Intest Res.
2020 Jul;18(3):297-305. 10.5217/ir.2019.00105.
Hypermethylated promoters of tumor suppressor genes were identified in Crohn’s disease patients
- Affiliations
-
- 1Department of Internal Medicine, Inje University Haeundae Paik Hospital, Busan, Korea
- 2Department of Microbiology and Immunology, Inje University College of Medicine, Busan, Korea
- KMID: 2504585
- DOI: http://doi.org/10.5217/ir.2019.00105
Abstract
- Background/Aims
Overwhelming evidence suggests that inflammatory bowel disease (IBD) is caused by a complicated interplay between the multiple genes and abnormal epigenetic regulation in response to environmental factors. It is becoming apparent that epigenetic factors are significantly associated with the development of the disease. DNA methylation remains the most studied epigenetic modification, and hypermethylation of gene promoters is associated with gene silencing.
Methods
DNA methylation alterations may contribute to the many complex diseases development by regulating the interplay between external and internal environmental factors and gene transcriptional expression. In this study, we used 15 tumor suppressor genes (TSGs), originally identified in colon cancer, to detect promoter methylation in patients with Crohn’s disease (CD). Methylation specific polymerase chain reaction and bisulfite sequencing analyses were performed to assess methylation level of TSGs in CD patients.
Results
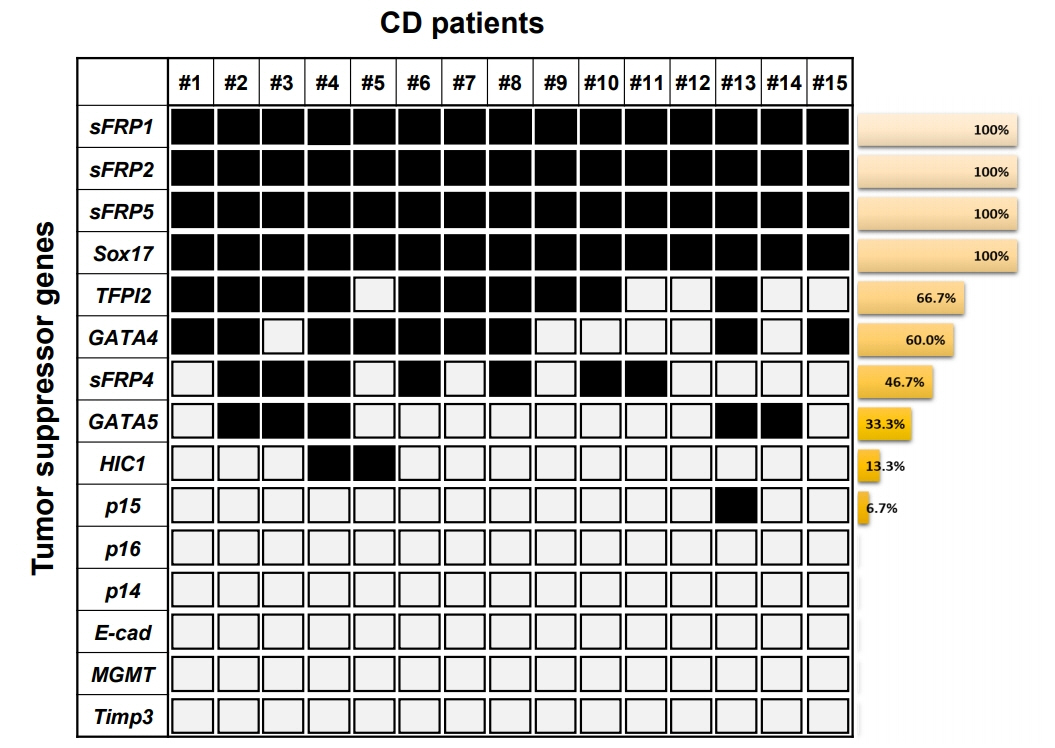
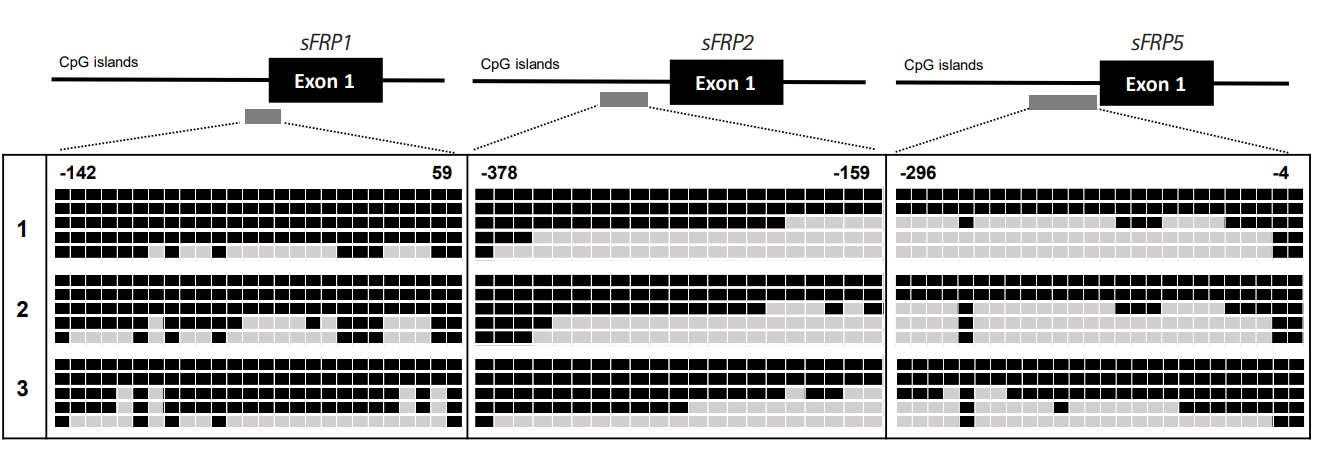
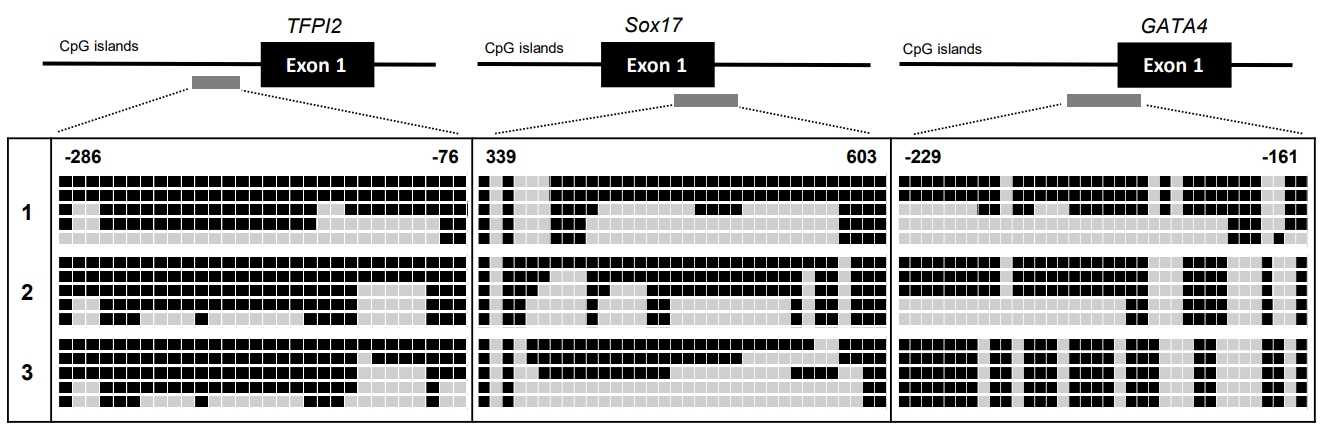
We found 6 TSGs (sFRP1, sFRP2, sFRP5, TFPI2, Sox17, and GATA4) are robustly hypermethylated in CD patient samples. Bisulfite sequencing analysis confirmed the methylation levels of the sFRP1, sFRP2, sFRP5, TFPI2, Sox17, and GATA4 promoters in the representative CD patient samples.
Conclusions
In this study, the promoter hypermethylation of the TSGs observed indicates that CD exhibits specific DNA methylation signatures with potential clinical applications for the noninvasive diagnosis of IBD and the prognosis for patients with IBD.
Figure
Reference
-
1. Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002; 347:417–429.
Article2. Rivas MA, Beaudoin M, Gardet A, et al. Deep resequencing of GWAS loci identifies independent rare variants associated with inflammatory bowel disease. Nat Genet. 2011; 43:1066–1073.3. Franke A, McGovern DP, Barrett JC, et al. Genome-wide metaanalysis increases to 71 the number of confirmed Crohn’s disease susceptibility loci. Nat Genet. 2010; 42:1118–1125.4. Liu JZ, van Sommeren S, Huang H, et al. Association analyses identify 38 susceptibility loci for inflammatory bowel disease and highlight shared genetic risk across populations. Nat Genet. 2015; 47:979–986.
Article5. Jones PA, Baylin SB. The epigenomics of cancer. Cell. 2007; 128:683–692.
Article6. Schuebel KE, Chen W, Cope L, et al. Comparing the DNA hypermethylome with gene mutations in human colorectal cancer. PLoS Genet. 2007; 3:1709–1723.
Article7. Esteller M. Epigenetic gene silencing in cancer: the DNA hypermethylome. Hum Mol Genet. 2007; 16. Spec No 1:R50-R59.
Article8. Shen L, Kondo Y, Guo Y, et al. Genome-wide profiling of DNA methylation reveals a class of normally methylated CpG island promoters. PLoS Genet. 2007; 3:2023–2036.
Article9. Robertson KD. DNA methylation and human disease. Nat Rev Genet. 2005; 6:597–610.
Article10. Maeda O, Ando T, Watanabe O, et al. DNA hypermethylation in colorectal neoplasms and inflammatory bowel disease: a mini review. Inflammopharmacology. 2006; 14:204–206.
Article11. Tahara T, Shibata T, Nakamura M, et al. Effect of MDR1 gene promoter methylation in patients with ulcerative colitis. Int J Mol Med. 2009; 23:521–527.
Article12. Heyn H, Esteller M. DNA methylation profiling in the clinic: applications and challenges. Nat Rev Genet. 2012; 13:679–692.
Article13. Kang K, Bae JH, Han K, Kim ES, Kim TO, Yi JM. A genomewide methylation approach identifies a new hypermethylated gene panel in ulcerative colitis. Int J Mol Sci. 2016; 17:E1291.
Article14. Kim TO, Park J, Kang MJ, et al. DNA hypermethylation of a selective gene panel as a risk marker for colon cancer in patients with ulcerative colitis. Int J Mol Med. 2013; 31:1255–1261.
Article15. Bae JH, Park J, Yang KM, Kim TO, Yi JM; IBD Study Group of Korean Association for Study of Intestinal Diseases (KASID). Detection of DNA hypermethylation in sera of patients with Crohn’s disease. Mol Med Rep. 2014; 9:725–729.
Article16. Hanauer SB. Inflammatory bowel disease: epidemiology, pathogenesis, and therapeutic opportunities. Inflamm Bowel Dis. 2006; 12–Suppl 1:S3-S9.
Article17. Bernstein CN, Blanchard JF, Kliewer E, Wajda A. Cancer risk in patients with inflammatory bowel disease: a populationbased study. Cancer. 2001; 91:854–862.
Article18. Bernstein CN, Blanchard JF, Rawsthorne P, Wajda A. Epidemiology of Crohn’s disease and ulcerative colitis in a central Canadian province: a population-based study. Am J Epidemiol. 1999; 149:916–924.
Article19. Colotta F, Allavena P, Sica A, Garlanda C, Mantovani A. Cancer-related inflammation, the seventh hallmark of cancer: links to genetic instability. Carcinogenesis. 2009; 30:1073–1081.
Article20. Rollin J, Iochmann S, Bléchet C, et al. Expression and methylation status of tissue factor pathway inhibitor-2 gene in nonsmall-cell lung cancer. Br J Cancer. 2005; 92:775–783.
Article21. Gerecke C, Scholtka B, Löwenstein Y, et al. Hypermethylation of ITGA4, TFPI2 and VIMENTIN promoters is increased in inflamed colon tissue: putative risk markers for colitis-associated cancer. J Cancer Res Clin Oncol. 2015; 141:2097–2107.
Article22. Glöckner SC, Dhir M, Yi JM, et al. Methylation of TFPI2 in stool DNA: a potential novel biomarker for the detection of colorectal cancer. Cancer Res. 2009; 69:4691–4699.23. Hibi K, Goto T, Kitamura YH, et al. Methylation of the TFPI2 gene is frequently detected in advanced gastric carcinoma. Anticancer Res. 2010; 30:4131–4133.24. Sun FK, Fan YC, Zhao J, et al. Detection of TFPI2 methylation in the serum of hepatocellular carcinoma patients. Dig Dis Sci. 2013; 58:1010–1015.
Article25. Kisiel JB, Yab TC, Taylor WR, et al. Stool DNA testing for the detection of pancreatic cancer: assessment of methylation marker candidates. Cancer. 2012; 118:2623–2631.
Article26. Dong Y, Tan Q, Tao L, et al. Hypermethylation of TFPI2 correlates with cervical cancer incidence in the Uygur and Han populations of Xinjiang, China. Int J Clin Exp Pathol. 2015; 8:1844–1854.27. Rasmussen SL, Krarup HB, Sunesen KG, Pedersen IS, Madsen PH, Thorlacius-Ussing O. Hypermethylated DNA as a biomarker for colorectal cancer: a systematic review. Colorectal Dis. 2016; 18:549–561.
Article28. Hibi K, Goto T, Shirahata A, et al. Detection of TFPI2 methylation in the serum of colorectal cancer patients. Cancer Lett. 2011; 311:96–100.
Article29. Sohn J, Natale J, Chew LJ, et al. Identification of Sox17 as a transcription factor that regulates oligodendrocyte development. J Neurosci. 2006; 26:9722–9735.
Article30. Matsui T, Kanai-Azuma M, Hara K, et al. Redundant roles of Sox17 and Sox18 in postnatal angiogenesis in mice. J Cell Sci. 2006; 119(Pt 17):3513–3526.31. Park KS, Wells JM, Zorn AM, Wert SE, Whitsett JA. Sox17 influences the differentiation of respiratory epithelial cells. Dev Biol. 2006; 294:192–202.
Article32. Kim I, Saunders TL, Morrison SJ. Sox17 dependence distinguishes the transcriptional regulation of fetal from adult hematopoietic stem cells. Cell. 2007; 130:470–483.
Article33. Hulbert A, Jusue-Torres I, Stark A, et al. Early detection of lung cancer using DNA promoter hypermethylation in plasma and sputum. Clin Cancer Res. 2017; 23:1998–2005.
Article34. Oishi Y, Watanabe Y, Yoshida Y, et al. Hypermethylation of Sox17 gene is useful as a molecular diagnostic application in early gastric cancer. Tumour Biol. 2012; 33:383–393.
Article35. Jia Y, Yang Y, Liu S, Herman JG, Lu F, Guo M. SOX17 antagonizes WNT/beta-catenin signaling pathway in hepatocellular carcinoma. Epigenetics. 2010; 5:743–749.
Article36. Fu DY, Wang ZM, et al. Sox17, the canonical Wnt antagonist, is epigenetically inactivated by promoter methylation in human breast cancer. Breast Cancer Res Treat. 2010; 119:601–612.
Article37. Zhang W, Glöckner SC, Guo M, et al. Epigenetic inactivation of the canonical Wnt antagonist SRY-box containing gene 17 in colorectal cancer. Cancer Res. 2008; 68:2764–2772.
Article38. Clevers H. Wnt/beta-catenin signaling in development and disease. Cell. 2006; 127:469–480.39. Gao X, Sedgwick T, Shi YB, Evans T. Distinct functions are implicated for the GATA-4, -5, and -6 transcription factors in the regulation of intestine epithelial cell differentiation. Mol Cell Biol. 1998; 18:2901–2911.
Article40. Akiyama Y, Watkins N, Suzuki H, et al. GATA-4 and GATA-5 transcription factor genes and potential downstream antitumor target genes are epigenetically silenced in colorectal and gastric cancer. Mol Cell Biol. 2003; 23:8429–8439.
Article41. Guo M, Akiyama Y, House MG, et al. Hypermethylation of the GATA genes in lung cancer. Clin Cancer Res. 2004; 10:7917–7924.
Article42. Guo M, House MG, Akiyama Y, et al. Hypermethylation of the GATA gene family in esophageal cancer. Int J Cancer. 2006; 119:2078–2083.43. Wakana K, Akiyama Y, Aso T, Yuasa Y. Involvement of GATA4/-5 transcription factors in ovarian carcinogenesis. Cancer Lett. 2006; 241:281–288.
Article44. Suzuki H, Watkins DN, Jair KW, et al. Epigenetic inactivation of SFRP genes allows constitutive WNT signaling in colorectal cancer. Nat Genet. 2004; 36:417–422.
Article45. Cheng YY, Yu J, Wong YP, et al. Frequent epigenetic inactivation of secreted frizzled-related protein 2 (SFRP2) by promoter methylation in human gastric cancer. Br J Cancer. 2007; 97:895–901.
Article46. Su HY, Lai HC, Lin YW, et al. Epigenetic silencing of SFRP5 is related to malignant phenotype and chemoresistance of ovarian cancer through Wnt signaling pathway. Int J Cancer. 2010; 127:555–567.
Article47. Fukui T, Kondo M, Ito G, et al. Transcriptional silencing of secreted frizzled related protein 1 (SFRP 1) by promoter hypermethylation in non-small-cell lung cancer. Oncogene. 2005; 24:6323–6327.
Article48. Nojima M, Suzuki H, Toyota M, et al. Frequent epigenetic inactivation of SFRP genes and constitutive activation of Wnt signaling in gastric cancer. Oncogene. 2007; 26:4699–4713.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Clinical Analysis of Methylation Status of Tumor Suppressor Genes in Breast Cancer
- Methylation Patterns of Tumor Suppressor Genes in Breast DCIS Tumors
- Cancer Metastasis and Metastasis Suppressors
- Identification of Novel Methylation Markers in Hepatocellular Carcinoma using a Methylation Array
- The Immunohistochemical Study of Oncogene and Tumor Suppressor Gene Proteins on Bone Tumor