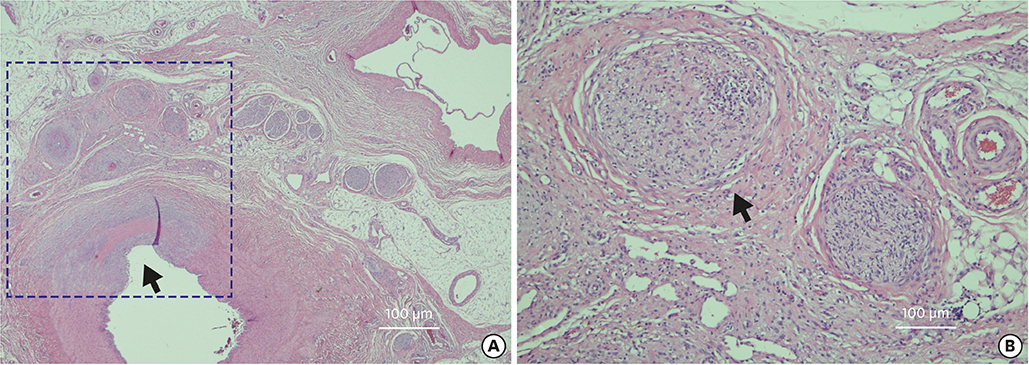
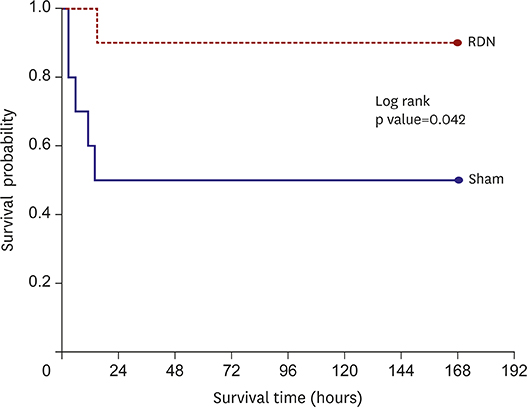
Korean Circ J.
2020 Jan;50(1):38-49. 10.4070/kcj.2019.0106.
Effect of Renal Denervation on Suppression of PVC and QT Prolongation in a Porcine Model of Acute Myocardial Infarction
- Affiliations
-
- 1Department of Cardiology, Chosun University Hospital, Gwangju, Korea.
- 2Department of Cardiovascular Medicine, Chonnam National University Medical School, Gwangju, Korea. mdhwp@naver.com, myungho@chollian.net
- 3Futuristic Animal Resource and Research Center, Korea Research Institute of Bioscience and Biotechnology, Ochang, Korea.
- 4Biomedical Research Institute, Chonnam National University Hospital, Gwangju, Korea.
- KMID: 2465662
- DOI: http://doi.org/10.4070/kcj.2019.0106
Abstract
- BACKGROUND AND OBJECTIVES
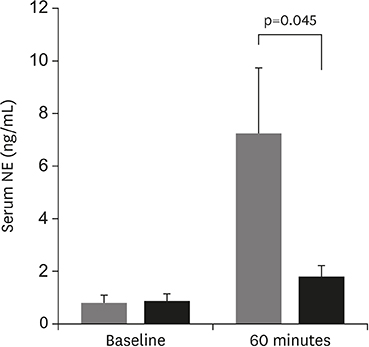
Antiarrhythmic effect of renal denervation (RDN) after acute myocardial infarction (AMI) remains unclear. The goal of this study was to evaluate the effect of RDN on ventricular arrhythmia (VA) after AMI in a porcine model.
METHODS
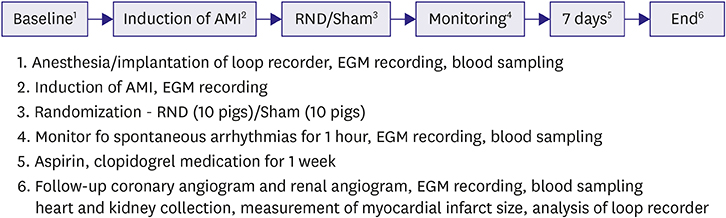
Twenty pigs were randomly divided into 2 groups based on RDN (RDN, n=10; Sham, n=10). After implanting a loop recorder, AMI was induced by occlusion of the middle left anterior descending coronary artery. Catheter-based RDN was performed for each renal artery immediately after creating AMI. Sham procedure used the same method, but a radiofrequency current was not delivered. Electrocardiography was monitored for 1 hour to observe VA. One week later, the animals were euthanized and the loop recorder data were analyzed.
RESULTS
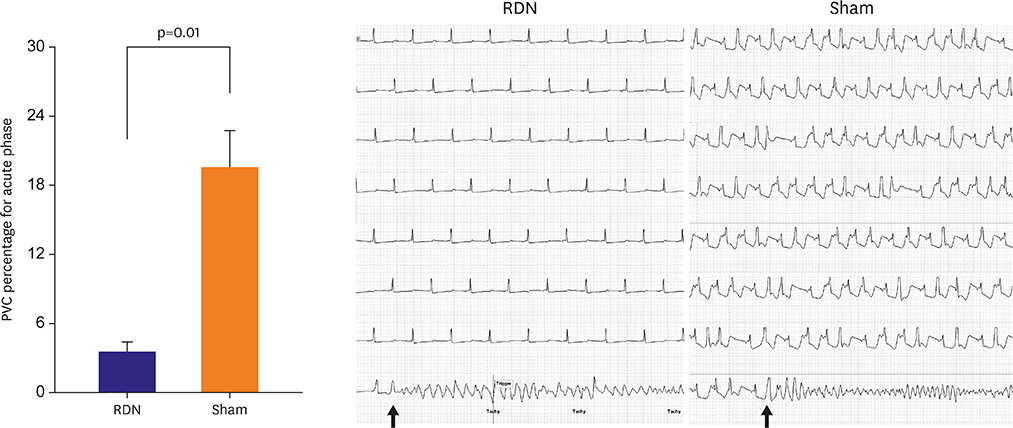
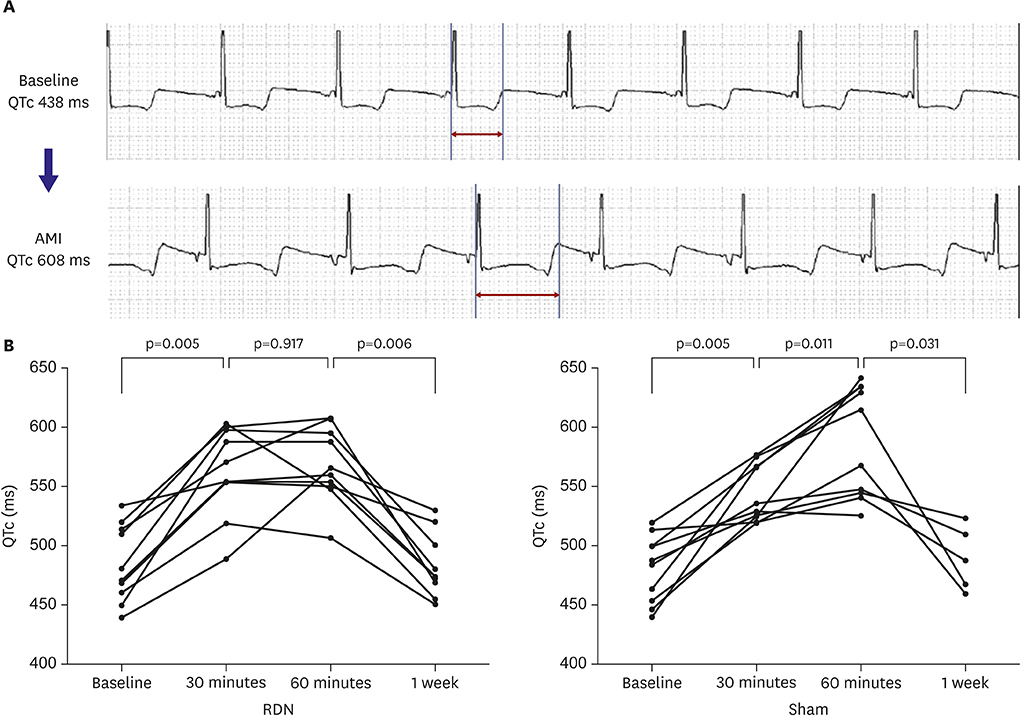
Ventricular fibrillation event rate and the interval from AMI creation to first VA in acute phase were not different between the 2 groups. However, the incidence of premature ventricular complex (PVC) was lower in the RDN than in the Sham. Additionally, RDN inhibited prolongation of the corrected QT (QTc) interval after AMI. The frequency of non-sustained or sustained ventricular tachycardia, arrhythmic death was lower in the RDN group in the early period.
CONCLUSIONS
RDN reduced the incidence of PVC, inhibited prolongation of the QTc interval, and reduced VA in the early period following an AMI. These results suggest that RDN might be a therapeutic option in patients with electrical instability after AMI.
MeSH Terms
Figure
Cited by 3 articles
-
Renal Denervation, Come Back Time?
Sang-Ho Jo
Korean Circ J. 2020;51(1):56-57. doi: 10.4070/kcj.2020.0479.An Open-label, Single-arm, Multicenter Feasibility Study Evaluating the Safety of Catheter-based Renal Denervation with DENEX™ in Patients with Uncontrolled Hypertension on Standard Medical Therapy
Chan Joon Kim, Kiyuk Chang, Byeong-Keuk Kim, Chang Gyu Park, Yangsoo Jang
Korean Circ J. 2020;51(1):43-55. doi: 10.4070/kcj.2020.0391.Is Renal Denervation Using Radiofrequency a Treatment Option for the Ventricular Arrhythmia Following Acute Myocardial Infarction?
Young Soo Lee
Korean Circ J. 2020;50(1):50-51. doi: 10.4070/kcj.2019.0361.
Reference
-
1. Schömig A. Catecholamines in myocardial ischemia. Systemic and cardiac release. Circulation. 1990; 82:II13–22.2. O'Brien E. Renal sympathetic denervation for resistant hypertension. Lancet. 2009; 373:2109–2110.3. Remo BF, Preminger M, Bradfield J, et al. Safety and efficacy of renal denervation as a novel treatment of ventricular tachycardia storm in patients with cardiomyopathy. Heart Rhythm. 2014; 11:541–546.
Article4. Hoffmann BA, Steven D, Willems S, Sydow K. Renal sympathetic denervation as an adjunct to catheter ablation for the treatment of ventricular electrical storm in the setting of acute myocardial infarction. J Cardiovasc Electrophysiol. 2013; 24:1175–1178.
Article5. Scholz EP, Raake P, Thomas D, Vogel B, Katus HA, Blessing E. Rescue renal sympathetic denervation in a patient with ventricular electrical storm refractory to endo- and epicardial catheter ablation. Clin Res Cardiol. 2015; 104:79–84.
Article6. Ukena C, Bauer A, Mahfoud F, et al. Renal sympathetic denervation for treatment of electrical storm: first-in-man experience. Clin Res Cardiol. 2012; 101:63–67.
Article7. Armaganijan LV, Staico R, Moreira DA, et al. 6-Month outcomes in patients with implantable cardioverter-defibrillators undergoing renal sympathetic denervation for the treatment of refractory ventricular arrhythmias. JACC Cardiovasc Interv. 2015; 8:984–990.
Article8. Kiuchi MG, Vitorio FP, da Silva GR, Paz LM, Souto GL. A case report of renal sympathetic denervation for the treatment of polymorphic ventricular premature complexes: expanding horizons. Medicine (Baltimore). 2015; 94:e2287.9. Bloch Thomsen PE, Jons C, Raatikainen MJ, et al. Long-term recording of cardiac arrhythmias with an implantable cardiac monitor in patients with reduced ejection fraction after acute myocardial infarction: the Cardiac Arrhythmias and Risk Stratification After Acute Myocardial Infarction (CARISMA) study. Circulation. 2010; 122:1258–1264.10. Jackson N, Gizurarson S, Azam MA, et al. Effects of renal artery denervation on ventricular arrhythmias in a postinfarct model. Circ Cardiovasc Interv. 2017; 10:e004172.
Article11. Sakakura K, Ladich E, Edelman ER, et al. Methodological standardization for the pre-clinical evaluation of renal sympathetic denervation. JACC Cardiovasc Interv. 2014; 7:1184–1193.12. Kaplinsky E, Ogawa S, Balke CW, Dreifus LS. Two periods of early ventricular arrhythmia in the canine acute myocardial infarction model. Circulation. 1979; 60:397–403.
Article13. Schwartz PJ, Stone HL, Brown AM. Effects of unilateral stellate ganglion blockade on the arrhythmias associated with coronary occlusion. Am Heart J. 1976; 92:589–599.
Article14. Linz D, Wirth K, Ukena C, et al. Renal denervation suppresses ventricular arrhythmias during acute ventricular ischemia in pigs. Heart Rhythm. 2013; 10:1525–1530.
Article15. Ruberman W, Weinblatt E, Goldberg JD, Frank CW, Shapiro S. Ventricular premature beats and mortality after myocardial infarction. N Engl J Med. 1977; 297:750–757.
Article16. Naito M, Michelson EL, Kaplinsky E, Dreifus LS, David D, Blenko TM. Role of early cycle ventricular extrasystoles in initiation of ventricular tachycardia and fibrillation: evaluation of the R on T phenomenon during acute ischemia in a canine model. Am J Cardiol. 1982; 49:317–322.
Article17. Janse MJ, Kleber AG, Capucci A, Coronel R, Wilms-Schopman F. Electrophysiological basis for arrhythmias caused by acute ischemia. Role of the subendocardium. J Mol Cell Cardiol. 1986; 18:339–355.18. Katra RP, Laurita KR. Cellular mechanism of calcium-mediated triggered activity in the heart. Circ Res. 2005; 96:535–542.
Article19. Curran J, Hinton MJ, Ríos E, Bers DM, Shannon TR. Beta-adrenergic enhancement of sarcoplasmic reticulum calcium leak in cardiac myocytes is mediated by calcium/calmodulin-dependent protein kinase. Circ Res. 2007; 100:391–398.20. Kenigsberg DN, Khanal S, Kowalski M, Krishnan SC. Prolongation of the QTc interval is seen uniformly during early transmural ischemia. J Am Coll Cardiol. 2007; 49:1299–1305.
Article21. Zhou S, Cao JM, Tebb ZD, et al. Modulation of QT interval by cardiac sympathetic nerve sprouting and the mechanisms of ventricular arrhythmia in a canine model of sudden cardiac death. J Cardiovasc Electrophysiol. 2001; 12:1068–1073.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Dispersion of QT Interval and Other Repolarization Indexes in Acute Myocardial Infarction
- Is Renal Denervation Using Radiofrequency a Treatment Option for the Ventricular Arrhythmia Following Acute Myocardial Infarction?
- A Case of Ofloxacin-induced Torsades de Pointes and Abnormal ECG Change Mimicking Acute Myocardial Infarction
- Clinical Experiences about Effect of IV Disopyramide Phosphate Injection on PVC
- The Effects of Trimetazidine on the Enhancement Patterns of Multi-Detector Computed Tomography in a Porcine Myocardial Infarction Model: What is the Meaning of the MDCT Enhancement Pattern in this Myocardial Infarction Model?