Allergy Asthma Immunol Res.
2020 Mar;12(2):364-370. 10.4168/aair.2020.12.2.364.
Primary Nasal Epithelial Cells From Allergic and Non-allergic Individuals Show Comparable Barrier Function
- Affiliations
-
- 1Department of Otorhinolaryngology, Medical University of Vienna, Vienna, Austria. verena.niederberger@meduniwien.ac.at
- 2Department of Pathophysiology and Allergy Research, Center for Pathophysiology, Infectiology and Immunology, Medical University of Vienna, Vienna, Austria.
- 3NRC Institute of Immunology FMBA of Russia, Moscow, Russia.
- 4Laboratory for Immunopathology, Department of Clinical Immunology and Allergy, Sechenov First Moscow State Medical University, Moscow, Russia.
- KMID: 2468468
- DOI: http://doi.org/10.4168/aair.2020.12.2.364
Abstract
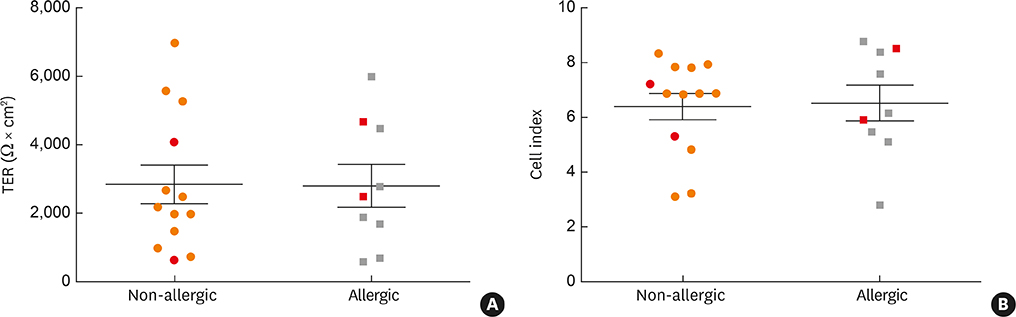
- Previous reports suggested that ex vivo cultured primary nasal epithelial cells from allergic patients differ from those from non-allergic individuals by genuinely reduced barrier function. By contrast, we found that primary nasal epithelial cells from allergic and non-allergic individuals showed comparable barrier function and secretion of cytokines.
Keyword
MeSH Terms
Figure
Reference
-
1. Egger C, Horak F, Vrtala S, Valenta R, Niederberger V. Nasal application of rBet v 1 or non-IgE-reactive T-cell epitope-containing rBet v 1 fragments has different effects on systemic allergen-specific antibody responses. J Allergy Clin Immunol. 2010; 126:1312–1315.e4.
Article2. Holgate ST. The sentinel role of the airway epithelium in asthma pathogenesis. Immunol Rev. 2011; 242:205–219.
Article3. Buckle FG, Cohen AB. Nasal mucosal hyperpermeability to macromolecules in atopic rhinitis and extrinsic asthma. J Allergy Clin Immunol. 1975; 55:213–221.
Article4. Steelant B, Seys SF, Van Gerven L, Van Woensel M, Farré R, Wawrzyniak P, et al. Histamine and T helper cytokine-driven epithelial barrier dysfunction in allergic rhinitis. J Allergy Clin Immunol. 2018; 141:951–963.e8.
Article5. Soyka MB, Wawrzyniak P, Eiwegger T, Holzmann D, Treis A, Wanke K, et al. Defective epithelial barrier in chronic rhinosinusitis: the regulation of tight junctions by IFN-γ and IL-4. J Allergy Clin Immunol. 2012; 130:1087–1096.e10.
Article6. Reisinger J, Triendl A, Küchler E, Bohle B, Krauth MT, Rauter I, et al. IFN-gamma-enhanced allergen penetration across respiratory epithelium augments allergic inflammation. J Allergy Clin Immunol. 2005; 115:973–981.7. Steelant B, Farré R, Wawrzyniak P, Belmans J, Dekimpe E, Vanheel H, et al. Impaired barrier function in patients with house dust mite-induced allergic rhinitis is accompanied by decreased occludin and zonula occludens-1 expression. J Allergy Clin Immunol. 2016; 137:1043–1053.e5.
Article8. Waltl EE, Selb R, Eckl-Dorna J, Mueller CA, Cabauatan CR, Eiwegger T, et al. Betamethasone prevents human rhinovirus- and cigarette smoke- induced loss of respiratory epithelial barrier function. Sci Rep. 2018; 8:9688.
Article9. van Hage M, Hamsten C, Valenta R. ImmunoCAP assays: pros and cons in allergology. J Allergy Clin Immunol. 2017; 140:974–977.
Article10. Lupinek C, Wollmann E, Baar A, Banerjee S, Breiteneder H, Broecker BM, et al. Advances in allergen-microarray technology for diagnosis and monitoring of allergy: the MeDALL allergen-chip. Methods. 2014; 66:106–119.
Article11. Kamekura R, Kojima T, Koizumi J, Ogasawara N, Kurose M, Go M, et al. Thymic stromal lymphopoietin enhances tight-junction barrier function of human nasal epithelial cells. Cell Tissue Res. 2009; 338:283–293.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Expression of Neurokinin 1 Receptor in Allergic Rhinitis
- In Vitro Culture of Human Nasal Epithelial Cells by Monolayer Culture of Dissociated Cells
- Expression of TARC in Nasal Epithelial Cells by IL-4/IL-13 and TNF-alpha in Allergic Rhinitis
- Upregulation of the Vitamin D Receptor in the Nasal Mucosa of Patients With Allergic Rhinitis
- Expression of NF-K B and I-K B in Allergic Rhinitis