr Res Pract. 2018; 12:486–493. PMID: 30515276.22. Lee S, Lee D, Jang TS, Kang KS, Nam JW, Lee HJ, Kim KH. Anti-inflammatory phenolic metabolites from the edible fungus Phellinus baumii in LPS-stimulated RAW264.7 cells. Molecules. 2017; 22:1583.23. Sun H, Chen L, Wang J, Wang K, Zhou J. Structure-function relationship of the saponins from the roots of Platycodon grandiflorum for hemolytic and adjuvant activity. Int Immunopharmacol. 2011; 11:2047–2056. PMID: 21945665.24. Spelman K, Burns J, Nichols D, Winters N, Ottersberg S, Tenborg M. Modulation of cytokine expression by traditional medicines: a review of herbal immunomodulators. Altern Med Rev. 2006; 11:128–150. PMID: 16813462.25. Choi EY, Jin JY, Hyeon JY, Choe SH, Keum BR, Lim JM, Park DC, Cho KK, Choi IS. Immunomodulatory activity of commercial β-glucan in murine macrophage cell line RAW 264.7. J Environ Biol. 2018; 39:166–174.26. Choi EY, Lee SS, Hyeon JY, Choe SH, Keum BR, Lim JM, Park DC, Choi IS, Cho KK. Effects of β-glucan on the release of nitric oxide by macrophages stimulated with lipopolysaccharide. Asian-Australas J Anim Sci. 2016; 29:1664–1674. PMID: 27488844.
Article27. Park SY, Kim HB, Kim JH, Lee JM, Kim SR, Shin HS, Yi TH. Immunostimulatory effect of fermented red ginseng in the mouse model. Prev Nutr Food Sci. 2014; 19:10–18. PMID: 24772404.
Article28. Kang S, Min H. Ginseng, the ‘immunity boost’: the effects of Panax ginseng on immune system. J Ginseng Res. 2012; 36:354–368. PMID: 23717137.29. Kim HL, Park J, Park H, Jung Y, Youn DH, Kang J, Jeong MY, Um JY. Platycodon grandiflorum A. De candolle ethanolic extract inhibits adipogenic regulators in 3T3-L1 cells and induces mitochondrial biogenesis in primary brown preadipocytes. J Agric Food Chem. 2015; 63:7721–7730. PMID: 26244589.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Immunostimulatory activity of hydrolyzed and fermented Platycodon grandiflorum extract occurs via the MAPK and NF-κB signaling pathway in RAW 264.7 cells
- Galangin Regulates Mucin 5AC Gene Expression via the Nuclear Factor-κB Inhibitor α/Nuclear Factor-κB p65 Pathway in Human Airway Epithelial Cells
- Polysaccharide from Hizikia Fusiformis Enhances the Immunomodulatory Activity of Macrophages
- Anti-inflammatory effects of proanthocyanidin-rich red rice extract via suppression of MAPK, AP-1 and NF-κB pathways in Raw 264.7 macrophages
- Immunomodulatory effects mixed with Weissella cibaria JW15 and Black soybean (Glycine max (L.) Merr.) Extract