J Korean Med Sci.
2020 Jul;35(26):e246. 10.3346/jkms.2020.35.e246.
Screening Clinic for Coronavirus Disease 2019 to Prevent Intrahospital Spread in Daegu, Korea: a Single-Center Report
- Affiliations
-
- 1Division of Pulmonology, Department of Internal Medicine, Keimyung University Dongsan Hospital, Keimyung University School of Medicine, Daegu, Korea
- 2Division of Infectious Disease, Department of Internal Medicine, Keimyung University Dongsan Hospital, Keimyung University School of Medicine, Daegu, Korea
- KMID: 2503693
- DOI: http://doi.org/10.3346/jkms.2020.35.e246
Abstract
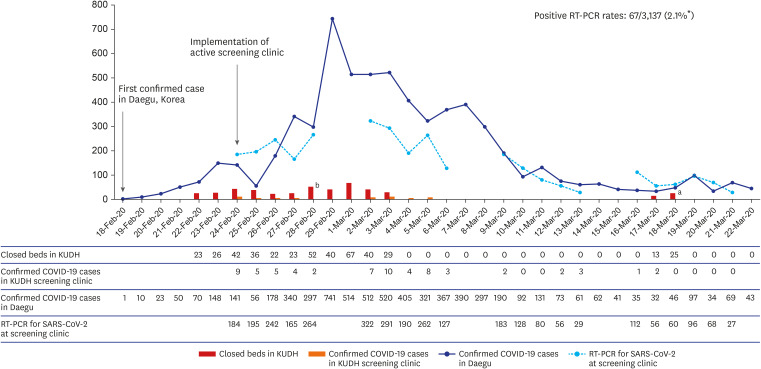
- There is still a paucity of studies on real-world outcome of screening clinic for hospital protection from coronavirus disease 2019 (COVID-19). As the number of COVID-19 cases was growing rapidly in Daegu, Korea, we started operating an active screening clinic outside the hospital premises. Over two weeks, 2,087 patients were screened using real-time reverse transcriptase polymerase chain reaction testing for severe acute respiratory syndrome coronavirus 2, with 42 confirmed cases. Before the screening clinic period, an average of 36 beds (maximum 67 beds) per day were closed due to unrecognized COVID-19 patients entering the hospital. In contrast, after the screening clinic operated well, only one event of closing emergency room (25 beds) occurred due to a confirmed COVID-19 case of asymptomatic patient. We report the operational process of screening clinic for COVID-19 and its effectiveness in maintaining the function of tertiary hospitals.
Figure
Cited by 2 articles
-
Clinical Pathway for Emergency Brain Surgery during COVID-19 Pandemic and Its Impact on Clinical Outcomes
Shin-heon Lee, Ju-sung Jang, Jin-Won Chung, Jeong-taik Kwon, Yong-sook Park
J Korean Med Sci. 2020;36(2):e16. doi: 10.3346/jkms.2021.36.e16.Analysis of a COVID-19 Prescreening Process in an Outpatient Clinic at a University Hospital during the COVID-19 Pandemic
Ui Yoon Choi, Seung Eun Jung, Mi Sook Kim, Hyang Sook Oh, Young Mi Kwon, Jehoon Lee, Jung-Hyun Choi
J Korean Med Sci. 2021;36(42):e295. doi: 10.3346/jkms.2021.36.e295.
Reference
-
1. Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020; 382(8):727–733. PMID: 31978945.
Article2. Zhou P, Yang XL, Wang XG, Hu B, Zhang L, Zhang W, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020; 579(7798):270–273. PMID: 32015507.3. Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020; 382(18):1708–1720. PMID: 32109013.4. Korea Centers for Disease Control & Prevention. The updates on COVID-19 in Korea as of 17 April. Updated 2020. Accessed April 18, 2020. https://www.cdc.go.kr/board/board.es?mid=a20501000000&bid=0015&list_no=366942&act=view.5. Korea Centers for Disease Control & Prevention. Guidelines for the operation of medical institutions with COVID0-19 patients. Updated 2020. Accessed June 8, 2020. http://ncov.mohw.go.kr/shBoardView.do?brdId=2&brdGubun=24&ncvContSeq=1181.6. Centers for Disease Control and Prevention. Interim infection prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID-19) in healthcare settings. Updated 2020. Accessed March 31, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html.7. Centers for Disease Control and Prevention. Interim guidelines for collecting, handling, and testing clinical specimens from persons for coronavirus disease 2019 (COVID-19). Updated 2020. Accessed March 31, 2020. https://www.cdc.gov/coronavirus/2019-ncov/lab/guidelines-clinical-specimens.html.8. Centers for Disease Control and Prevention. Interim U.S. guidance for risk assessment and public health management of healthcare personnel with potential exposure in a healthcare setting to patients with coronavirus disease (COVID-19). Updated 2020. Accessed March 31, 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/guidance-risk-assesment-hcp.html.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The analysis of the status of emergency department-based screening clinic according to the spread of coronavirus disease 2019: a retrospective single-center study
- Prevention of exposure to and spread of COVID-19 using air purifiers: challenges and concerns
- Yeungnam University type drive-through (YU-Thru) coronavirus disease 2019 (COVID-19) screening system: a rapid and safe screening system
- COVID-19 and Smell Loss
- Clinical and Epidemiological Characteristics of Coronavirus Disease 2019 in the Early Stage of Outbreak