J Gastric Cancer.
2020 Mar;20(1):29-40. 10.5230/jgc.2020.20.e2.
Genetic Characterization of Molecular Targets in Korean Patients with Gastrointestinal Stromal Tumors
- Affiliations
-
- 1Department of Laboratory Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 2Department of Surgery, College of Medicine, The Catholic University of Korea, Seoul, Korea. kalgs@catholic.ac.kr
- 3Department of Pathology, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- 4Division of Gastroenterology, Department of Internal Medicine, College of Medicine, The Catholic University of Korea, Seoul, Korea.
- KMID: 2471923
- DOI: http://doi.org/10.5230/jgc.2020.20.e2
Abstract
- PURPOSE
Gastrointestinal stromal tumors (GISTs) frequently harbor activating gene mutations in either KIT or platelet-derived growth factor receptor A (PDGFRA) and are highly responsive to several selective tyrosine kinase inhibitors. In this study, a targeted next-generation sequencing (NGS) assay with an Oncomine Focus Assay (OFA) panel was used for the genetic characterization of molecular targets in 30 Korean patients with GIST.
MATERIALS AND METHODS
Using the OFA that enables rapid and simultaneous detection of hotspots, single nucleotide variants (SNVs), insertion and deletions (Indels), copy number variants (CNVs), and gene fusions across 52 genes relevant to solid tumors, targeted NGS was performed using genomic DNA extracted from formalin-fixed and paraffin-embedded samples of 30 GISTs.
RESULTS
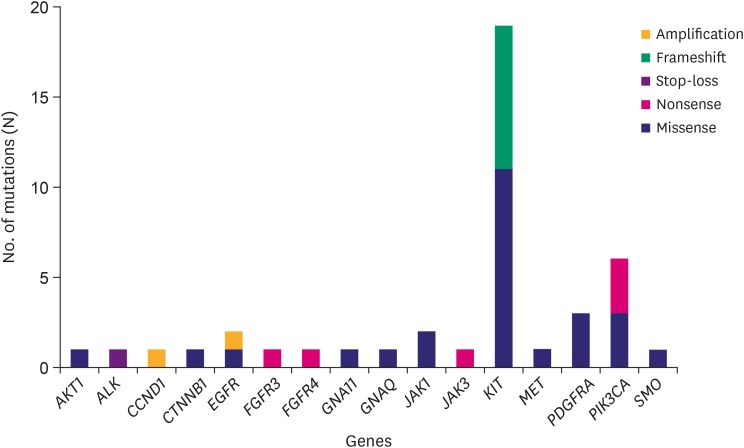
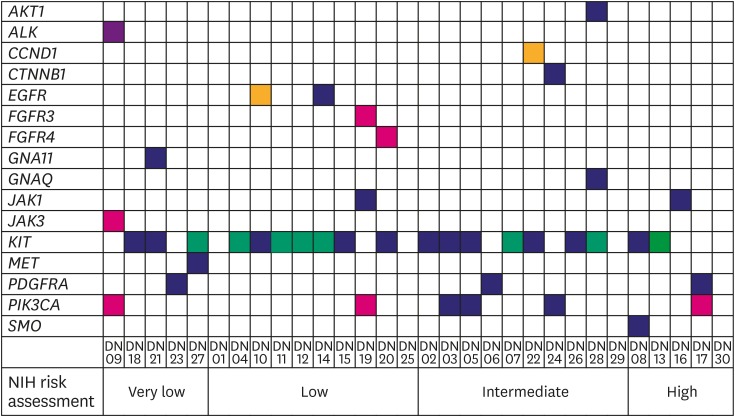
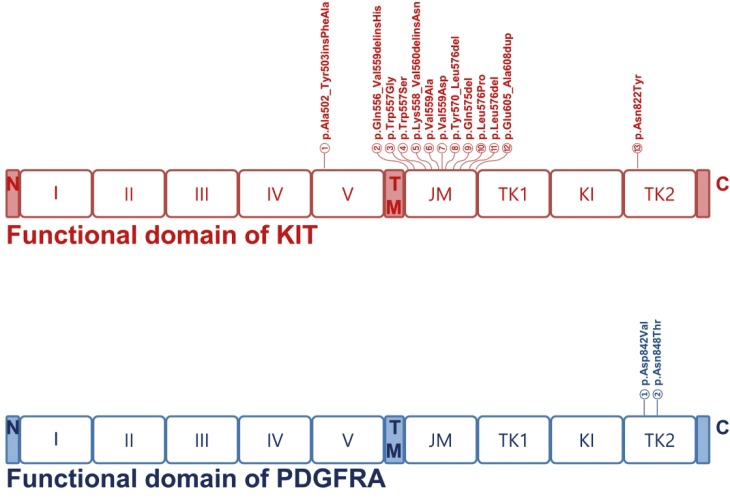
Forty-three hotspot/other likely pathogenic variants (33 SNVs, 8 Indels, and 2 amplifications) in 16 genes were identified in 26 of the 30 GISTs. KIT variants were most frequent (44%, 19/43), followed by 6 variants in PIK3CA, 3 in PDGFRA, 2 each in JAK1 and EGFR, and 1 each in AKT1, ALK, CCND1, CTNNB1, FGFR3, FGFR4, GNA11, GNAQ, JAK3, MET, and SMO. Based on the mutation types, majority of the variants carried missense mutations (60%, 26/43), followed by 8 frameshifts, 6 nonsense, 1 stop-loss, and 2 amplifications.
CONCLUSIONS
Our study confirmed the advantage of using targeted NGS with a cancer gene panel to efficiently identify mutations associated with GISTs. These findings may provide a molecular genetic basis for developing new drugs targeting these gene mutations for GIST therapy.
Keyword
MeSH Terms
Figure
Reference
-
1. Joensuu H, Wardelmann E, Sihto H, Eriksson M, Sundby Hall K, Reichardt A, et al. Effect of KIT and PDGFRA mutations on survival in patients with gastrointestinal stromal tumors treated with adjuvant imatinib: an exploratory analysis of a randomized clinical trial. JAMA Oncol. 2017; 3:602–609. PMID: 28334365.2. Xu Z, Huo X, Tang C, Ye H, Nandakumar V, Lou F, et al. Frequent KIT mutations in human gastrointestinal stromal tumors. Sci Rep. 2014; 4:5907. PMID: 25080996.
Article3. Fisher KE, Zhang L, Wang J, Smith GH, Newman S, Schneider TM, et al. Clinical validation and implementation of a targeted next-generation sequencing assay to detect somatic variants in non-small cell lung, melanoma, and gastrointestinal malignancies. J Mol Diagn. 2016; 18:299–315. PMID: 26801070.
Article4. Hamblin A, Wordsworth S, Fermont JM, Page S, Kaur K, Camps C, et al. Clinical applicability and cost of a 46-gene panel for genomic analysis of solid tumours: Retrospective validation and prospective audit in the UK National Health Service. PLoS Med. 2017; 14:e1002230. PMID: 28196074.
Article5. Giardina T, Robinson C, Grieu-Iacopetta F, Millward M, Iacopetta B, Spagnolo D, et al. Implementation of next generation sequencing technology for somatic mutation detection in routine laboratory practice. Pathology. 2018; 50:389–401. PMID: 29752127.
Article6. Shahsiah R, DeKoning J, Samie S, Latifzadeh SZ, Kashi ZM. Validation of a next generation sequencing panel for detection of hotspot cancer mutations in a clinical laboratory. Pathol Res Pract. 2017; 213:98–105. PMID: 28049581.
Article7. Williams HL, Walsh K, Diamond A, Oniscu A, Deans ZC. Validation of the Oncomine™ focus panel for next-generation sequencing of clinical tumour samples. Virchows Arch. 2018; 473:489–503. PMID: 30105577.
Article8. Fletcher CD, Berman JJ, Corless C, Gorstein F, Lasota J, Longley BJ, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol. 2002; 33:459–465. PMID: 12094370.
Article9. Jones RL. Practical aspects of risk assessment in gastrointestinal stromal tumors. J Gastrointest Cancer. 2014; 45:262–267. PMID: 24802226.
Article10. 1000 Genomes Project Consortium. Auton A, Brooks LD, Durbin RM, Garrison EP, Kang HM, et al. A global reference for human genetic variation. Nature. 2015; 526:68–74. PMID: 26432245.11. Shi E, Chmielecki J, Tang CM, Wang K, Heinrich MC, Kang G, et al. FGFR1 and NTRK3 actionable alterations in “Wild-Type” gastrointestinal stromal tumors. J Transl Med. 2016; 14:339. PMID: 27974047.
Article12. Szucs Z, Thway K, Fisher C, Bulusu R, Constantinidou A, Benson C, et al. Molecular subtypes of gastrointestinal stromal tumors and their prognostic and therapeutic implications. Future Oncol. 2017; 13:93–107. PMID: 27600498.
Article13. Joensuu H, DeMatteo RP. The management of gastrointestinal stromal tumors: a model for targeted and multidisciplinary therapy of malignancy. Annu Rev Med. 2012; 63:247–258. PMID: 22017446.
Article14. Mayr P, Märkl B, Agaimy A, Kriening B, Dintner S, Schenkirsch G, et al. Malignancies associated with GIST: a retrospective study with molecular analysis of KIT and PDGFRA. Langenbecks Arch Surg. 2019; 404:605–613. PMID: 30877378.15. Yamamoto H, Oda Y. Gastrointestinal stromal tumor: recent advances in pathology and genetics. Pathol Int. 2015; 65:9–18. PMID: 25414046.
Article16. Mei L, Du W, Idowu M, von Mehren M, Boikos SA. Advances and challenges on management of gastrointestinal stromal tumors. Front Oncol. 2018; 8:135. PMID: 29868467.
Article17. Charville GW, Longacre TA. Surgical pathology of gastrointestinal stromal tumors: practical implications of morphologic and molecular heterogeneity for precision medicine. Adv Anat Pathol. 2017; 24:336–353. PMID: 28820749.
Article18. El-Menyar A, Mekkodathil A, Al-Thani H. Diagnosis and management of gastrointestinal stromal tumors: an up-to-date literature review. J Cancer Res Ther. 2017; 13:889–900. PMID: 29237949.19. Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998; 279:577–580. PMID: 9438854.20. Chen LL, Holden JA, Choi H, Zhu J, Wu EF, Jones KA, et al. Evolution from heterozygous to homozygous KIT mutation in gastrointestinal stromal tumor correlates with the mechanism of mitotic nondisjunction and significant tumor progression. Mod Pathol. 2008; 21:826–836. PMID: 18488000.21. Sciot R, Debiec-Rychter M, Daugaard S, Fisher C, Collin F, van Glabbeke M, et al. Distribution and prognostic value of histopathologic data and immunohistochemical markers in gastrointestinal stromal tumours (GISTs): an analysis of the EORTC phase III trial of treatment of metastatic GISTs with imatinib mesylate. Eur J Cancer. 2008; 44:1855–1860. PMID: 18653326.
Article22. Wozniak A, Rutkowski P, Schöffski P, Ray-Coquard I, Hostein I, Schildhaus HU, et al. Tumor genotype is an independent prognostic factor in primary gastrointestinal stromal tumors of gastric origin: a European multicenter analysis based on ConticaGIST. Clin Cancer Res. 2014; 20:6105–6116. PMID: 25294914.
Article23. Lasota J, Miettinen M. KIT and PDGFRA mutations in gastrointestinal stromal tumors (GISTs). Semin Diagn Pathol. 2006; 23:91–102. PMID: 17193822.24. Lasota J, Stachura J, Miettinen M. GISTs with PDGFRA exon 14 mutations represent subset of clinically favorable gastric tumors with epithelioid morphology. Lab Invest. 2006; 86:94–100. PMID: 16258521.25. Lasota J, Felisiak-Golabek A, Wasag B, Kowalik A, Zieba S, Chlopek M, et al. Frequency and clinicopathologic profile of PIK3CA mutant GISTs: molecular genetic study of 529 cases. Mod Pathol. 2016; 29:275–282. PMID: 26796526.26. Tornillo L, Duchini G, Carafa V, Lugli A, Dirnhofer S, Di Vizio D, et al. Patterns of gene amplification in gastrointestinal stromal tumors (GIST). Lab Invest. 2005; 85:921–931. PMID: 15864317.
Article27. Lopes LF, Bacchi CE. EGFR and gastrointestinal stromal tumor: an immunohistochemical and FISH study of 82 cases. Mod Pathol. 2007; 20:990–994. PMID: 17643098.28. Boikos SA, Pappo AS, Killian JK, LaQuaglia MP, Weldon CB, George S, et al. Molecular subtypes of KIT/PDGFRA wild-type gastrointestinal stromal tumors: a report from the National Institutes of Health Gastrointestinal Stromal Tumor Clinic. JAMA Oncol. 2016; 2:922–928. PMID: 27011036.29. Heinrich MC, Corless CL, Demetri GD, Blanke CD, von Mehren M, Joensuu H, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003; 21:4342–4349. PMID: 14645423.
Article30. Gopie P, Mei L, Faber AC, Grossman SR, Smith SC, Boikos SA. Classification of gastrointestinal stromal tumor syndromes. Endocr Relat Cancer. 2018; 25:R49–R58. PMID: 29170162.
Article31. Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST). Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. J Clin Oncol. 2010; 28:1247–1253. PMID: 20124181.32. Cai H, Jing C, Chang X, Ding D, Han T, Yang J, et al. Mutational landscape of gastric cancer and clinical application of genomic profiling based on target next-generation sequencing. J Transl Med. 2019; 17:189. PMID: 31164161.
Article33. Luthra R, Patel KP, Routbort MJ, Broaddus RR, Yau J, Simien C, et al. A Targeted high-throughput next-generation sequencing panel for clinical screening of mutations, gene amplifications, and fusions in solid tumors. J Mol Diagn. 2017; 19:255–264. PMID: 28017569.
Article34. Dudley JC, Gurda GT, Tseng LH, Anderson DA, Chen G, Taube JM, et al. Tumor cellularity as a quality assurance measure for accurate clinical detection of BRAF mutations in melanoma. Mol Diagn Ther. 2014; 18:409–418. PMID: 24604154.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A case of huge Gastrointestinal stromal tumor masquerading as an ovarian malignancy
- A Case of Massive Bleeding from Jejunal Stromal Tumor Diagnosed by Intraoperative Enteroscopy: A Case of Jejunal Stromal Tumor Bleeding
- Clinical Relevance of the Location of Gastric Gastrointestinal Stromal Tumors
- Gastrointestinal Stromal Tumors associated with Neurofibromatosis Type I: A Report of Two Cases
- Multiple Gastrointestinal Stromal Tumors of the Small Intestine