Korean J Transplant.
2020 Mar;34(1):31-37. 10.4285/kjt.2020.34.1.31.
Long-term patency and complications of ringed polytetrafluoroethylene grafts used for middle hepatic vein reconstruction in living-donor liver transplantation
- Affiliations
-
- 1Division of Hepatobiliary Surgery and Liver Transplantation, Department of Surgery, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea
- 2Department of Surgery, Busan Paik Hospital, Inje University College of Medicine, Seoul, Korea
- KMID: 2503780
- DOI: http://doi.org/10.4285/kjt.2020.34.1.31
Abstract
- Background
Homologous vein allografts are adequate for reconstruction of the middle hepatic vein (MHV) in living-donor liver transplantation (LDLT). However, supply is a matter of concern. To replace homologous vein allografts, polytetrafluoroethylene (PTFE) grafts were used. This study aimed to assess the long-term patency rates and complications of PTFE grafts used for MHV reconstruction of LDLT in a high-volume liver transplantation center.
Methods
We analyzed the patency rates of PTFE-interposed MHV in 100 LDLT recipients and reviewed complications including PTFE graft migration.
Results
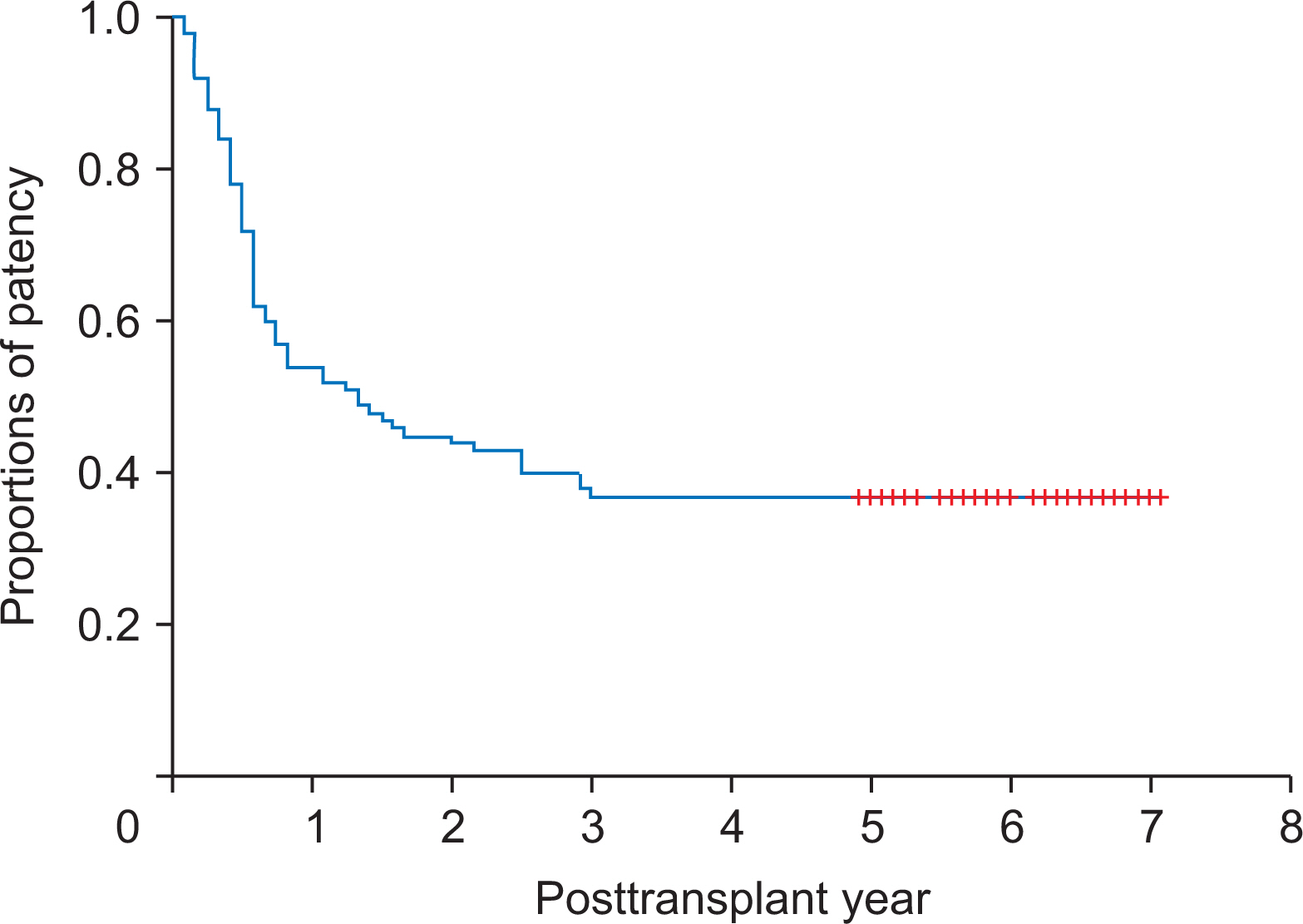
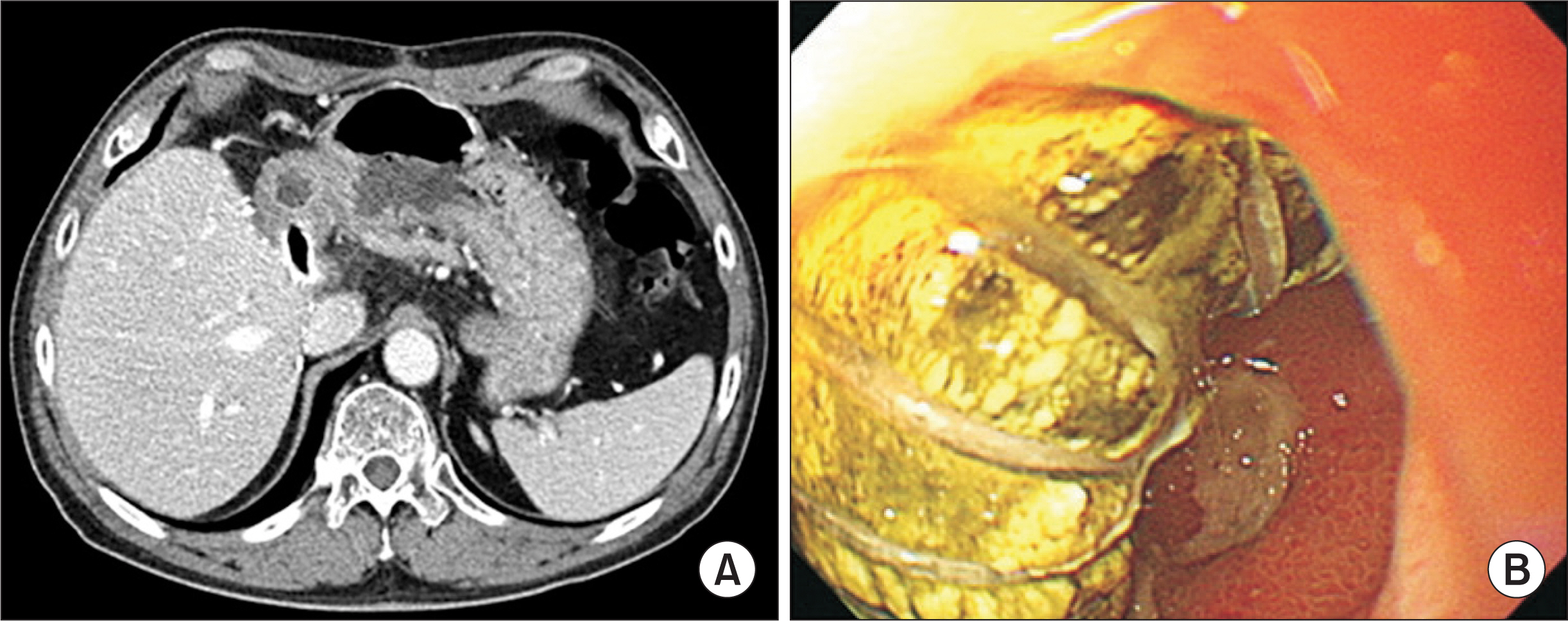
The mean age was 53.5±5.4 years and male to female ratio was 73:27. Primary diagnoses were hepatitis B virus infection (n=71) and other (n=28). Mean model for endstage liver disease score was 16.2±8.3. V5 reconstruction was performed as either single anastomosis (n=85) or double anastomoses (n=14). No V5 reconstruction was required in one patient. V8 reconstruction was performed as single anastomosis, double anastomoses, and no reconstruction in 75, 0, and 25 patients, respectively. During a mean follow-up of 6 years, three recipients required early MHV stenting within 2 weeks. After 3 months, there were no episodes of congestion-associated infarct, regardless of MHV patency. Patency rates of PTFE-interposed MHV were 54.0%, 37.0%, and 37.0% at 1, 3, and 5 years, respectively. Unwanted PTFE graft migration occurred in two recipients, and the actual incidence was 2% at 5 years.
Conclusions
PTFE grafts combined with small-artery patches demonstrated acceptably high short- and long-term patency rates. Since the risk of unwanted migration of PTFE graft is not negligibly low, lifelong surveillance is necessary to detect unexpected rare complications.
Figure
Reference
-
Hwang S., Lee SG., Lee YJ., Sung KB., Park KM., Kim KH, et al. 2006. Lessons learned from 1,000 living donor liver transplantations in a single center: how to make living donations safe. Liver Transpl. 12:920–7. DOI: 10.1002/lt.20734. PMID: 16721780.
ArticleHwang S., Lee SG., Ahn CS., Park KM., Kim KH., Moon DB, et al. 2005. Cryopreserved iliac artery is indispensable interposition graft material for middle hepatic vein reconstruction of right liver grafts. Liver Transpl. 11:644–9. DOI: 10.1002/lt.20430. PMID: 15915499.
ArticleSugawara Y., Makuuchi M., Akamatsu N., Kishi Y., Niiya T., Kaneko J, et al. 2004. Refinement of venous reconstruction using cryopreserved veins in right liver grafts. Liver Transpl. 10:541–7. DOI: 10.1002/lt.20129. PMID: 15048798.
ArticleHwang S., Lee SG., Song GW., Lee HJ., Park JI., Ryu JH. 2007. Use of endarterectomized atherosclerotic artery allograft for hepatic vein reconstruction of living donor right lobe graft. Liver Transpl. 13:306–8. DOI: 10.1002/lt.21045. PMID: 17256786.
ArticleHwang S., Jung DH., Ha TY., Ahn CS., Moon DB., Kim KH, et al. 2012. Usability of ringed polytetrafluoroethylene grafts for middle hepatic vein reconstruction during living donor liver transplantation. Liver Transpl. 18:955–65. DOI: 10.1002/lt.23456. PMID: 22511404.
ArticleHa TY., Hwang S., Jung DH., Ahn CS., Kim KH., Moon DB, et al. 2014. Complications analysis of polytetrafluoroethylene grafts used for middle hepatic vein reconstruction in living-donor liver transplantation. Transplant Proc. 46:845–9. DOI: 10.1016/j.transproceed.2013.10.054. PMID: 24767363.
ArticleHsu SC., Thorat A., Yang HR., Poon KS., Li PC., Yeh CC, et al. 2017. Assessing the safety of expanded polytetrafluoroethylene synthetic grafts in living donor liver transplantation: graft migration into hollow viscous organs. Diagnosis and treatment options. Med Sci Monit. 23:3284–92. DOI: 10.12659/MSM.902636. PMID: 28683053. PMCID: PMC5510995.Koc C., Akbulut S., Ozdemir F., Kose A., Isik B., Yologlu S, et al. 2019. Analysis of risk factors affecting the development of infection in artificial vascular grafts used for reconstruction of middle hepatic vein tributaries in living donor liver transplantation. Transplantation. 103:1871–6. DOI: 10.1097/TP.0000000000002583. PMID: 30747841.
ArticleChung MH., Chuang CC., Liaw LF., Chen CY., Chen IM., Hsu CP, et al. 2018. Thrombotic ringed polytetrafluoroethylene graft with infection after living-donor liver transplantation. Transplant Proc. 50:2606–10. DOI: 10.1016/j.transproceed.2018.04.050. PMID: 30401360.
ArticleGoja S., Yadav SK., Roy R., Soin AS. 2018. A retrospective comparative study of venous vs nonringed expanded polytetrafluoroethylene etension grafts for anterior sector outflow reconstruction in right lobe living donor liver transplantation. Clin Transplant. 32:e13344. DOI: 10.1111/ctr.13344. PMID: 29981524.Thorat A., Hsu SC., Yang HR., Li PC., Li ML., Yeh CC, et al. 2016. Reconstruction of isolated inferior right hepatic vein(s) in right lobe living donor liver transplantation using polytetrafluoroethylene grafts: a new feasible concept, technique of ¡®bridging conduit venoplasty¡¯ and outcomes. Ann Transplant. 21:735–44. DOI: 10.12659/AOT.900871. PMID: 27909288.
ArticleKo GY., Sung KB., Yoon HK., Kim JH., Song HY., Seo TS, et al. 2002. Endovascular treatment of hepatic venous outflow obstruction after living-donor liver transplantation. J Vasc Interv Radiol. 13:591–9. DOI: 10.1016/S1051-0443(07)61652-2. PMID: 12050299.
ArticleKo GY., Sung KB., Yoon HK., Kim KR., Kim JH., Gwon DI, et al. 2008. Early posttransplant hepatic venous outflow obstruction: long-term efficacy of primary stent placement. Liver Transpl. 14:1505–11. DOI: 10.1002/lt.21560. PMID: 18825710.
ArticleJung DH., Lee SG., Hwang S., Hong HN. 2009. Feasibility assessment of interposition vessel graft substitutes in dog models for later clinical application to middle hepatic vein reconstruction during living donor liver transplantation. J Korean Surg Soc. 77:15–28. DOI: 10.4174/jkss.2009.77.1.15.
ArticlePapanicolaou G., Beach KW., Zierler RE., Detmer PR., Strandness DE Jr Jr. 1995. Hemodynamics of stenotic infrainguinal vein grafts: theoretic considerations. Ann Vasc Surg. 9:163–71. DOI: 10.1007/BF02139659. PMID: 7786702.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Patency of middle hepatic vein reconstruction using Hemashield grafts compared with ringed polytetrafluoroethylene grafts in living donor liver transplantation
- A critical complication of the interposition grafts using a cryopreserved aortic allograft for middle hepatic vein reconstruction in living donor liver transplantation
- Human dermis as a new substitute for middle hepatic vein during living donor liver transplantation: early results from ongoing clinical trial
- Patency of Hemashield grafts versus ringed Gore-Tex grafts in middle hepatic vein reconstruction for living donor liver transplantation
- Migration of synthetic vascular graft used for middle hepatic vein reconstruction during living donor liver transplantation using a modified right liver graft: A collective review of worldwide cases