J Bacteriol Virol.
2020 Mar;50(1):25-34. 10.4167/jbv.2020.50.1.025.
Expression and Purification of Recombinant Mayaro Virus Envelope Glycoproteins E1 and E2 to Develop a Mayaro Virus Detection System
- Affiliations
-
- 1BioNano Health Guard Research Center, 125 Gwahak-ro, Yuseong-gu, Daejeon 34141, Republic of Korea. marsp@kribb.re.kr, ybshin@kribb.re.kr
- 2Department of Biochemistry, College of Natural Sciences, Chungnam National University, 99 Daehak-ro, Yuseong-gu, Daejeon 34134, Republic of Korea.
- 3Department of Clinical Laboratory Science, Daejeon Health Institute of Technology, Daejeon 34504, Republic of Korea.
- 4Department of General Education, Daejeon Health Institute of Technology, Daejeon 34504, Republic of Korea.
- 5Bionanotechnology Research Center, Korea Research Institute of Bioscience and Biotechnology (KRIBB), 125 Gwahak-ro, Yuseong-gu, Daejeon 34141, Republic of Korea.
- KMID: 2471835
- DOI: http://doi.org/10.4167/jbv.2020.50.1.025
Abstract
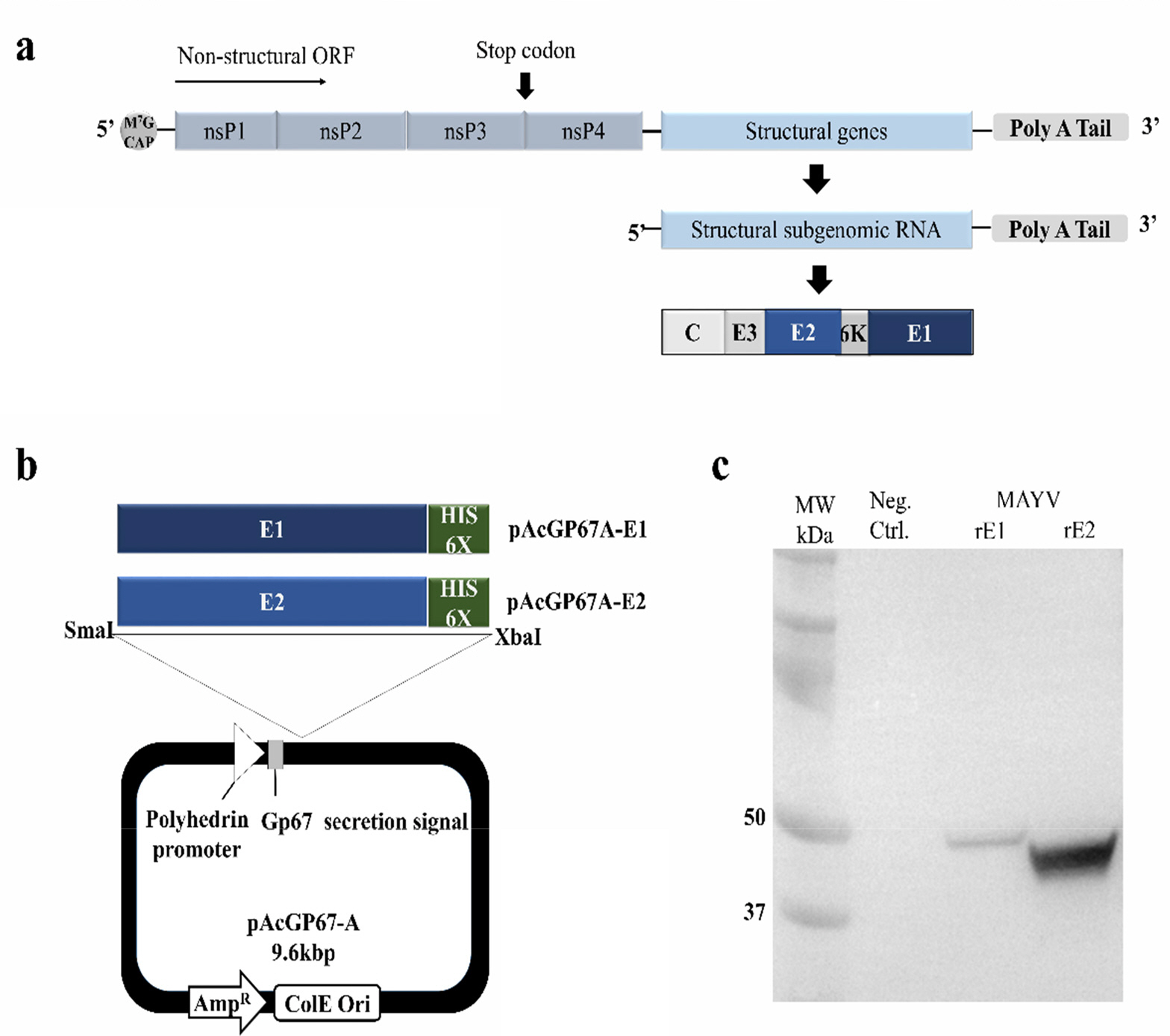
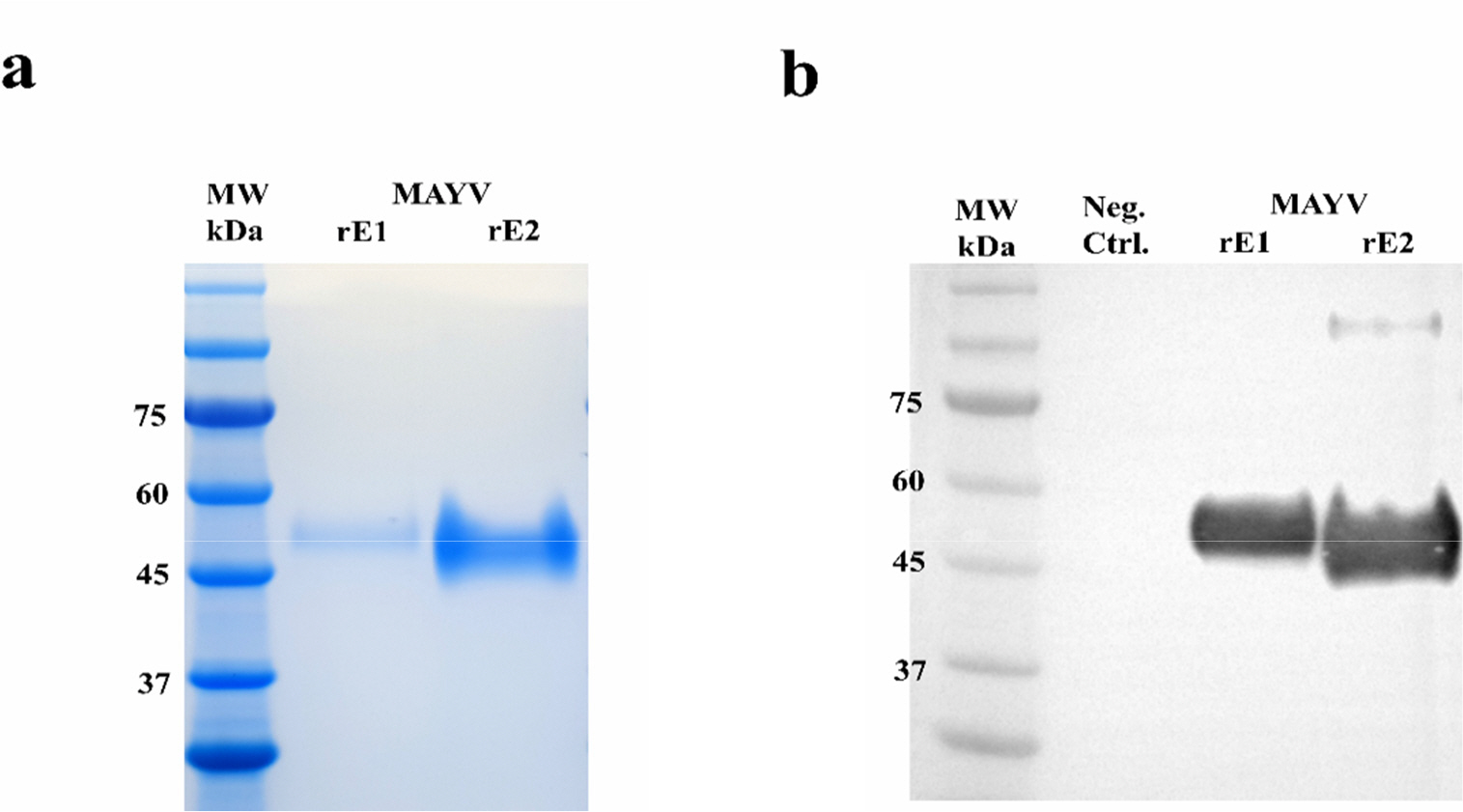
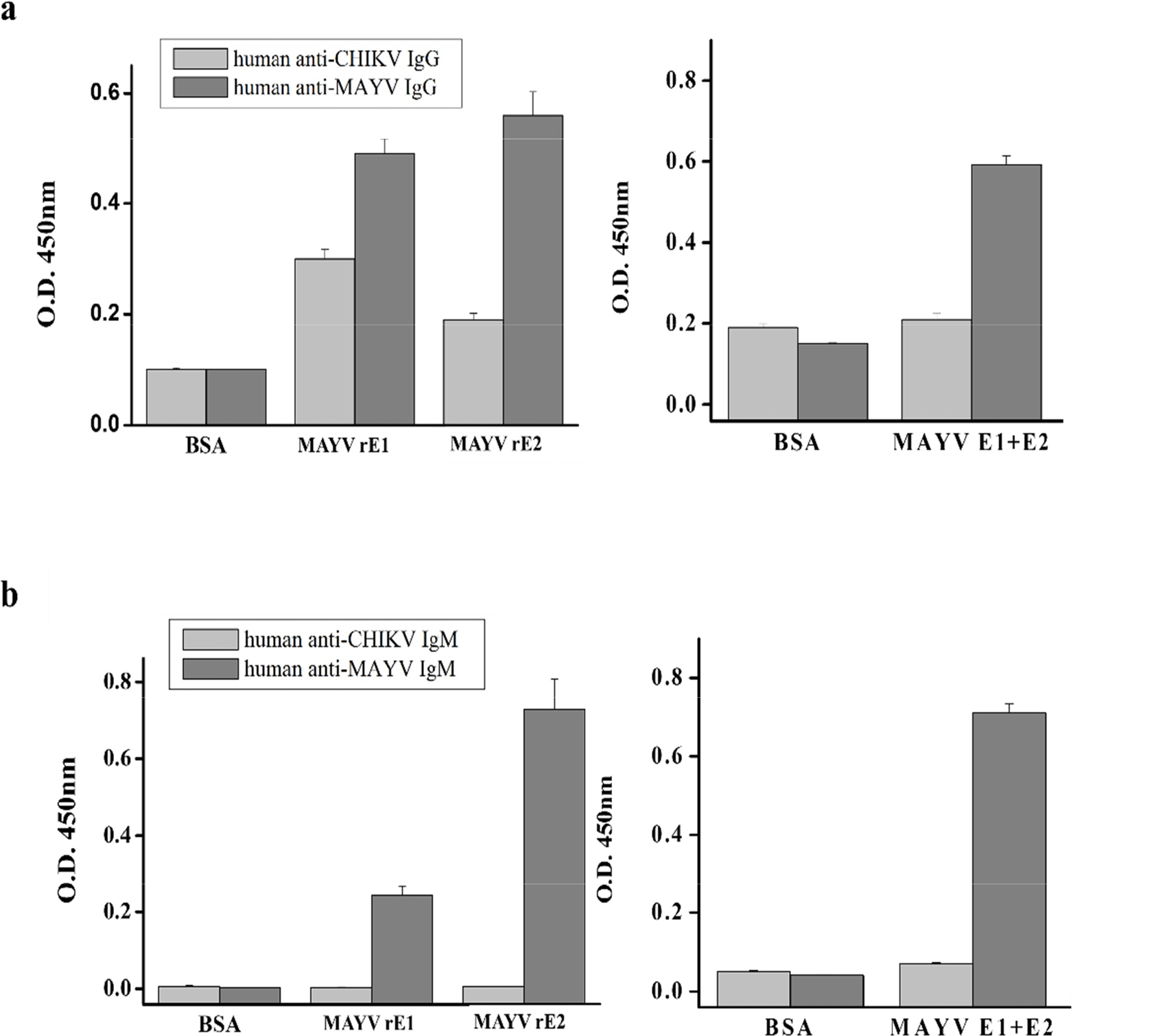
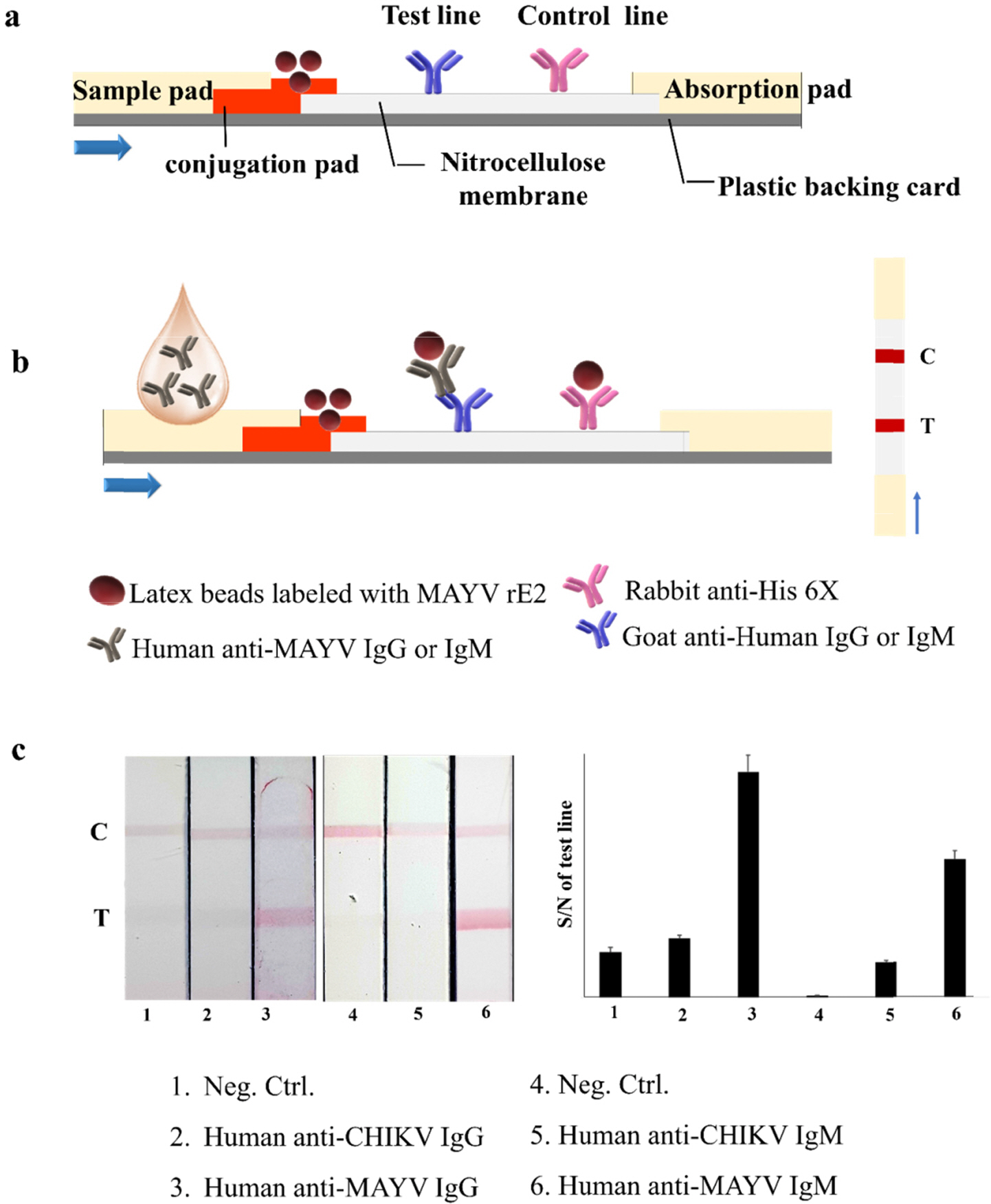
- Mayaro virus (MAYV) is a mosquito-transmitted alphavirus that produces an acute, usually non-fatal, febrile illness including Mayaro fever. Like other alphaviruses, the MAYV E1 and E2 envelope glycoproteins are major viral surface antigens that play a key role in host recognition and infection. Here, we report expression and purification methods for recombinant MAYV E1 (rE1) and rE2 using a baculovirus system. Enzyme-linked immunosorbent assays (ELISA) revealed that rE1 and rE2 were antigenic and reacted with human anti-MAYV IgG and IgM. Cross-reactivity was also confirmed with human anti-Chikungunya virus (CHIKV) IgG and IgM. Furthermore, we developed an immunochromatographic strip test (IST) with rE2 to diagnose MAYV infection. Thus, purified rE2 may be valuable tool for rapidly diagnosing MAYV infection.
Keyword
MeSH Terms
Figure
Reference
-
1). Muñ oz M, Navarro JC. [Mayaro: a re-emerging Arbovirus in Venezuela and Latin America]. Biomedica. 2012; 32:286–302.2). Smith JL, Pugh CL, Cisney ED, Keasey SL, Guevara C, Ampuero JS, et al. Human Antibody Responses to Emerging Mayaro Virus and Cocirculating Alphavirus Infections Examined by Using Structural Proteins from Nine New and Old World Lineages. mSphere. 2018; 3.
Article3). Suhrbier A, Jaffar-Bandjee MC, Gasque P. Arthritogenic alphaviruses–an overview. Nat Rev Rheumatol. 2012; 8:420–9.
Article4). Choi H, Kudchodkar SB, Reuschel EL, Asija K, Borole P, Ho M, et al. Protective immunity by an engineered DNA vaccine for Mayaro virus. PLoS Negl Trop Dis. 2019; 13:e0007042.
Article5). Powers AM, Aguilar PV, Chandler LJ, Brault AC, Meakins TA, Watts D, et al. Genetic relationships among Mayaro and Una viruses suggest distinct patterns of transmission. Am J Trop Med Hyg. 2006; 75:461–9.
Article6). Rodrí guez-Morales AJ, Paniz-Mondolfi AE, Villamil-Gó mez WE, Navarro JC. Mayaro, Oropouche and Venezuelan Equine Encephalitis viruses: Following in the footsteps of Zika? Travel Med Infect Dis. 2017; 15:72–3.7). Auguste AJ, Liria J, Forrester NL, Giambalvo D, Moncada M, Long KC, et al. Evolutionary and Ecological Characterization of Mayaro Virus Strains Isolated during an Outbreak, Venezuela, 2010. Emerg Infect Dis. 2015; 21:1742–50.
Article8). Tesh RB, Watts DM, Russell KL, Damodaran C, Calampa C, Cabezas C, et al. Mayaro virus disease: an emerging mosquito-borne zoonosis in tropical South America. Clin Infect Dis. 1999; 28:67–73.
Article9). Esposito DLA, Fonseca BALD. Will Mayaro virus be responsible for the next outbreak of an arthropod-borne virus in Brazil? Braz J Infect Dis. 2017; 21:540–4.
Article10). Cheng RH, Kuhn RJ, Olson NH, Rossmann MG, Choi HK, Smith TJ, et al. Nucleocapsid and glycoprotein organization in an enveloped virus. Cell. 1995; 80:621–30.
Article11). Tello D, Rodrí guez-Rodrí guez M, Yé lamos B, Gó mez-Gutié rrez J, Peterson DL, Gavilanes F. High-yield production of a chimeric glycoprotein based on permuted E1 and E2 HCV envelope ectodomains. J Virol Methods. 2015; 213:38–44.
Article12). Masoomi Dezfooli S, Tan WS, Tey BT, Ooi CW, Hussain SA. Expression and purification of the matrix protein of Nipah virus in baculovirus insect cell system. Biotechnol Prog. 2016; 32:171–7.
Article13). Tello D, Rodrí guez-Rodrí guez M, Yé lamos B, Gó mez-Gutié rrez J, Ortega S, Pacheco B, et al. Expression and structural properties of a chimeric protein based on the ectodomains of E1 and E2 hepatitis C virus envelope glycoproteins. Protein Expr Purif. 2010; 71:123–31.
Article14). Lombana L, Ortega-Atienza S, Gó mez-Gutié rrez J, Yé lamos B, Peterson DL, Gavilanes F. The deletion of residues 268-292 of E1 impairs the ability of HCV envelope proteins to induce pore formation. Virus Res. 2016; 217:63–70.
Article15). Mackay IM, Arden KE. Mayaro virus: a forest virus primed for a trip to the city? Microbes Infect. 2016; 18:724–34.
Article16). Pinheiro FP, Freitas RB, Travassos da Rosa JF, Gabbay YB, Mello WA, LeDuc JW. An outbreak of Mayaro virus disease in Belterra, Brazil. I. Clinical and virological findings. Am J Trop Med Hyg. 1981; 30:674–81.17). Fumagalli MJ, de Souza WM, Romeiro MF, de Souza Costa MC, Slhessarenko RD, Figueiredo LTM. Development of an Enzyme-Linked Immunosorbent Assay To Detect Antibodies Targeting Recombinant Envelope Protein 2 of Mayaro Virus. J Clin Microbiol. 2019; 57.
Article18). Rodrí guez-Rodrí guez M, Tello D, Yé lamos B, Gó mez-Gutié rrez J, Pacheco B, Ortega S, et al. Structural properties of the ectodomain of hepatitis C virus E2 envelope protein. Virus Res. 2009; 139:91–9.19). Powers AM, Brault AC, Shirako Y, Strauss EG, Kang W, Strauss JH, et al. Evolutionary relationships and systematics of the alphaviruses. J Virol. 2001; 75:10118–31.
Article20). Song C, Li J, Liu J, Liu Q. Simple sensitive rapid detection of Escherichia coli O157: H7 in food samples by label-free immunofluorescence strip sensor. Talanta. 2016; 156-157:42–7.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression and Evaluation of Chikungunya Virus E1 and E2 Envelope Proteins for Serodiagnosis of Chikungunya Virus Infection
- Expression and Characterization of Human Immunodeficiency Virus Type 1 Envelope Mutant Glycoproteins by Using Baculovirus Expression System
- Characterization of Monoclonal Antibody Specific for Hepatitis C Virus E2 Envelope Protein
- Western Blot Detection of Human Anti-Chikungunya Virus Antibody with Recombinant Envelope 2 Protein
- Expression of Open Reading Frame 5 Protein of Porcine Reproductive and Respiratory Syndrome Virus Using Semliki Forest Virus Expression System