J Liver Cancer.
2019 Sep;19(2):136-142. 10.17998/jlc.19.2.136.
Radiation-induced Myositis after Proton Beam Therapy to Huge Hepatocellular Carcinoma
- Affiliations
-
- 1Department of Medicine, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea.
- 2Department of Radiation Oncology, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. hee.ro.park@samsung.com
- KMID: 2463614
- DOI: http://doi.org/10.17998/jlc.19.2.136
Abstract
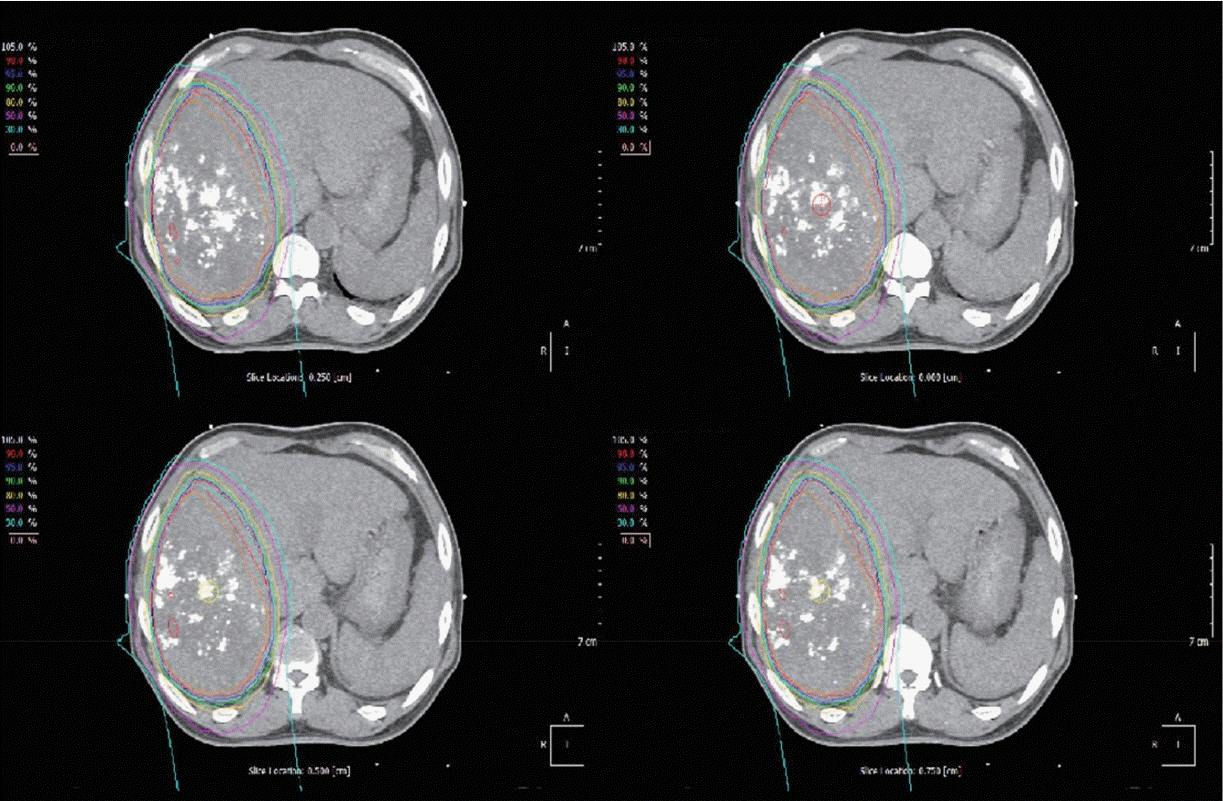
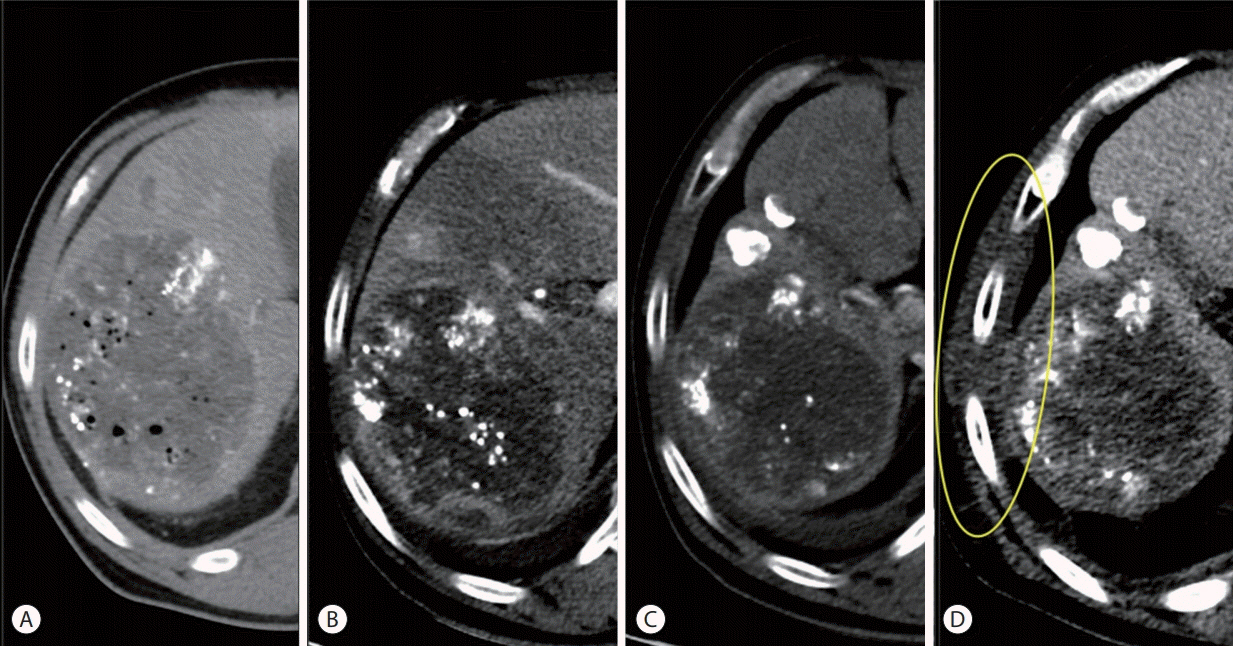
- Proton beam therapy (PBT) is one of the advances in radiotherapy techniques, which enables dose escalation with lower probability of radiation-induced liver or gastrointestinal injuries. However, the chest wall proximal to the tumor can be affected by high dose irradiation. Here, we report on a 58-year-old male patient who presented with huge hepatocellular carcinoma, received treatment with transarterial chemoembolization and PBT, and developed severe chest wall pain due to radiation-induced myositis. The patient's symptoms were controlled by oral steroids.
Keyword
MeSH Terms
Figure
Reference
-
1. Yoo GS, Yu JI, Park HC. Proton therapy for hepatocellular carcinoma: current knowledges and future perspectives. World J Gastroenterol. 2018; 24:3090–3100.2. Klein J, Dawson LA. Hepatocellular carcinoma radiation therapy: review of evidence and future opportunities. Int J Radiat Oncol Biol Phys. 2013; 87:22–32.3. Choi SH, Seong J. Strategic application of radiotherapy for hepatocellular carcinoma. Clin Mol Hepatol. 2018; 24:114–134.4. Kanemoto A, Mizumoto M, Okumura T, Takahashi H, Hashimoto T, Oshiro Y, et al. Dose-volume histogram analysis for risk factors of radiation-induced rib fracture after hypofractionated proton beam therapy for hepatocellular carcinoma. Acta Oncol. 2013; 52:538–544.5. Yeung R, Bowen SR, Chapman TR, MacLennan GT, Apisarnthanarax S. Chest wall toxicity after hypofractionated proton beam therapy for liver malignancies. Pract Radiat Oncol. 2018; 8:287–293.6. Welsh JS, Torre TG, DeWeese TL, O'Reilly S. Radiation myositis. Ann Oncol. 1999; 10:1105–1108.7. Gillette EL, Mahler PA, Powers BE, Gillette SM, Vujaskovic Z. Late radiation injury to muscle and peripheral nerves. Int J Radiat Oncol Biol Phys. 1995; 31:1309–1318.8. Lockney DT, Jia AY, Lis E, Lockney NA, Liu C, Hopkins B, et al. Myositis following spine radiosurgery for metastatic disease: a case series. J Neurosurg Spine. 2018; 28:416–421.9. Borroni G, Vassallo C, Brazzelli V, Martinoli S, Ardigò M, Alessandrino PE, et al. Radiation recall dermatitis, panniculitis, and myositis following cyclophosphamide therapy: histopathologic findings of a patient affected by multiple myeloma. Am J Dermatopathol. 2004; 26:213–216.10. Burstein HJ. Side effects of chemotherapy. Case 1. Radiation recall dermatitis from gemcitabine. J Clin Oncol. 2000; 18:693–694.11. Maeng CH, Park JS, Lee SA, Kim DH, Yun DH, Yoo SD, et al. Radiation recall phenomenon presenting as myositis triggered by carboplatin plus paclitaxel and related literature review. J Cancer Res Ther. 2014; 10:1093–1097.12. Shrimali RK, McPhail NJ, Correa PD, Fraser J, Rizwanullah M. Trastuzumab-induced radiation recall dermatitis--first reported case. Clin Oncol (R Coll Radiol). 2009; 21:634–635.13. Kang SK. Images in clinical medicine. Radiation recall reaction after antimicrobial therapy. N Engl J Med. 2006; 354:622.14. Phillips TL, Fu KK. Quantification of combined radiation therapy and chemotherapy effects on critical normal tissues. Cancer. 1976; 37(2 Suppl):1186–1200.15. Kim GE, Song HS, Ahn KJ, Kim YS. Radiation recall dermatitis triggered by sorafenib after radiation therapy for hepatocellular carcinoma. Radiat Oncol J. 2017; 35:289–294.16. Mehta K, Kaubisch A, Tang J, Pirlamarla A, Kalnicki S. Radiation recall dermatitis in patients treated with sorafenib. Case Rep Oncol Med. 2018; 2018:2171062.17. Smitaman E, Flores DV, Mejía Gómez C, Pathria MN. MR imaging of atraumatic muscle disorders. Radiographics. 2018; 38:500–522.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Current role of proton beam therapy in patients with hepatocellular carcinoma
- A Case of Hepatocellular Carcinoma Treated by Proton Beam Therapy as 1st Choice
- Proton Therapy Review: Proton Therapy from a Medical
- Novel paradigm in the treatment of hepatocellular carcinoma: Anticipating breakthroughs with particle therapy
- Current evidence and the potential role of proton beam therapy for hepatocellular carcinoma