Cancer Res Treat.
2020 Apr;52(2):586-593. 10.4143/crt.2019.242.
Risk Factors for Cognitive Impairment in High-Grade Glioma PatientsTreated with Postoperative Radiochemotherapy
- Affiliations
-
- 1Department of Radiation Oncology, Shandong Cancer Hospital Aliated to Shandong University, Jinan, China
- 2Department of Oncology, Laiwu Central Hospital of Xinwen Mining Group, Jinan, China
- 3Department of Radiation Oncology, Qilu Hospital of Shandong University, Jinan, China
- 4Department of Oncology, No. 88 Hospital of People's Liberation Army, Tai'an, China
- KMID: 2500343
- DOI: http://doi.org/10.4143/crt.2019.242
Abstract
- Purpose
Fractionated radiotherapy as well as concomitant and adjuvant chemotherapy such as temozolomide for postoperative high-grade glioma (HGG) patients improves progressionfree survival and overall survival. Multiple factors such as chemotherapy, radiotherapy, tumor grade, residual tumor volume, and genetic modifications might play a role in the formation of cognitive impairment. The risk factors of cognitive impairment in postoperative patients with HGG receiving radiotherapy and chemotherapy remains a concern in this population. The purpose of this study was to identify risk factors for cognitive impairment in patients of postoperative HGG.
Materials and Methods
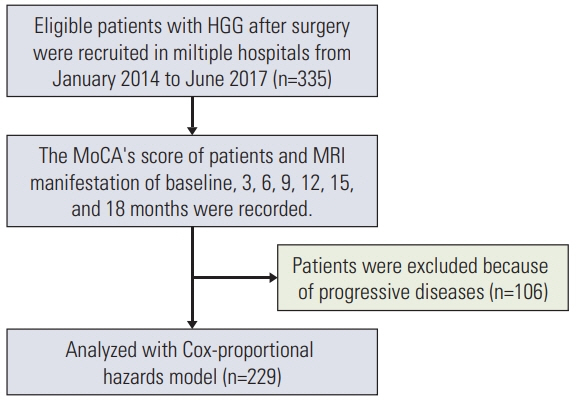
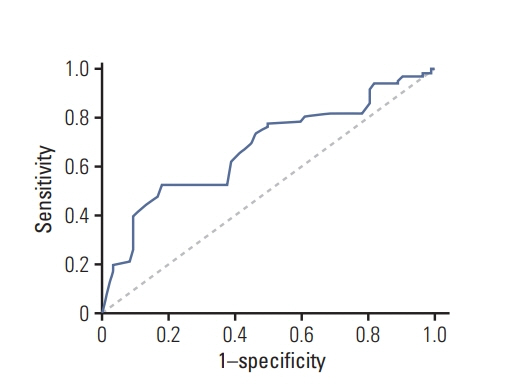
A total of 229 patients with HGG who underwent surgery were analyzed. Cognitive impairment was defined as a decrease of Cognitive Assessment Montreal (MoCA)’s score in at least two cognitive domains or any MoCA’s score of less than 26 points at the time of study compared with baseline level. Multiple potential risk factors including methylated status of the O6-methylguanine-DNA methyltransferase (MGMT) promoter, glioma World Health Organization (WHO) grade, residual tumor volume, education, and sex were analyzed. Cox univariate and multivariate regression analysis was used to detect the significant risk factors for cognitive impairment.
Results
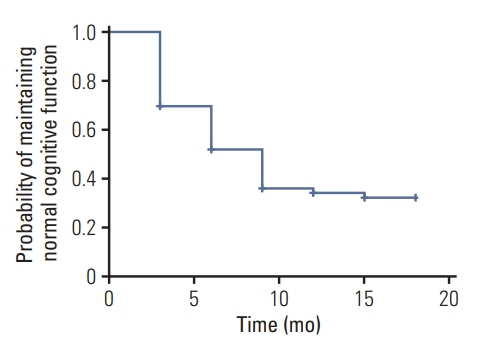
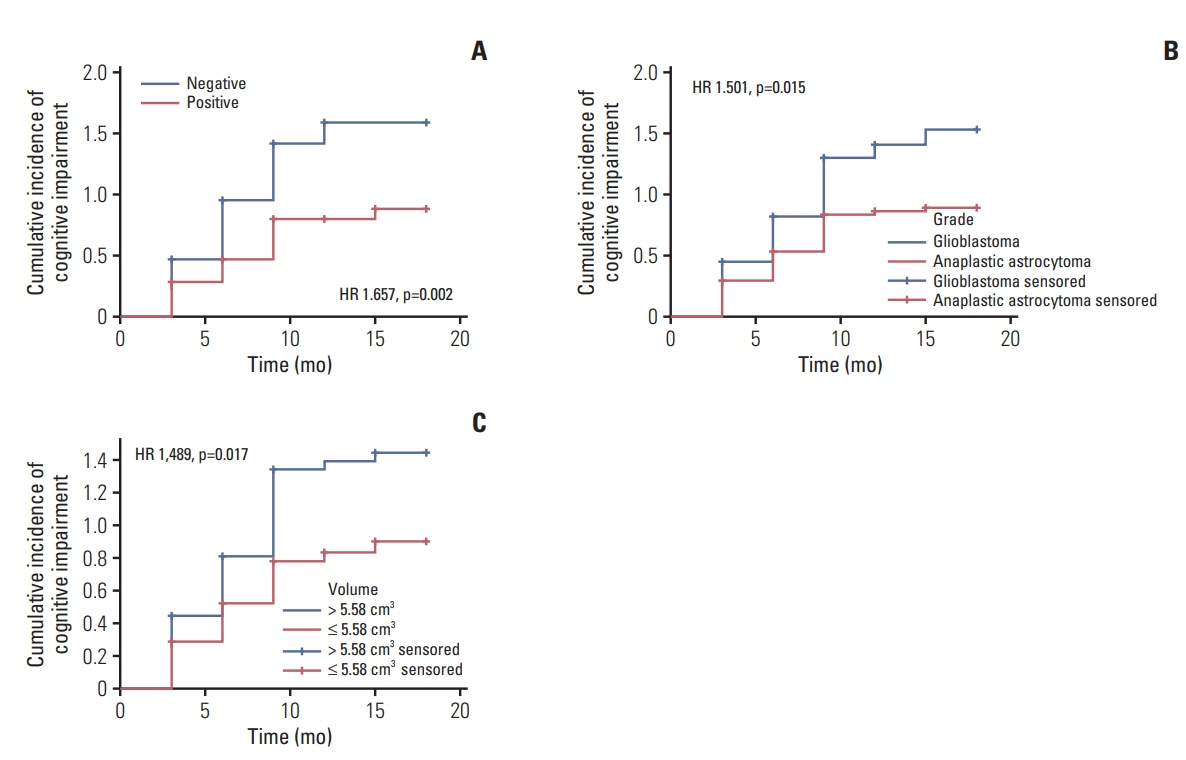
At the end of follow-up among the 229 patients, 147 patients (67%) developed cognitive impairment. 82 patients (36%) remained in normal cognitive condition. In multivariate analysis, unmethylated MGMT promoter (hazard ratio [HR], 1.679; 95% confidence interval [CI], 1.212 to 2.326; p=0.002), glioblastoma (HR, 1.550; 95% CI, 1.117 to 2.149; p=0.009), and residual tumor volume > 5.58 cm3 (HR, 1.454; 95% CI, 1.047 to 2.020; p=0.026) were independent risk factors for cognitive impairment.
Conclusion
Methylated status of the MGMT promoter, glioma WHO grade, and residual tumor volume might be risk factors for the cognitive impairment in postoperative patients with HGG.
Figure
Reference
-
References
1. Ostrom QT, Gittleman H, Fulop J, Liu M, Blanda R, Kromer C, et al. CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2008-2012. Neuro Oncol. 2015; 17 Suppl 4:iv1–62.
Article2. Klein M, Taphoorn MJ, Heimans JJ, van der Ploeg HM, Vandertop WP, Smit EF, et al. Neurobehavioral status and healthrelated quality of life in newly diagnosed high-grade glioma patients. J Clin Oncol. 2001; 19:4037–47.
Article3. Tucha O, Smely C, Preier M, Lange KW. Cognitive deficits before treatment among patients with brain tumors. Neurosurgery. 2000; 47:324–33.
Article4. Sarkissian V. The sequelae of cranial irradiation on human cognition. Neurosci Lett. 2005; 382:118–23.
Article5. Armstrong CL, Hunter JV, Ledakis GE, Cohen B, Tallent EM, Goldstein BH, et al. Late cognitive and radiographic changes related to radiotherapy: initial prospective findings. Neurology. 2002; 59:40–8.
Article6. Wang M, Cairncross G, Shaw E, Jenkins R, Scheithauer B, Brachman D, et al. Cognition and quality of life after chemotherapy plus radiotherapy (RT) vs. RT for pure and mixed anaplastic oligodendrogliomas: radiation therapy oncology group trial 9402. Int J Radiat Oncol Biol Phys. 2010; 77:662–9.
Article7. Laack NN, Brown PD, Ivnik RJ, Furth AF, Ballman KV, Hammack JE, et al. Cognitive function after radiotherapy for supratentorial low-grade glioma: a North Central Cancer Treatment Group prospective study. Int J Radiat Oncol Biol Phys. 2005; 63:1175–83.
Article8. Klein M, Heimans JJ, Aaronson NK, van der Ploeg HM, Grit J, Muller M, et al. Effect of radiotherapy and other treatmentrelated factors on mid-term to long-term cognitive sequelae in low-grade gliomas: a comparative study. Lancet. 2002; 360:1361–8.
Article9. Verger E, Gil M, Yaya R, Vinolas N, Villa S, Pujol T, et al. Temozolomide and concomitant whole brain radiotherapy in patients with brain metastases: a phase II randomized trial. Int J Radiat Oncol Biol Phys. 2005; 61:185–91.
Article10. Antonadou D, Paraskevaidis M, Sarris G, Coliarakis N, Economou I, Karageorgis P, et al. Phase II randomized trial of temozolomide and concurrent radiotherapy in patients with brain metastases. J Clin Oncol. 2002; 20:3644–50.
Article11. Saito K, Mukasa A, Narita Y, Tabei Y, Shinoura N, Shibui S, et al. Toxicity and outcome of radiotherapy with concomitant and adjuvant temozolomide in elderly patients with glioblastoma: a retrospective study. Neurol Med Chir (Tokyo). 2014; 54:272–9.
Article12. Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005; 53:695–9.
Article13. Smith T, Gildeh N, Holmes C. The Montreal Cognitive Assessment: validity and utility in a memory clinic setting. Can J Psychiatry. 2007; 52:329–32.
Article14. Wang Q, Qi F, Song X, Di J, Zhang L, Zhou Y, et al. A prospective longitudinal evaluation of cognition and depression in postoperative patients with high-grade glioma following radiotherapy and chemotherapy. J Cancer Res Ther. 2018; 14(Suppl):S1048–51.15. Durand T, Jacob S, Lebouil L, Douzane H, Lestaevel P, Rahimian A, et al. EpiBrainRad: an epidemiologic study of the neurotoxicity induced by radiotherapy in high grade glioma patients. BMC Neurol. 2015; 15:261.
Article16. Dhermain F. Radiotherapy of high-grade gliomas: current standards and new concepts, innovations in imaging and radiotherapy, and new therapeutic approaches. Chin J Cancer. 2014; 33:16–24.
Article17. Kim BS, Seol HJ, Nam DH, Park CK, Kim IH, Kim TM, et al. Concurrent chemoradiotherapy with temozolomide followed by adjuvant temozolomide for newly diagnosed glioblastoma patients: a retrospective multicenter observation study in Korea. Cancer Res Treat. 2017; 49:193–203.
Article18. Kole AJ, Park HS, Yeboa DN, Rutter CE, Corso CD, Aneja S, et al. Concurrent chemoradiotherapy versus radiotherapy alone for "biopsy-only" glioblastoma multiforme. Cancer. 2016; 122:2364–70.
Article19. Woo PY, Ho JM, Tse TP, Lam SW, Mak CH, Chan DT, et al. Determining a cut-off residual tumor volume threshold for patients with newly diagnosed glioblastoma treated with temozolomide chemoradiotherapy: a multicenter cohort study. J Clin Neurosci. 2019; 63:134–41.
Article20. Yang K, Jung SW, Shin H, Lim DH, Lee JI, Kong DS, et al. Cancer genetic markers according to radiotherapeutic response in patients with primary glioblastoma: radiogenomic approach for precision medicine. Radiother Oncol. 2019; 131:66–74.21. Wefel JS, Noll KR, Rao G, Cahill DP. Neurocognitive function varies by IDH1 genetic mutation status in patients with malignant glioma prior to surgical resection. Neuro Oncol. 2016; 18:1656–63.
Article22. Kesler SR, Noll K, Cahill DP, Rao G, Wefel JS. The effect of IDH1 mutation on the structural connectome in malignant astrocytoma. J Neurooncol. 2017; 131:565–74.
Article23. Jiang X, Reardon DA, Desjardins A, Vredenburgh JJ, Quinn JA, Austin AD, et al. O6-methylguanine-DNA methyltransferase (MGMT) immunohistochemistry as a predictor of resistance to temozolomide in primary CNS lymphoma. J Neurooncol. 2013; 114:135–40.24. Binabaj MM, Bahrami A, ShahidSales S, Joodi M, Joudi Mashhad M, Hassanian SM, et al. The prognostic value of MGMT promoter methylation in glioblastoma: a meta-analysis of clinical trials. J Cell Physiol. 2018; 233:378–86.
Article25. Smrdel U, Popovic M, Zwitter M, Bostjancic E, Zupan A, Kovac V, et al. Long-term survival in glioblastoma: methyl guanine methyl transferase (MGMT) promoter methylation as independent favourable prognostic factor. Radiol Oncol. 2016; 50:394–401.
Article26. Liu Y, Zhou R, Sulman EP, Scheurer ME, Boehling N, Armstrong GN, et al. Genetic modulation of neurocognitive function in glioma patients. Clin Cancer Res. 2015; 21:3340–6.
Article27. Miotto EC, Junior AS, Silva CC, Cabrera HN, Machado MA, Benute GR, et al. Cognitive impairments in patients with low grade gliomas and high grade gliomas. Arq Neuropsiquiatr. 2011; 69:596–601.
Article28. Noll KR, Sullaway C, Ziu M, Weinberg JS, Wefel JS. Relationships between tumor grade and neurocognitive functioning in patients with glioma of the left temporal lobe prior to surgical resection. Neuro Oncol. 2015; 17:580–7.
Article29. Surma-aho O, Niemela M, Vilkki J, Kouri M, Brander A, Salonen O, et al. Adverse long-term effects of brain radiotherapy in adult low-grade glioma patients. Neurology. 2001; 56:1285–90.
Article30. Talacchi A, Santini B, Savazzi S, Gerosa M. Cognitive effects of tumour and surgical treatment in glioma patients. J Neurooncol. 2011; 103:541–9.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Factors Associated With Neurocognitive Impairment Following Chemoradiotherapy in Patients With High-Grade Glioma: Results of a Prospective Trial
- Risk and Protective Factors for the Onset of Cognitive Impairment in Korea: A 10-Year Longitudinal Panel Study
- Endocrine Risk Factors for Cognitive Impairment
- Association between Hearing Difficulty, Masticatory Difficulty, and Risk of Cognitive Impairment: Analysis of the Korean Longitudinal Study of Aging Data (2006-2020)
- Epidemiology of Age-Associated Memory Impairment