J Vet Sci.
2020 Mar;21(2):e30. 10.4142/jvs.2020.21.e30.
ImprovingMycoplasma ovipneumoniae culture medium by a comparative transcriptome method
- KMID: 2506184
- DOI: http://doi.org/10.4142/jvs.2020.21.e30
Abstract
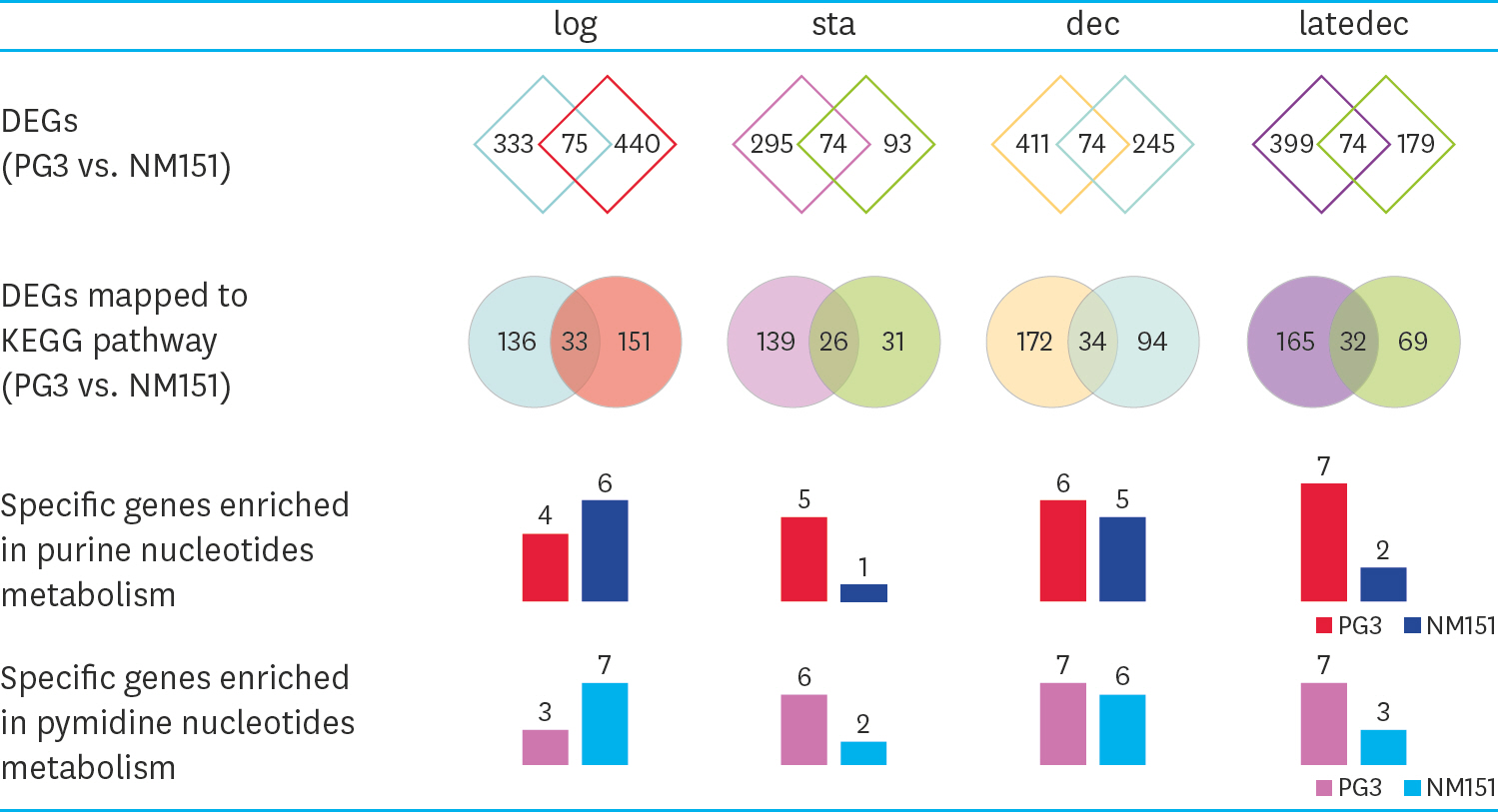
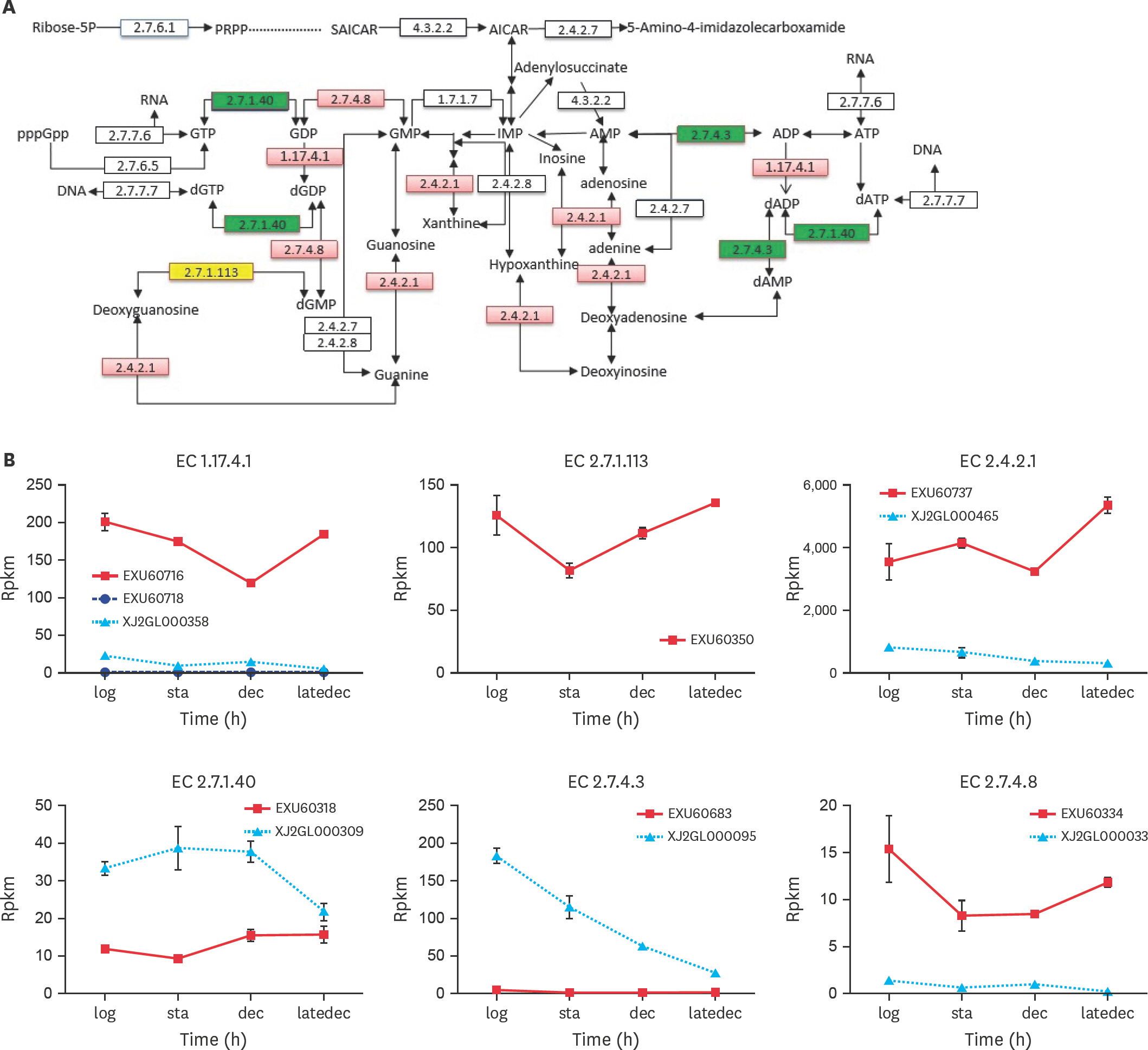
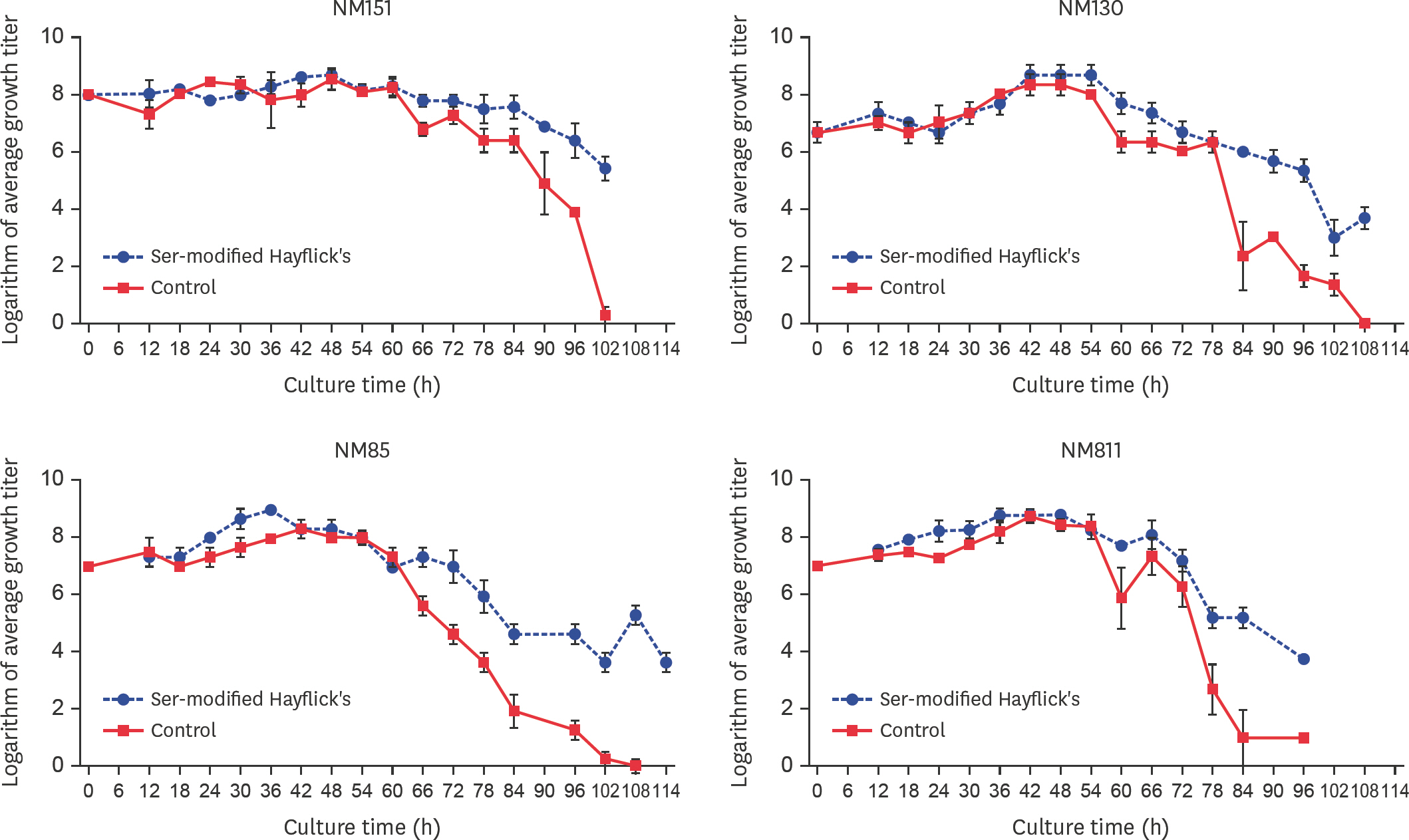
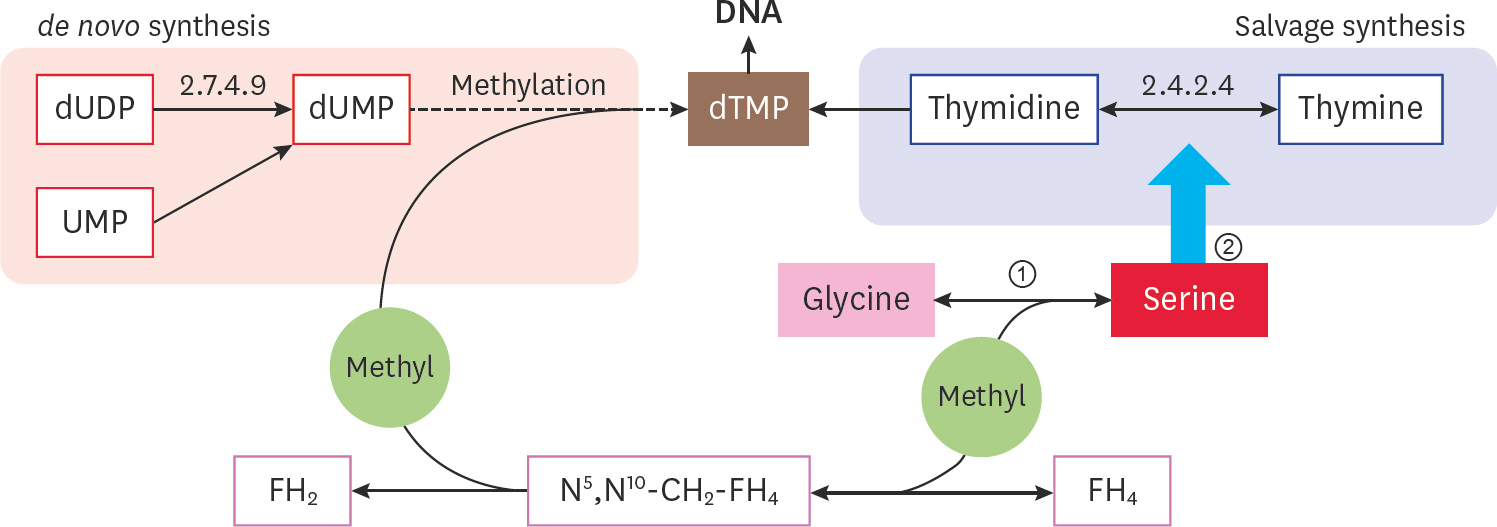
- Mycoplasma ovipneumoniae (Mo) is difficult to culture, resulting in many difficulties in related research and application. Since nucleotide metabolism is a basic metabolism affects growth, this study conducted a “point-to-point” comparison of the corresponding growth phases between the Mo NM151 strain and theMycoplasma mycoides subsp. capri (Mmc) PG3 strain. The results showed that the largest difference in nucleotide metabolism was found in the stationary phase. Nucleotide synthesis in PG3 was mostly de novo, while nucleotide synthesis in NM151 was primarily based on salvage synthesis. Compared with PG3, the missing reactions of NM151 referred to the synthesis of deoxythymine monophosphate. We proposed and validated a culture medium with added serine to fill this gap and prolong the stationary phase of NM151. This solved the problem of the fast death of Mo, which is significant for related research and application.
Figure
Reference
-
References
1. Wang XH, Wang YF, Huang HB, Bai F, Shi X, Ma C, Gao Y, Zhang J, Zhang W, Hao Y. Understanding the metabolism of Mycoplasma mycoides subsp. capri in vitro by a transcriptomic analysis. J Integr Agric. 2018; 17:428–435.2. Razin S, Yogev D, Naot Y. Molecular biology and pathogenicity of mycoplasmas. Microbiol Mol Biol Rev. 1998; 62:1094–1156.
Article3. Miles RJ. Catabolism in mollicutes. J Gen Microbiol. 1992; 138:1773–1783.
Article4. Wodke JA, Puchałka J, Lluch-Senar M, Marcos J, Yus E, Godinho M, Gutiérrez-Gallego R, dos Santos VA, Serrano L, Klipp E, Maier T. Dissecting the energy metabolism in Mycoplasma pneumoniae through genome-scale metabolic modeling. Mol Syst Biol. 2013; 9:653.5. Yus E, Maier T, Michalodimitrakis K, van Noort V, Yamada T, Chen WH, Wodke JA, Güell M, Martínez S, Bourgeois R, Kühner S, Raineri E, Letunic I, Kalinina OV, Rode M, Herrmann R, Gutiérrez-Gallego R, Russell RB, Gavin AC, Bork P, Serrano L. Impact of genome reduction on bacterial metabolism and its regulation. Science. 2009; 326:1263–1268.
Article6. Wang XH, Huang HB, Cheng C, Wang RC, Zheng JQ, Hao YQ, Zhang W. Complete genome sequence of Mycoplasma ovipneumoniae strain NM2010, which was isolated from a sheep in China. J Integr Agric. 2014; 13:2562–2563.7. Xu CG, Hao YQ, Zhang L, Hao RX, Liu XL, Huang ZY. Molecular cloning and immune response analysis of putative variable lipoproteins from Mycoplasma mycoides subsp capri. Genet Mol Res. 2014; 13:1527–1539.8. Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods. 2012; 9:357–359.
Article9. McClure R, Balasubramanian D, Sun Y, Bobrovskyy M, Sumby P, Genco CA, Vanderpool CK, Tjaden B. Computational analysis of bacterial RNA-Seq data. Nucleic Acids Res. 2013; 41:e140.
Article10. Sanavia T, Finotello F, Di Camillo B. FunPat: function-based pattern analysis on RNA-seq time series data. BMC Genomics. 2015; 16:S2.
Article11. Kanehisa M, Goto S, Hattori M, Aoki-Kinoshita KF, Itoh M, Kawashima S, Katayama T, Araki M, Hirakawa M. From genomics to chemical genomics: new developments in KEGG. Nucleic Acids Res. 2006; 34:D354–D357.
Article12. Kanehisa M, Araki M, Goto S, Hattori M, Hirakawa M, Itoh M, Katayama T, Kawashima S, Okuda S, Tokimatsu T, Yamanishi Y. KEGG for linking genomes to life and the environment. Nucleic Acids Res. 2008; 36:D480–D484.
Article13. Sun S, Xuan F, Ge X, Fu H, Zhu J, Zhang S. Identification of differentially expressed genes in hepatopancreas of oriental river prawn, Macrobrachium nipponense exposed to environmental hypoxia. Gene. 2014; 534:298–306.14. Ziemkowski P, Felczak K, Poznański J, Kulikowski T, Zieliński Z, Cieś la J, Rode W. Interactions of 2′-fluoro-substituted dUMP analogues with thymidylate synthase. Biochem Biophys Res Commun. 2007; 362:37–43.
Article15. Wang JY. Biochemistry. Beijing: Higher Education Press;2002. 400 p.16. Sinigaglia F, Scheidegger D, Talmadge K, Garotta G. A sensitive and quantitative microassay for the detection of mycoplasma contamination: inhibition of IL-2 dependent cell line proliferation. J Immunol Methods. 1985; 76:85–92.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Verification of the Mycoplasma IST3 for urogenital mycoplasma culture in comparison to the Mycoplasma IST2
- A clinical study of mycoplasma pneumonia in children during recent 5 years
- Development of a rapid isolation and culture method of wild type Mycoplasma spp. in pigs
- Epidemiology of Mycoplasma pneumoniae Infection in Childhood
- Mycoplasma pneumoniae hepatitis in children