Anat Cell Biol.
2020 Jun;53(2):201-215. 10.5115/acb.19.236.
Cytoprotective and antioxidant effects of aged garlic extract against adriamycin-induced cardiotoxicity in adult male rats
- Affiliations
-
- 1Department of Anatomy, Faculty of Medicine, King Abdulaziz University, Jeddah, Saudi Arabia
- 2Department of Anatomy, Faculty of Medicine, Zagazig University, Zagazig, Egypt
- KMID: 2503450
- DOI: http://doi.org/10.5115/acb.19.236
Abstract
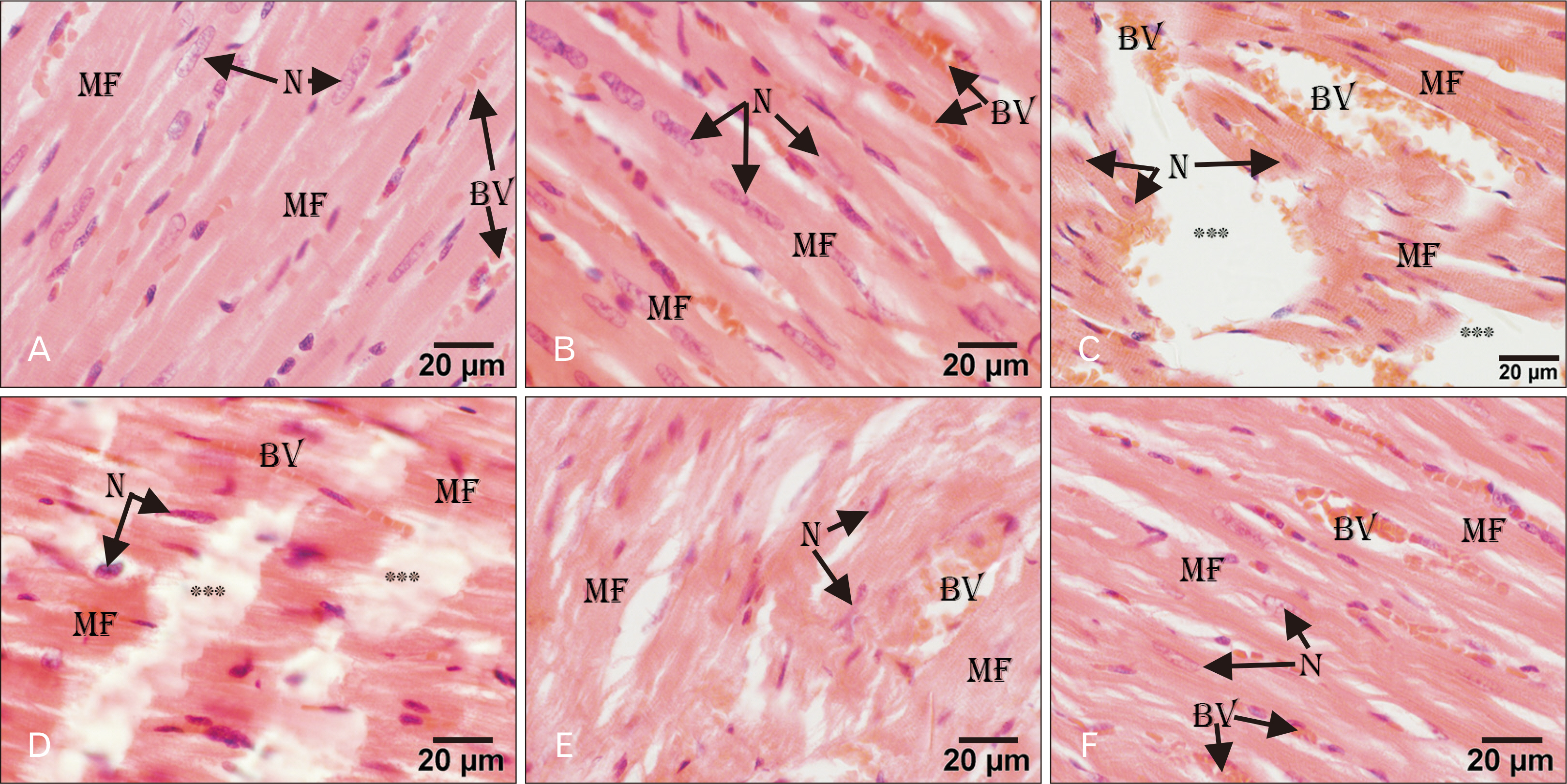
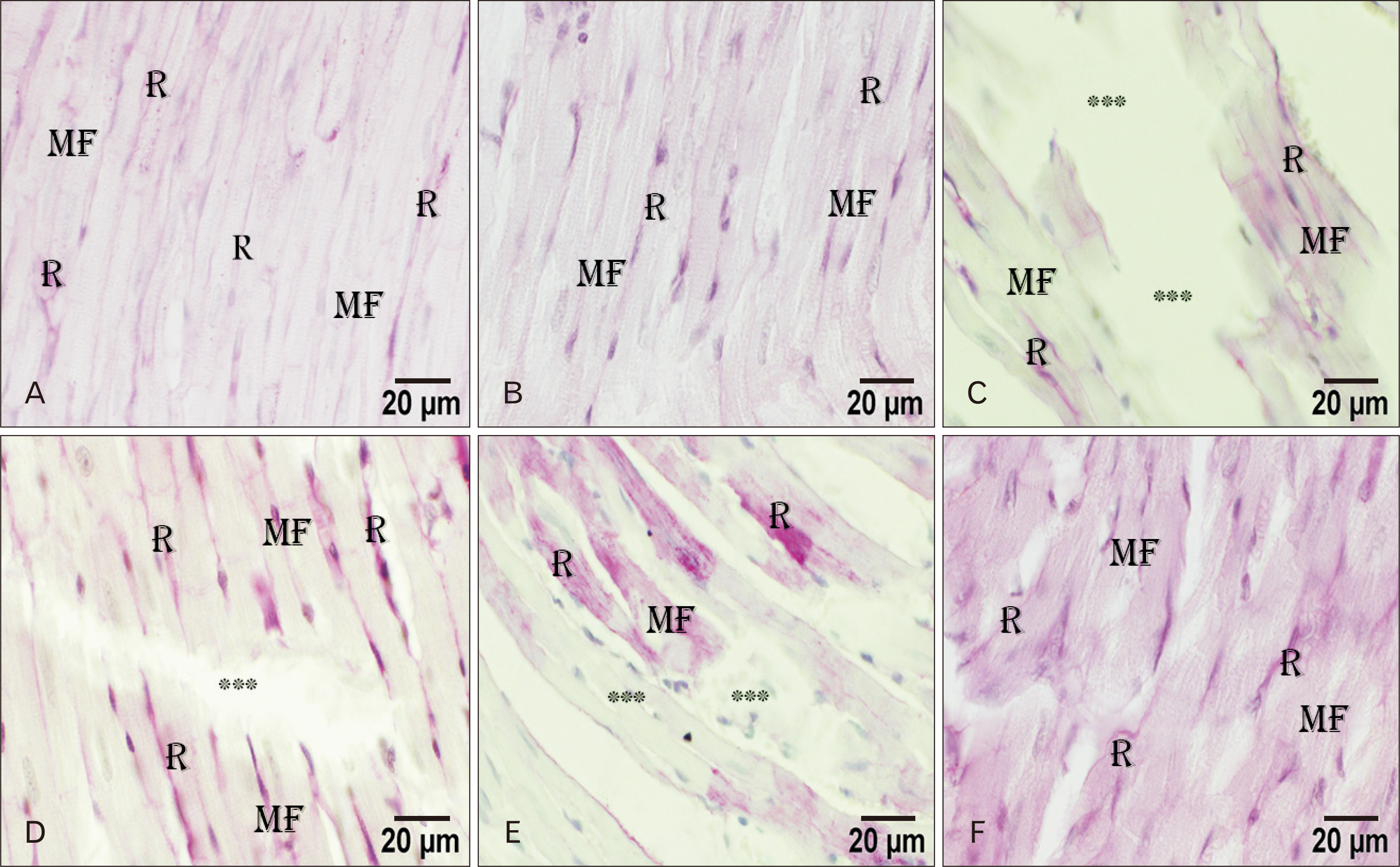
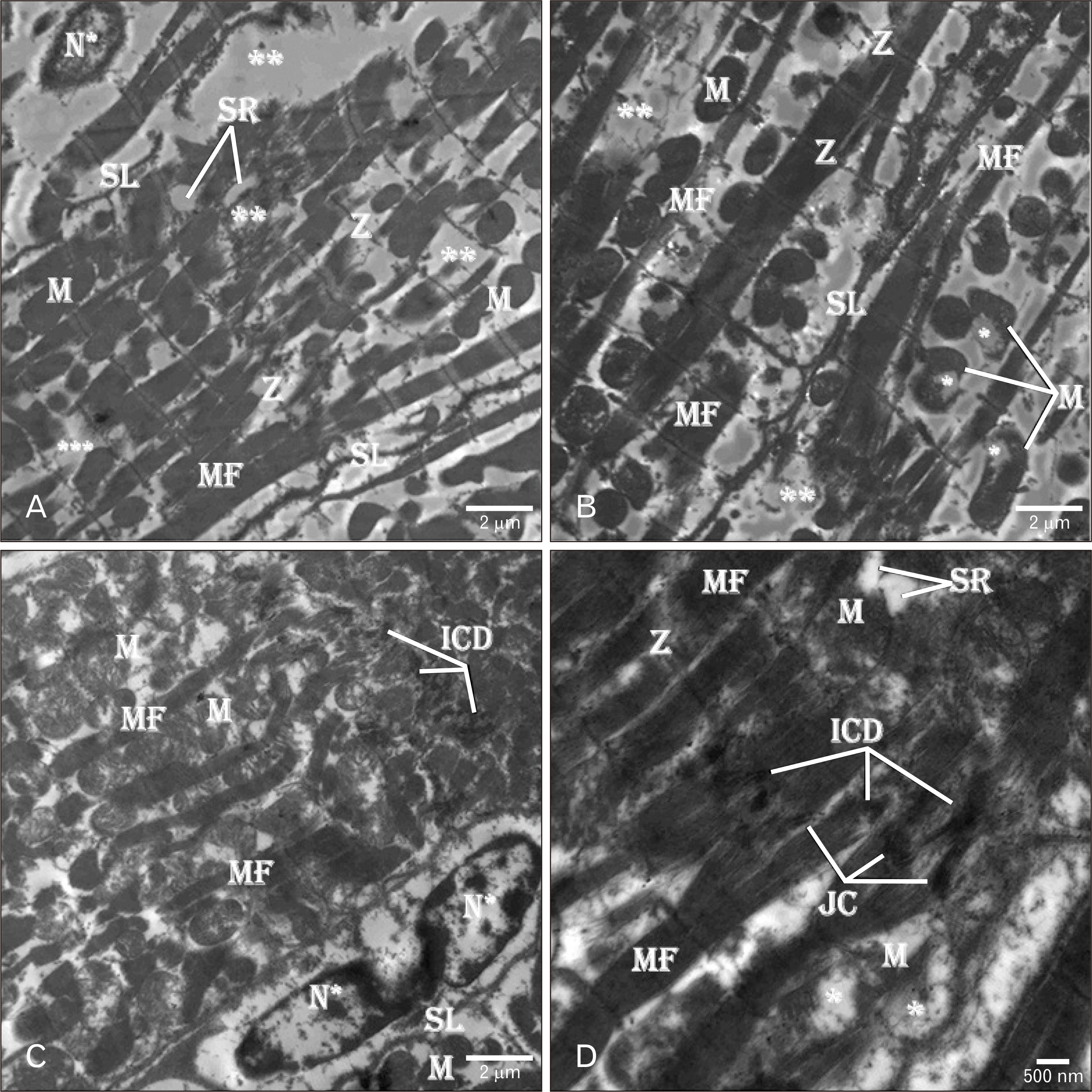
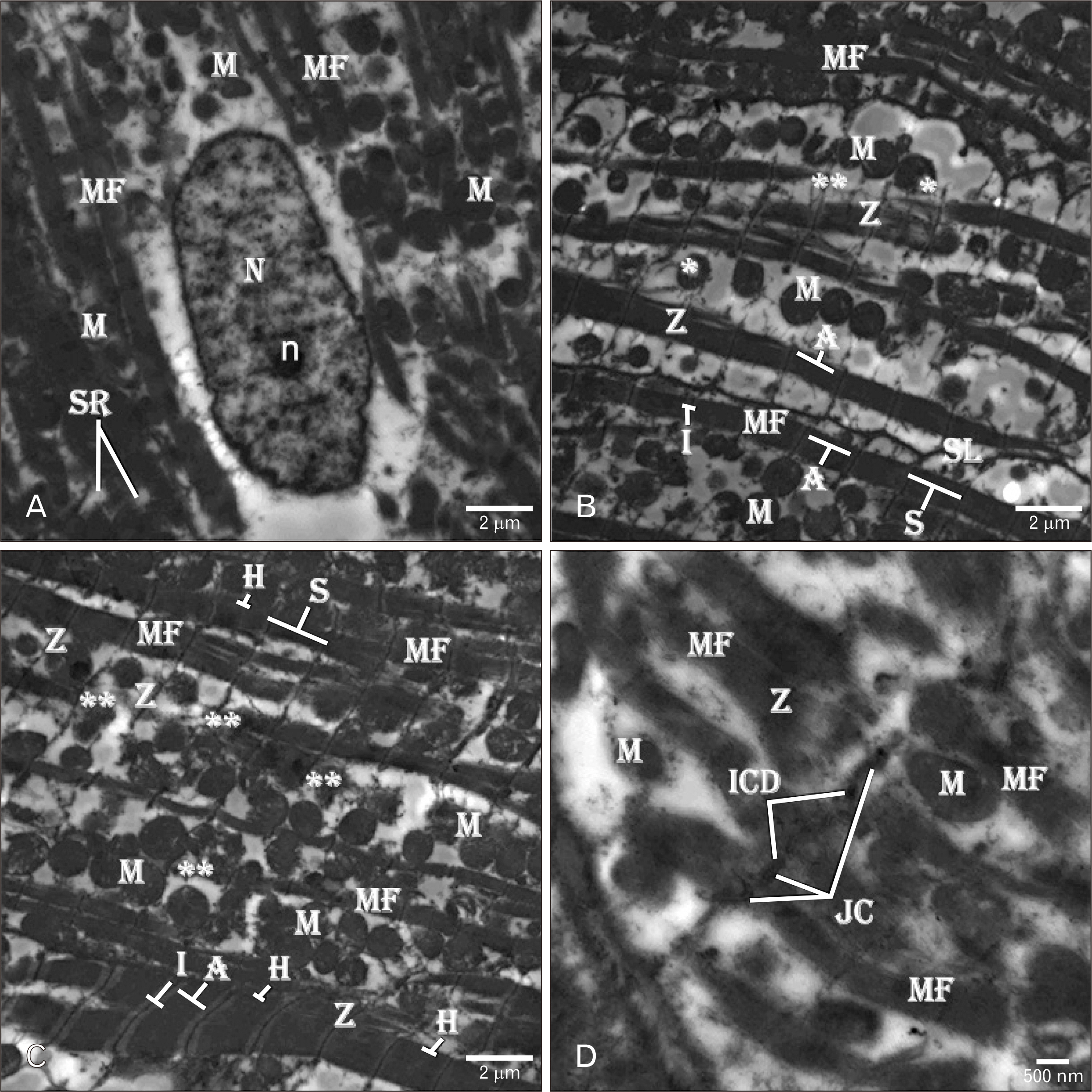
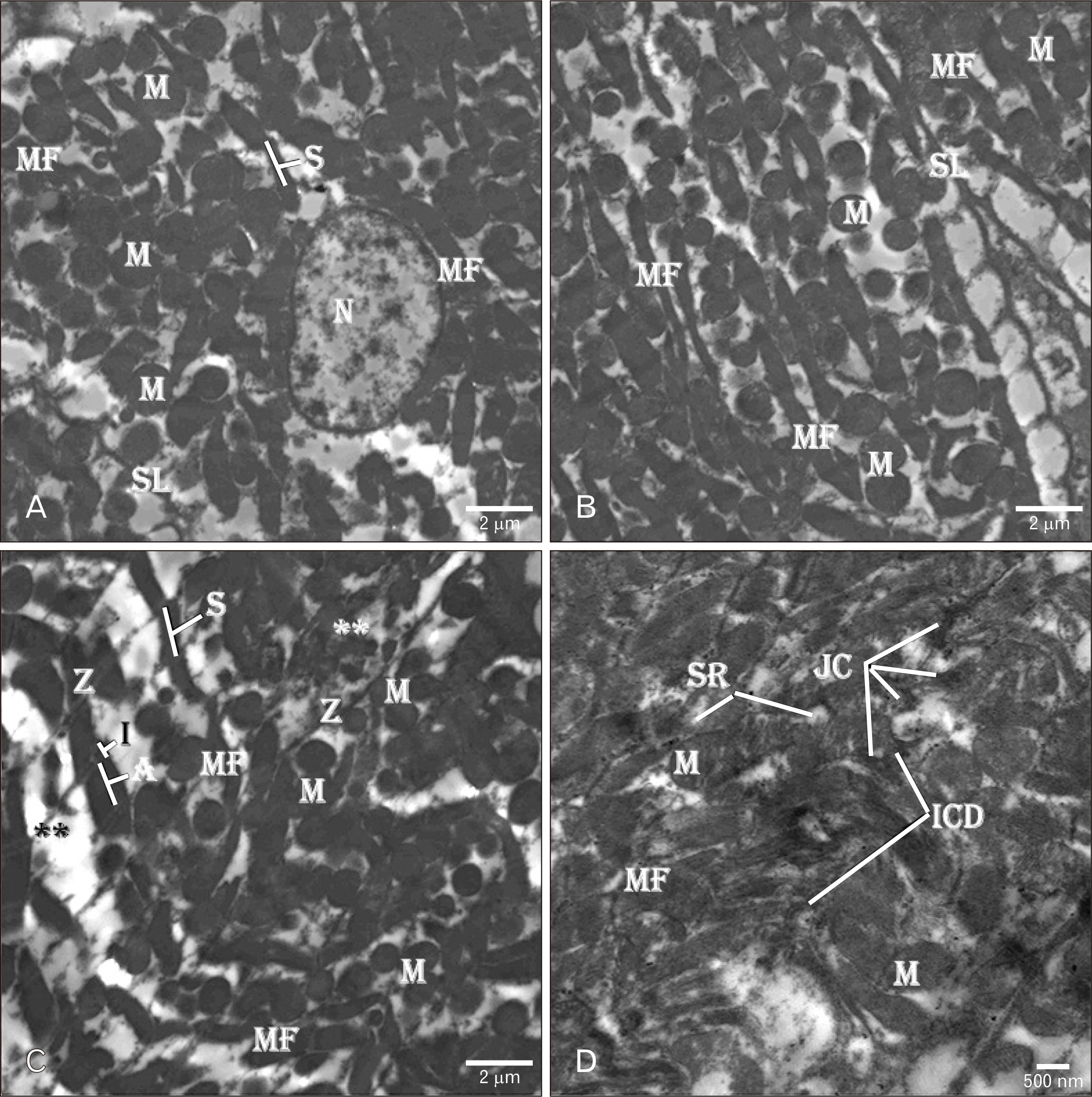
- Adriamycin (ADR) efficacy in cancer chemotherapy is well-established. However, ADR-induced cardiotoxicity remains a significant challenge. Aged garlic extract (AGE) is a natural polyphenol with high antioxidant potential. This study was planned to determine the cytoprotective and antioxidant actions of AGE against the cardiotoxic effect of ADR in rats. Six equal groups, control, ADR-treated (single dose of 10 mg/kg on day 8); AGE-treated (one dose of 250 mg/kg for 14 days); AGE plus ADR-treated (one dose of 250 mg/kg AGE for one week plus ADR injection of 10 mg/kg on day 8); ADR plus AGE-treated (single ADR injection of 10 mg/kg on day 8 plus AGE of 250 mg/kg once from 8th to 14th day); combined AGE plus ADR plus AGE-treated (one dose of 250 mg/kg AGE for 14 days plus single ADR injection of 10 mg/kg on day 8). Sera and cardiac samples were collected on day 15 and prepared for histological, ultrastructural and biochemical study. Disorganization, focal degeneration and necrosis with apoptotic changes of the cardiac myofibrils were observed in ADR-treated rats. Also, reduction in level of total creatine kinase, lactic dehydrogenase, alkaline phosphatase enzymes, glutathione, glutathione- peroxidase, superoxide dismutase, and catalase activities and elevation in malondialdehyde concentration were detected in ADR-treated rats. However, combination of AGE attenuated most of the histopathological, ultrastructural, and biochemical changes induced by ADR. Combination of AGE attenuated the cardiotoxic effects-induced by ADR through its antioxidant and cytoprotective potentials. Therefore, AGE can use as adjunct during administration of ADR in cancer therapy.
Keyword
Figure
Reference
-
1. Songbo M, Lang H, Xinyong C, Bin X, Ping Z, Liang S. 2019; Oxidative stress injury in doxorubicin-induced cardiotoxicity. Toxicol Lett. 307:41–8. DOI: 10.1016/j.toxlet.2019.02.013. PMID: 30817977.
Article2. Renu K, V G A, P B TP, Arunachalam S. 2018; Molecular mechanism of doxorubicin-induced cardiomyopathy - an update. Eur J Pharmacol. 818:241–53. DOI: 10.1016/j.ejphar.2017.10.043. PMID: 29074412.
Article3. Tham YK, Bernardo BC, Ooi JY, Weeks KL, McMullen JR. 2015; Pathophysiology of cardiac hypertrophy and heart failure: signaling pathways and novel therapeutic targets. Arch Toxicol. 89:1401–38. DOI: 10.1007/s00204-015-1477-x. PMID: 25708889.
Article4. Yu J, Wang C, Kong Q, Wu X, Lu JJ, Chen X. 2018; Recent progress in doxorubicin-induced cardiotoxicity and protective potential of natural products. Phytomedicine. 40:125–39. DOI: 10.1016/j.phymed.2018.01.009. PMID: 29496165.
Article5. Cheung KG, Cole LK, Xiang B, Chen K, Ma X, Myal Y, Hatch GM, Tong Q, Dolinsky VW. 2015; Sirtuin-3 (SIRT3) protein attenuates doxorubicin-induced oxidative stress and improves mitochondrial respiration in H9c2 cardiomyocytes. J Biol Chem. 290:10981–93. DOI: 10.1074/jbc.M114.607960. PMID: 25759382. PMCID: PMC4409259.
Article6. Shaker RA, Abboud SH, Assad HC, Hadi N. 2018; Enoxaparin attenuates doxorubicin induced cardiotoxicity in rats via interfering with oxidative stress, inflammation and apoptosis. BMC Pharmacol Toxicol. 19:3. DOI: 10.1186/s40360-017-0184-z. PMID: 29321061. PMCID: PMC5763526.
Article7. Octavia Y, Kararigas G, de Boer M, Chrifi I, Kietadisorn R, Swinnen M, Duimel H, Verheyen FK, Brandt MM, Fliegner D, Cheng C, Janssens S, Duncker DJ, Moens AL. 2017; Folic acid reduces doxorubicin-induced cardiomyopathy by modulating endothelial nitric oxide synthase. J Cell Mol Med. 21:3277–87. DOI: 10.1111/jcmm.13231. PMID: 28608983. PMCID: PMC5706529.
Article8. De Angelis A, urbanek K, Cappetta D, Piegari E, Ciuffreda LP, Rivellino A, Russo R, Esposito G, Rossi F, Berrino L. 2016; Doxorubicin cardiotoxicity and target cells: a broader perspective. Cardio-Oncology. 2:2. DOI: 10.1186/s40959-016-0012-4.
Article9. Lam W, Jiang Z, Guan F, Huang X, Hu R, Wang J, Bussom S, Liu SH, Zhao H, Yen Y, Cheng YC. 2015; PHY906(KD018), an adjuvant based on a 1800-year-old Chinese medicine, enhanced the anti-tumor activity of Sorafenib by changing the tumor microenvironment. Sci Rep. 5:9384. DOI: 10.1038/srep09384. PMID: 25819872. PMCID: PMC4377583.
Article10. Moutia M, Habti N, Badou A. 2018; In vitro and in vivo immunomodulator activities of allium sativum L. Evid Based Complementary Altern Med. 2018:4984659. DOI: 10.1155/2018/4984659. PMID: 30008785. PMCID: PMC6020507.
Article11. Arreola R, Quintero-Fabián S, López-Roa RI, Flores-Gutiérrez EO, Reyes-Grajeda JP, Carrera-Quintanar L, Ortuño-Sahagún D. 2015; Immunomodulation and anti-inflammatory effects of garlic compounds. J Immunol Res. 2015:401630. DOI: 10.1155/2015/401630. PMID: 25961060. PMCID: PMC4417560.
Article12. Wang X, Zhang M, Yang Y. 2019; The vivo antioxidant activity of self-made aged garlic extract on the d-galactose-induced mice and its mechanism research via gene chip analysis. RSC Adv. 9:3669–78. DOI: 10.1039/C8RA10308A.
Article13. Jeong YY, Ryu JH, Shin JH, Kang MJ, Kang JR, Han J, Kang D. 2016; Comparison of anti-oxidant and anti-inflammatory effects between fresh and aged black garlic extracts. Molecules. 21:430. DOI: 10.3390/molecules21040430. PMID: 27043510. PMCID: PMC6274159.
Article14. Nasr AY, Saleh HA. 2014; Aged garlic extract protects against oxidative stress and renal changes in cisplatin-treated adult male rats. Cancer Cell Int. 14:92. DOI: 10.1186/s12935-014-0092-x. PMID: 25298749. PMCID: PMC4189163.
Article15. Nasr AY. 2017; The impact of aged garlic extract on adriamycin-induced testicular changes in adult male Wistar rats. Acta Histochem. 119:648–62. DOI: 10.1016/j.acthis.2017.07.006. PMID: 28784287.
Article16. Zhang J, Cui L, Han X, Zhang Y, Zhang X, Chu X, Zhang F, Zhang Y, Chu L. 2017; Protective effects of tannic acid on acute doxorubicin-induced cardiotoxicity: Involvement of suppression in oxidative stress, inflammation, and apoptosis. Biomed Pharmacother. 93:1253–60. DOI: 10.1016/j.biopha.2017.07.051. PMID: 28738542.
Article17. Bancroft JD, Layton C. Bancroft JD, Layton C, Suvarna KS, editors. 2012. The hematoxylins and eosin. Theory and practice of histological techniques. 7th ed. Elsevier;Philadelphia: p. 172–214.
Article18. Hayat MA. Hayat MA, editor. 2000. Chemical Fixation. Principles and techniques of electron microscopy: biological applications. 4th ed. Cambridge University Press;Cambridge: p. 4–85.
Article19. Buhl SN, Jackson KY. 1978; Optimal conditions and comparison of lactate dehydrogenase catalysis of the lactate-to-pyruvate and pyruvate-to-lactate reactions in human serum at 25, 30, and 37 degrees C. Clin Chem. 24:828–31. DOI: 10.1093/clinchem/24.5.828.
Article20. Tietz NW, Rinker AD, Shaw LM. 1983; International Federation of Clinical Chemistry. IFCC methods for the measurement of catalytic concentration of enzymes. Part 5. IFCC method for alkaline phosphatase (orthophosphoric-monoester phosphohydrolase, alkaline optimum, EC 3.1.3.1). IFCC document stage 2, draft 1, 1983-03 with a view to an IFCC recommendation. Clin Chim Acta. 135:339F–67F.21. Hørder M, Elser RC, Gerhardt W, Mathieu M, Sampson EJ. 1990; International Federation of Clinical Chemistry (IFCC): scientific division, committee on enzymes. IFCC methods for the measurement of catalytic concentration of enzymes. Part 7. IFCC method for creatine kinase (ATP: creatine (N-phosphotransferase, EC 2.7.3.2). IFCC recommendation. J Automat Chem. 12:22–40. DOI: 10.1155/S1463924690000049. PMID: 18925260. PMCID: PMC2547813.
Article22. Ohkawa H, Ohishi N, Yagi K. 1979; Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem. 95:351–8. DOI: 10.1016/0003-2697(79)90738-3. PMID: 36810.
Article23. Aebi H. 1984; Catalase in vitro. Methods Enzymol. 105:121–6. DOI: 10.1016/S0076-6879(84)05016-3. PMID: 6727660.24. Sun Y, Oberley LW, Li Y. 1988; A simple method for clinical assay of superoxide dismutase. Clin Chem. 34:497–500. DOI: 10.1093/clinchem/34.3.497. PMID: 3349599.
Article25. ELLMAN GL. 1959; Tissue sulfhydryl groups. Arch Biochem Biophys. 82:70–7. DOI: 10.1016/0003-9861(59)90090-6. PMID: 13650640.
Article26. Lawrence RA, Burk RF. 1976; Glutathione peroxidase activity in selenium-deficient rat liver. Biochem Biophys Res Commun. 71:952–8. DOI: 10.1016/0006-291X(76)90747-6. PMID: 971321.
Article27. Damiani RM, Moura DJ, Viau CM, Caceres RA, Henriques JAP, Saffi J. 2016; Pathways of cardiac toxicity: comparison between chemotherapeutic drugs doxorubicin and mitoxantrone. Arch Toxicol. 90:2063–76. DOI: 10.1007/s00204-016-1759-y. PMID: 27342245.
Article28. Chen X, Peng X, Luo Y, You J, Yin D, Xu Q, He H, He M. 2019; Quercetin protects cardiomyocytes against doxorubicin-induced toxicity by suppressing oxidative stress and improving mitochondrial function via 14-3-3γ. Toxicol Mech Methods. 29:344–54. DOI: 10.1080/15376516.2018.1564948. PMID: 30636491.
Article29. Abdel-Daim MM, Kilany OE, Khalifa HA, Ahmed AAM. 2017; Allicin ameliorates doxorubicin-induced cardiotoxicity in rats via suppression of oxidative stress, inflammation and apoptosis. Cancer Chemother Pharmacol. 80:745–53. DOI: 10.1007/s00280-017-3413-7. PMID: 28785995.
Article30. Alkreathy HM, Damanhouri ZA, Ahmed N, Slevin M, Osman AM. 2012; Mechanisms of cardioprotective effect of aged garlic extract against doxorubicin-induced cardiotoxicity. Integr Cancer Ther. 11:364–70. DOI: 10.1177/1534735411426726. PMID: 22172987.
Article31. Abd El-Halim SS, Mohamed MM. 2012; Garlic powder attenuates acrylamide-induced oxidative damage in multiple organs in rat. J Appl Sci Res. 8:168–73.32. Somade OT, Adedokun AH, Adeleke IK, Taiwo MA, Oyeniran MO. 2019; Diallyl disulfide, a garlic-rich compound ameliorates trichloromethane-induced renal oxidative stress, NFkB activation and apoptosis in rats. Clin Nutr Exp. 23:44–59. DOI: 10.1016/j.yclnex.2018.10.007.33. Wu R, Wang HL, Yu HL, Cui XH, Xu MT, Xu X, Gao JP. 2016; Doxorubicin toxicity changes myocardial energy metabolism in rats. Chem Biol Interact. 244:149–58. DOI: 10.1016/j.cbi.2015.12.010. PMID: 26721193.
Article34. Mantawy EM, Esmat A, El-Bakly WM, Salah ElDin RA, El-Demerdash E. 2017; Mechanistic clues to the protective effect of chrysin against doxorubicin-induced cardiomyopathy: Plausible roles of p53, MAPK and AKT pathways. Sci Rep. 7:4795. DOI: 10.1038/s41598-017-05005-9. PMID: 28684738. PMCID: PMC5500480.
Article35. Takemura G, Fujiwara H. 2007; Doxorubicin-induced cardiomyopathy from the cardiotoxic mechanisms to management. Prog Cardiovasc Dis. 49:330–52. DOI: 10.1016/j.pcad.2006.10.002. PMID: 17329180.36. Lončar-Turukalo T, Vasić M, Tasić T, Mijatović G, Glumac S, Bajić D, Japunžić-Žigon N. 2015; Heart rate dynamics in doxorubicin-induced cardiomyopathy. Physiol Meas. 36:727–39. DOI: 10.1088/0967-3334/36/4/727. PMID: 25798626.
Article37. Pisoschi AM, Pop A. 2015; The role of antioxidants in the chemistry of oxidative stress: A review. Eur J Med Chem. 97:55–74. DOI: 10.1016/j.ejmech.2015.04.040. PMID: 25942353.
Article38. Zhang QL, Yang JJ, Zhang HS. 2019; Carvedilol (CAR) combined with carnosic acid (CAA) attenuates doxorubicin-induced cardiotoxicity by suppressing excessive oxidative stress, inflammation, apoptosis and autophagy. Biomed Pharmacother. 109:71–83. DOI: 10.1016/j.biopha.2018.07.037. PMID: 30396094.
Article39. Halliwell B, Gutteridge J. 2007. Free radicals in biology and medicine. 4th ed. Oxford University Press;Oxford:40. Elberry AA, Abdel-Naim AB, Abdel-Sattar EA, Nagy AA, Mosli HA, Mohamadin AM, Ashour OM. 2010; Cranberry (Vaccinium macrocarpon) protects against doxorubicin-induced cardiotoxicity in rats. Food Chem Toxicol. 48:1178–84. DOI: 10.1016/j.fct.2010.02.008. PMID: 20146931.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- The Role of Apoptosis in Adriamycin Induced Cardiotoxicity and Preventive Effect of L-carnitine in Rat
- Garlic and allopurinol alleviate the apoptotic pathway in rats’ brain following exposure to fipronil insecticide
- The protective effect of melatonin administration against adriamycin-induced cardiotoxicity in rats
- Antioxidant Effect of Garlic Supplement Against Exercise-Induced Oxidative Stress in Rats
- Effect of organosulfur compounds on the expression of UDP-glucuronosyltransferase and thyroid hormone level in TCDD-treated rats