Lab Med Online.
2020 Jul;10(3):197-201. 10.3343/lmo.2020.10.3.197.
Performance Evaluation of Serum IgD Quantification by the SPAPLUS Turbidimetric Analyzer and Determination of a Reference Interval of IgD in the Korean Population
- Affiliations
-
- 1Department of Laboratory Medicine and Genetics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Korea
- 2Seoul Clinical Laboratories, Seoul, Korea
- KMID: 2504128
- DOI: http://doi.org/10.3343/lmo.2020.10.3.197
Abstract
- Background
IgD is an immunoglobulin isotype that accounts for <1% of the total serum immunoglobulin, and IgD level changes can be observed in specific clinical situations. Herein, we evaluated the analytical performances of serum IgD quantification by the SPAPLUS turbidimetric analyzer (The Binding Site, UK), and determined a reference interval of IgD in the Korean population.
Methods
Precision, linearity and comparison with BNII (Siemens Healthineers, Germany) were assessed, and a reference interval was established according to the Clinical and Laboratory Standards Institute guidelines.
Results
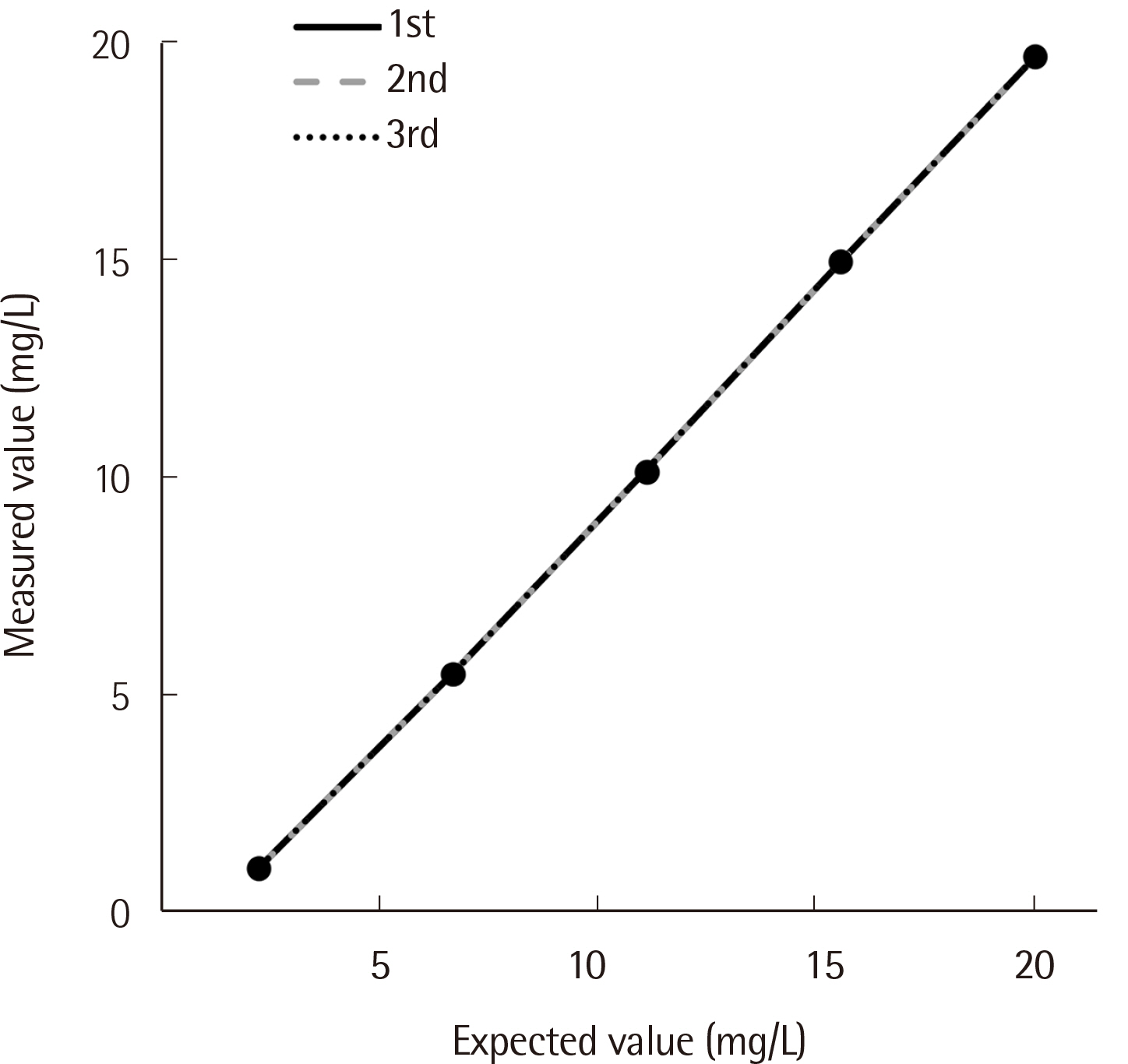
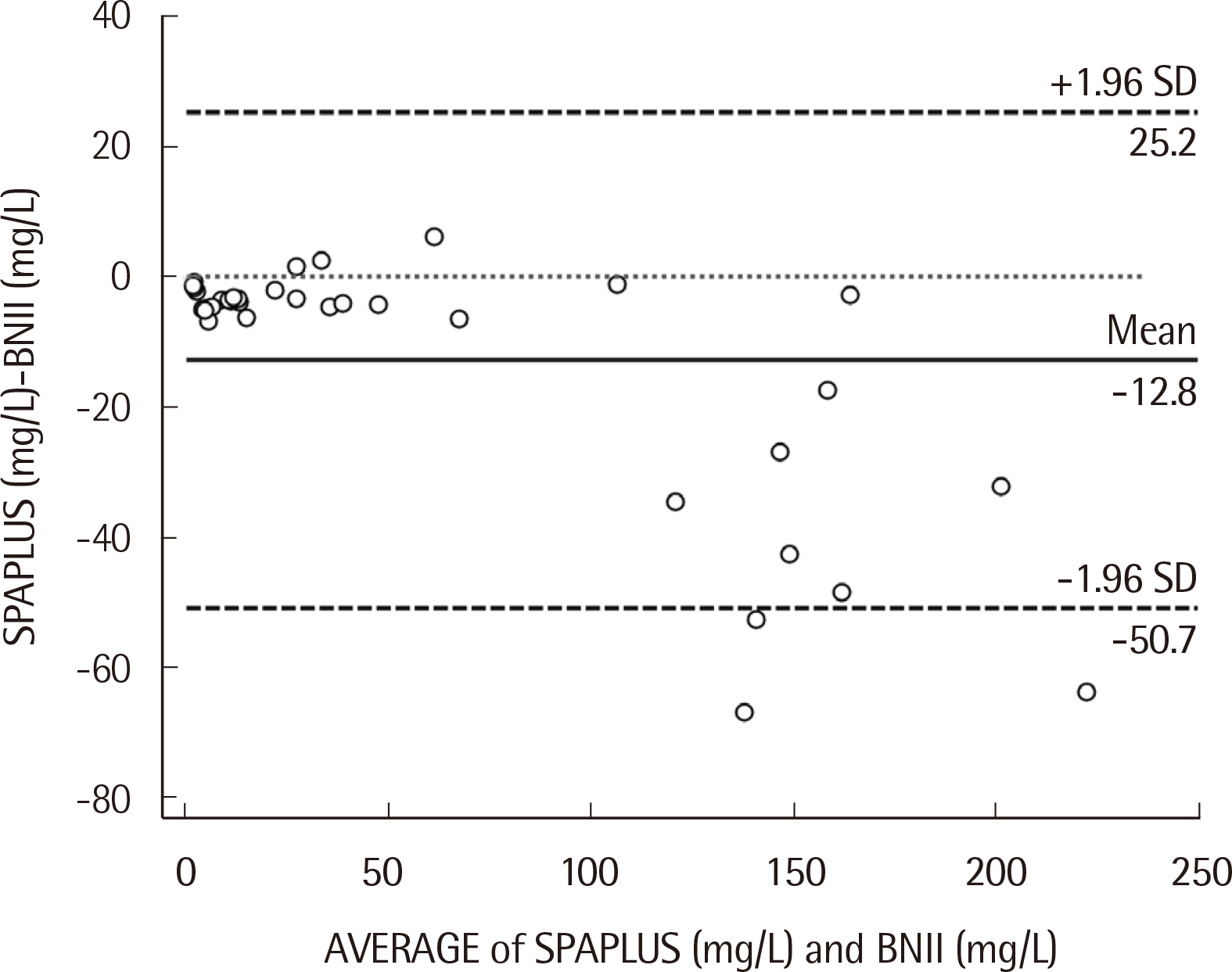
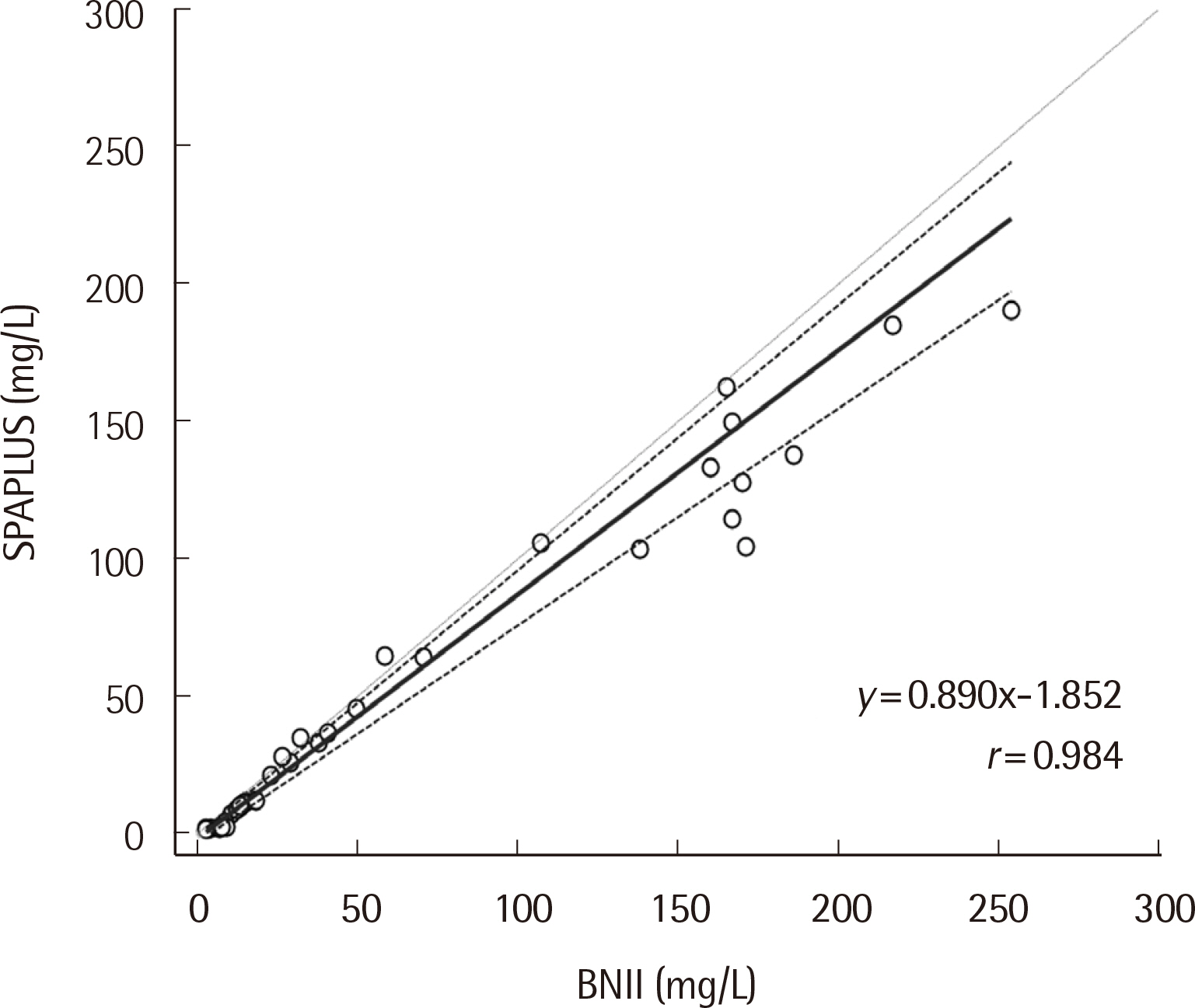
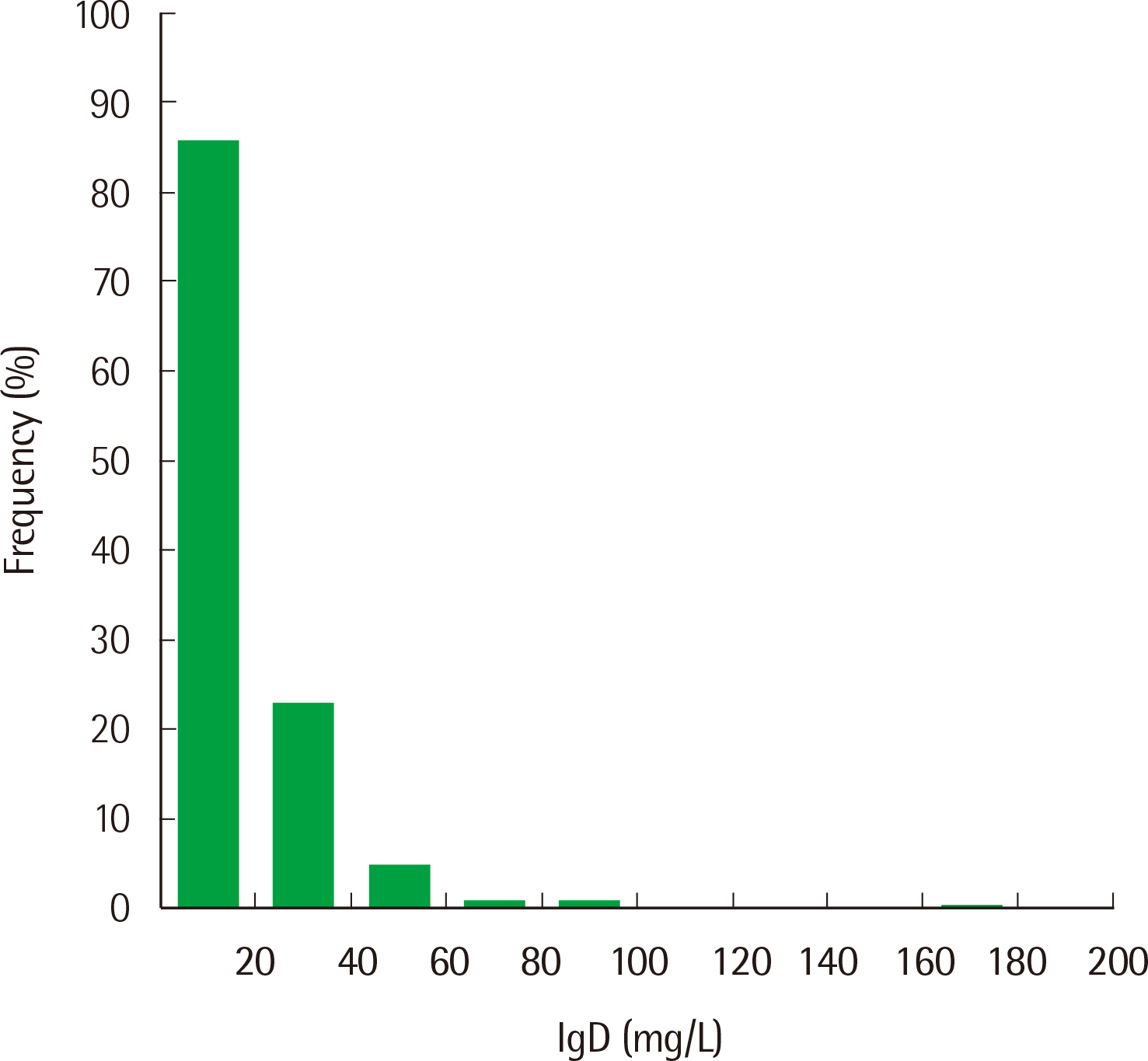
Repeatability ranged from 0.78% to 2.14%, and the within-laboratory precision ranged from 4.10% to 5.40% for three concentrations. The coefficient of determination (R2) was 0.9996 for the analytical measurement range. The IgD concentration showed a correlation between SPAPLUS and BNII (y=0.890x-1.852, r=0.984). However, IgD concentrations tended to be lower in SPAPLUS compared to those in BNII at concentrations >100 mg/L. The established upper limit of the reference interval was 53.6 mg/L.
Conclusions
The SPAPLUS shows good analytical performance for serum IgD quantification.
Figure
Reference
-
1. Rowe DS, Fahey JL. 1965; A new class of human immunoglobulins. I. A unique myeloma protein. J Exp Med. 121:171–84. DOI: 10.1084/jem.121.1.171. PMID: 14253482. PMCID: PMC2137973.2. Rigante D. 2016; The truth on IgD in the ploy of immune surveillance and inflammation. Immunol Res. 64:632–5. DOI: 10.1007/s12026-015-8744-z. PMID: 26603165.
Article3. van der Meer JW, Vossen JM, Radl J, van Nieuwkoop JA, Meyer CJ, Lobatto S, et al. 1984; Hyperimmunoglobulinaemia D and periodic fever: a new syndrome. Lancet. 1:1087–90. DOI: 10.1016/S0140-6736(84)92505-4. PMID: 6144826.
Article4. Pope RM, Keightley R, McDuffy S. 1982; Circulating autoantibodies to IgD in rheumatic diseases. J Immunol. 128:1860–3. PMID: 6977573.5. Pandey S, Kyle RA. 2013; Unusual myelomas: a review of IgD and IgE variants. Oncology (Williston Park). 27:798–803. PMID: 24133829.6. Bladé J, Lust JA, Kyle RA. 1994; Immunoglobulin D multiple myeloma: presenting features, response to therapy, and survival in a series of 53 cases. J Clin Oncol. 12:2398–404. DOI: 10.1200/JCO.1994.12.11.2398. PMID: 7964956.
Article7. Dunnette SL, Gleich GJ, Miller RD, Kyle RA. 1977; Measurement of IgD by a double antibody radioimmunoassay: demonstration of an apparent trimodal distribution of IgD levels in normal human sera. J Immunol. 119:1727–31. PMID: 915276.8. Rowe DS, Crabbé PA, Turner MW. 1968; Immunoglobulin D in serum, body fluids and lymphoid tissues. Clin Exp Immunol. 3:477–90. PMID: 4178229. PMCID: PMC1578945.9. Carballo I, Rabuñal N, Alvela L, Pérez LF, Vidal C, Alonso M, et al. 2017; Factors influencing serum concentrations of immunoglobulin D in the adult population: an observational study in Spain. Scand J Immunol. 85:272–9. DOI: 10.1111/sji.12529. PMID: 28128470.
Article10. Mosedale DE, Sandhu MS, Luan J, Goodall M, Grainger DJ. 2006; A new sensitive and specific enzyme-linked immunosorbent assay for IgD. J Immunol Methods. 313:74–80. DOI: 10.1016/j.jim.2006.03.012. PMID: 16714033.
Article11. Bahna SL, Heiner DC, Myhre BA. 1983; Changes in serum IgD in cigarette smokers. Clin Exp Immunol. 51:624–30. PMID: 6851250. PMCID: PMC1536788.12. Dunnette SL, Gleich GJ, Weinshilboum RM. 1978; Inheritance of low serum immunoglobulin D. J Clin Invest. 62:248–55. DOI: 10.1172/JCI109123. PMID: 678327. PMCID: PMC371760.
Article13. Gerrard JW, Heiner DC, Ko CG, Mink J, Meyers A, Dosman JA. 1980; Immunoglobulin levels in smokers and non-smokers. Ann Allergy. 44:261–2. PMID: 7377637.14. Clinical Laboratory Standards Institute. 2014; Evaluation of precision of quantitative measurement procedures; Approved guideline-Third edition. CLSI document EP05-A3. Clinical Laboratory Standards Institute;Wayne, PA:15. Clinical Laboratory Standards Institute. 2003; Evaluation of the linearity of quantitative measurement procedures: a statistical approach; Approved guideline. CLSI document EP06-A. Clinical Laboratory Standards Institute;Wayne, PA:16. Clinical Laboratory Standards Institute. 2013; Measurement procedure comparison and bias estimation using patient samples; Approved guideline-Third edition. CLSI document EP09-A3. Clinical Laboratory Standards Institute;Wayne, PA:17. Clinical Laboratory Standards Institute. 2010; De ning, establishing, and verifying reference intervals in the clinical laboratory; Approved guideline-Third edition. CLSI document EP28-A3C. Clinical Laboratory Standards Institute;Wayne, PA:18. Gerrard JW, Heiner DC, Ko CG, Mink JT, Myers A. 1981; IgD levels in white and Metis communities in Saskatchewan. Ann Allergy. 46:321–4. PMID: 7247082.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Relationship between Depression, Attention Deficit/Hyperactivity Disorder, and Internet Gaming Disorder Through Mediation Model
- The Structured Clinical Interview for DSM-5 Internet Gaming Disorder: Development and Validation for Diagnosing IGD in Adolescents
- Decreased Serum Glutamate Levels in Male Adults with Internet Gaming Disorder: A Pilot Study
- Effect of Parental Perception on the Prevalence of Adolescent Internet Gaming Disorder During the COVID-19 Pandemic
- Decreased Serum Cocaine- and Amphetamine-Regulated Transcript Level in Internet Gaming Disorder