Blood Res.
2020 Mar;55(1):57-61. 10.5045/br.2020.55.1.57.
Mantle cell lymphoma relapsed after autologous stem cell transplantation: a single-center experience
- Affiliations
-
- 1Department of Hospital and Specialty Medicine, Veterans Affairs Puget Sound Health Care System, Seattle, WA, USA. tchaunce@uw.edu
- 2Division of Oncology, University of Washington Medicine, Seattle, WA, USA.
- 3Clinical Research Division, Fred Hutchinson Cancer Research Center, Seattle, WA, USA.
- 4Division of Hematology, University of Washington Medicine, Seattle, WA, USA.
- 5Division of Pathology, University of Washington Medicine, Seattle, WA, USA.
- KMID: 2472009
- DOI: http://doi.org/10.5045/br.2020.55.1.57
Abstract
- BACKGROUND
Autologous stem cell transplantation (autoSCT) can extend remission of mantle cell lymphoma (MCL), but the management of subsequent relapse is challenging.
METHODS
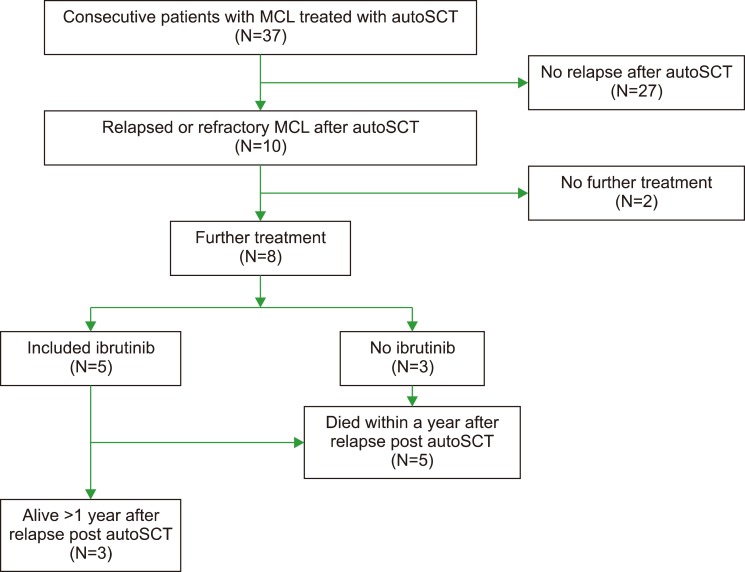
We examined consecutive patients with MCL who underwent autoSCT at Veterans Affairs Puget Sound Health Care System between 2009 and 2017 (N=37).
RESULTS
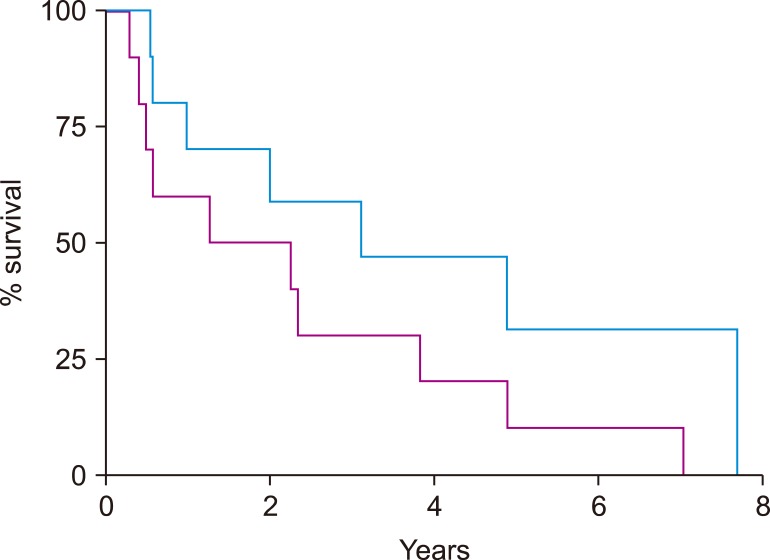
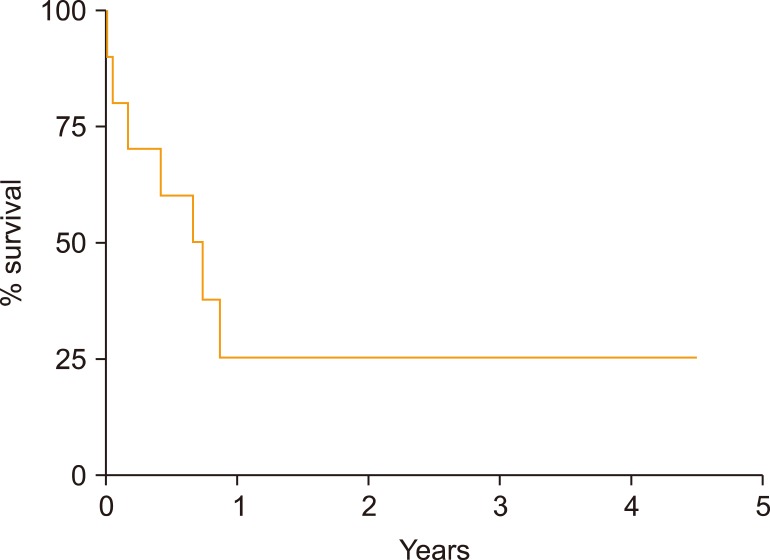
Ten patients experienced disease progression after autoSCT and were included in this analysis. Median progression free survival after autoSCT was 1.8 years (range, 0.3-7.1) and median overall survival after progression was only 0.7 years (range, 0.1 to not reached). The 3 patients who survived more than 1 year after progression were treated with ibrutinib.
CONCLUSION
Our findings suggest that ibrutinib can achieve relatively prolonged control of MCL progressing after autoSCT.
MeSH Terms
Figure
Reference
-
1. Gerson JN, Handorf E, Villa D, et al. Survival outcomes of younger patients with mantle cell lymphoma treated in the rituximab era. J Clin Oncol. 2019; 37:471–480. PMID: 30615550.2. Le Gouill S, Thieblemont C, Oberic L, et al. Rituximab after autologous stem-cell transplantation in mantle-cell lymphoma. N Engl J Med. 2017; 377:1250–1260. PMID: 28953447.
Article3. Hermine O, Hoster E, Walewski J, et al. Addition of high-dose cytarabine to immunochemotherapy before autologous stem-cell transplantation in patients aged 65 years or younger with mantle cell lymphoma (MCL Younger): a randomised, open-label, phase 3 trial of the European Mantle Cell Lymphoma Network. Lancet. 2016; 388:565–575. PMID: 27313086.4. Graf SA, Stevenson PA, Holmberg LA, et al. Maintenance rituximab after autologous stem cell transplantation in patients with mantle cell lymphoma. Ann Oncol. 2015; 26:2323–2328. PMID: 26347113.
Article5. Dietrich S, Boumendil A, Finel H, et al. Outcome and prognostic factors in patients with mantle-cell lymphoma relapsing after autologous stem-cell transplantation: a retrospective study of the European Group for Blood and Marrow Transplantation (EBMT). Ann Oncol. 2014; 25:1053–1058. PMID: 24585719.
Article6. Vaughn JE, Sorror ML, Storer BE, et al. Long-term sustained disease control in patients with mantle cell lymphoma with or without active disease after treatment with allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Cancer. 2015; 121:3709–3716. PMID: 26207349.
Article7. Stefoni V, Pellegrini C, Broccoli A, et al. Lenalidomide in pretreated mantle cell lymphoma patients: an Italian observational multicenter retrospective study in daily clinical practice (the Lenamant Study). Oncologist. 2018; 23:1033–1038. PMID: 29674440.
Article8. Visco C, Tisi MC, Evangelista A, et al. Time to progression of mantle cell lymphoma after high-dose cytarabine-based regimens defines patients risk for death. Br J Haematol. 2019; 185:940–944. PMID: 30407625.
Article9. Davids MS, Roberts AW, Seymour JF, et al. Phase I first-in-human study of venetoclax in patients with relapsed or refractory non-Hodgkin lymphoma. J Clin Oncol. 2017; 35:826–833. PMID: 28095146.10. Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for initial evaluation, staging, and response assessment of Hodgkin and non-Hodgkin lymphoma: the Lugano classification. J Clin Oncol. 2014; 32:3059–3068. PMID: 25113753.
Article11. Rule S, Dreyling M, Goy A, et al. Outcomes in 370 patients with mantle cell lymphoma treated with ibrutinib: a pooled analysis from three open-label studies. Br J Haematol. 2017; 179:430–438. PMID: 28832957.
Article12. Hoster E, Rosenwald A, Berger F, et al. Prognostic value of Ki-67 index, cytology, and growth pattern in mantle-cell lymphoma: results from randomized trials of the european mantle cell lymphoma network. J Clin Oncol. 2016; 34:1386–1394. PMID: 26926679.
Article13. Eskelund CW, Dahl C, Hansen JW, et al. TP53 mutations identify younger mantle cell lymphoma patients who do not benefit from intensive chemoimmunotherapy. Blood. 2017; 130:1903–1910. PMID: 28819011.
Article14. Scott DW, Abrisqueta P, Wright GW, et al. New molecular assay for the proliferation signature in mantle cell lymphoma applicable to formalin-fixed paraffin-embedded biopsies. J Clin Oncol. 2017; 35:1668–1677. PMID: 28291392.
Article15. Tam CS, Anderson MA, Pott C, et al. Ibrutinib plus venetoclax for the treatment of mantle-cell lymphoma. N Engl J Med. 2018; 378:1211–1223. PMID: 29590547.
Article16. Lin RJ, Ho C, Hilden PD, et al. Allogeneic haematopoietic cell transplantation impacts on outcomes of mantle cell lymphoma with TP53 alterations. Br J Haematol. 2019; 184:1006–1010. PMID: 30537212.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Case of Mantle Cell Lymphoma Treated with Autologous Stem Cell Transplantation and Rituximab
- Autoimmune Diseases after Autologous Hematopoietic Stem Cell Transplantation in Patients with Non-Hodgkin's Lymphoma
- Patient profiles and outcomes of lymphoma patients who underwent autologous stem cell transplant in National Kidney and Transplant Institute: a single-center analysis
- Treatment of Relapsed Hodgkin Lymphoma
- A Case of Scleroderma following Autologous Peripheral Stem Cell Transplantation