Allergy Asthma Immunol Res.
2020 Jan;12(1):24-41. 10.4168/aair.2020.12.1.24.
Critical Points on the Use of Biologicals in Allergic Diseases and Asthma
- Affiliations
-
- 1Department of Allergy and Clinical Immunology, Faculty of Medicine, Transylvania University of Brasov, Brasov, Romania. ibrumaru@unitbv.ro
- KMID: 2462570
- DOI: http://doi.org/10.4168/aair.2020.12.1.24
Abstract
- Improved understanding of the contribution of immune-inflammatory mechanisms in allergic diseases and asthma has encouraged development of biologicals and small molecules specifically targeting the innate and adaptive immune response. There are several critical points impacting the efficacy of this stratified approach, from the complexity of disease endotypes to the effectiveness in real-world settings. We discuss here how these barriers can be overcome to facilitate the development of implementation science for allergic diseases and asthma.
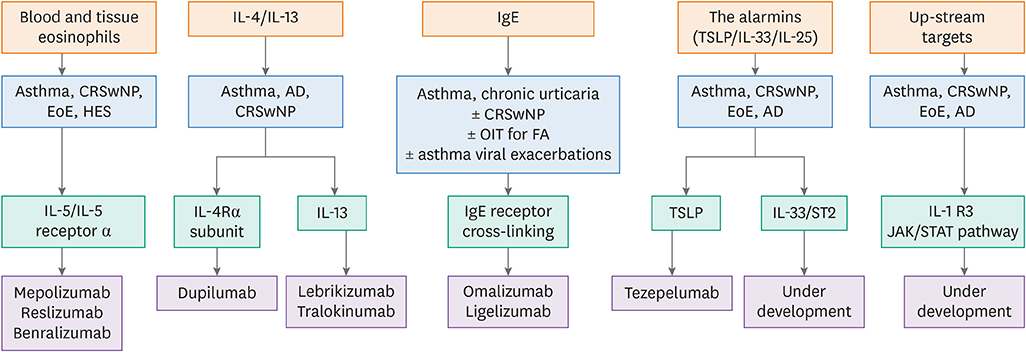
Figure
Reference
-
1. Agache I. Severe asthma phenotypes and endotypes. Semin Immunol. 2019; 101301:101301.
Article2. Agache I, Akdis CA. Precision medicine and phenotypes, endotypes, genotypes, regiotypes, and theratypes of allergic diseases. J Clin Invest. 2019; 130:1493–1503.
Article3. Agache I, Akdis CA. Endotypes of allergic diseases and asthma: an important step in building blocks for the future of precision medicine. Allergol Int. 2016; 65:243–252.
Article4. Agache I, Miller R, Gern JE, Hellings PW, Jutel M, Muraro A, et al. Emerging concepts and challenges in implementing the exposome paradigm in allergic diseases and asthma: a Practall document. Allergy. 2019; 74:449–463.
Article5. Agache I, Rogozea L. Asthma biomarkers: do they bring precision medicine closer to the clinic? Allergy Asthma Immunol Res. 2017; 9:466–476.
Article6. Agache I, Annesi-Maesano I, Bonertz A, Branca F, Cant A, Fras Z, et al. Prioritizing research challenges and funding for allergy and asthma and the need for translational research-the European Strategic Forum on Allergic Diseases. Allergy. 2019; 74:2064–2076.
Article7. Ravin KA, Loy M. The eosinophil in infection. Clin Rev Allergy Immunol. 2016; 50:214–227.
Article8. Jacobsen EA, Helmers RA, Lee JJ, Lee NA. The expanding role(s) of eosinophils in health and disease. Blood. 2012; 120:3882–3890.
Article9. Marichal T, Mesnil C, Bureau F. Homeostatic eosinophils: characteristics and functions. Front Med (Lausanne). 2017; 4:101.
Article10. Mesnil C, Raulier S, Paulissen G, Xiao X, Birrell MA, Pirottin D, et al. Lung-resident eosinophils represent a distinct regulatory eosinophil subset. J Clin Invest. 2016; 126:3279–3295.
Article11. Takatsu K. Interleukin-5 and IL-5 receptor in health and diseases. Proc Jpn Acad, Ser B, Phys Biol Sci. 2011; 87:463–485.
Article12. Molfino NA, Gossage D, Kolbeck R, Parker JM, Geba GP. Molecular and clinical rationale for therapeutic targeting of interleukin-5 and its receptor. Clin Exp Allergy. 2012; 42:712–737.
Article13. Gorski SA, Lawrence MG, Hinkelman A, Spano MM, Steinke JW, Borish L, et al. Expression of IL-5 receptor alpha by murine and human lung neutrophils. PLoS One. 2019; 14:e0221113.
Article14. Dent LA, Strath M, Mellor AL, Sanderson CJ. Eosinophilia in transgenic mice expressing interleukin 5. J Exp Med. 1990; 172:1425–1431.
Article15. Sridhar S, Liu H, Pham TH, Damera G, Newbold P. Modulation of blood inflammatory markers by benralizumab in patients with eosinophilic airway diseases. Respir Res. 2019; 20:14.
Article16. Agache IO. From phenotypes to endotypes to asthma treatment. Curr Opin Allergy Clin Immunol. 2013; 13:249–256.
Article17. Wang FP, Liu T, Lan Z, Li SY, Mao H. Efficacy and safety of anti-interleukin-5 therapy in patients with asthma: a systematic review and meta-analysis. PLoS One. 2016; 11:e0166833.
Article18. Haldar P, Brightling CE, Singapuri A, Hargadon B, Gupta S, Monteiro W, et al. Outcomes after cessation of mepolizumab therapy in severe eosinophilic asthma: a 12-month follow-up analysis. J Allergy Clin Immunol. 2014; 133:921–923.19. Kim YJ, Prussin C, Martin B, Law MA, Haverty TP, Nutman TB, et al. Rebound eosinophilia after treatment of hypereosinophilic syndrome and eosinophilic gastroenteritis with monoclonal anti-IL-5 antibody SCH55700. J Allergy Clin Immunol. 2004; 114:1449–1455.
Article20. Mukherjee M, Aleman Paramo F, Kjarsgaard M, Salter B, Nair G, LaVigne N, et al. Weight-adjusted intravenous reslizumab in severe asthma with inadequate response to fixed-dose subcutaneous mepolizumab. Am J Respir Crit Care Med. 2018; 197:38–46.
Article21. Mukherjee M, Lim HF, Thomas S, Miller D, Kjarsgaard M, Tan B, et al. Airway autoimmune responses in severe eosinophilic asthma following low-dose Mepolizumab therapy. Allergy Asthma Clin Immunol. 2017; 13:2.
Article22. Kelly EA, Esnault S, Liu LY, Evans MD, Johansson MW, Mathur S, et al. Mepolizumab attenuates airway eosinophil numbers, but not their functional phenotype in asthma. Am J Respir Crit Care Med. 2017; 196:1385–1395.
Article23. Lim HF, Nair P. Efficacy and safety of reslizumab in patients with moderate to severe eosinophilic asthma. Expert Rev Respir Med. 2015; 9:135–142.
Article24. Sehmi R, Lim HF, Mukherjee M, Huang C, Radford K, Newbold P, et al. Benralizumab attenuates airway eosinophilia in prednisone-dependent asthma. J Allergy Clin Immunol. 2018; 141:1529–1532.e8.
Article25. Laviolette M, Gossage DL, Gauvreau G, Leigh R, Olivenstein R, Katial R, et al. Effects of benralizumab on airway eosinophils in asthmatic patients with sputum eosinophilia. J Allergy Clin Immunol. 2013; 132:1086–1096.e5.
Article26. Liu LY, Sedgwick JB, Bates ME, Vrtis RF, Gern JE, Kita H, et al. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: I. Loss of membrane IL-5 receptor alpha on airway eosinophils and increased soluble IL-5 receptor alpha in the airway after allergen challenge. J Immunol. 2002; 169:6452–6458.27. Liu LY, Sedgwick JB, Bates ME, Vrtis RF, Gern JE, Kita H, et al. Decreased expression of membrane IL-5 receptor alpha on human eosinophils: II. IL-5 down-modulates its receptor via a proteinase-mediated process. J Immunol. 2002; 169:6459–6466.28. Stein ML, Villanueva JM, Buckmeier BK, Yamada Y, Filipovich AH, Assa’ad AH, et al. Anti-IL-5 (mepolizumab) therapy reduces eosinophil activation ex vivo and increases IL-5 and IL-5 receptor levels. J Allergy Clin Immunol. 2008; 121:1473–1483. 1483.e1–1474.
Article29. Kolbeck R, Kozhich A, Koike M, Peng L, Andersson CK, Damschroder MM, et al. MEDI-563, a humanized anti-IL-5 receptor alpha mAb with enhanced antibody-dependent cell-mediated cytotoxicity function. J Allergy Clin Immunol. 2010; 125:1344–1353.e2.30. Wright AK, Weston C, Rana BM, Brightling CE, Cousins DJ. Human group 2 innate lymphoid cells do not express the IL-5 receptor. J Allergy Clin Immunol. 2017; 140:1430–1433.e4.
Article31. Smith SG, Chen R, Kjarsgaard M, Huang C, Oliveria JP, O’Byrne PM, et al. Increased numbers of activated group 2 innate lymphoid cells in the airways of patients with severe asthma and persistent airway eosinophilia. J Allergy Clin Immunol. 2016; 137:75–86.e8.
Article32. Gandhi NA, Pirozzi G, Graham NM. Commonality of the IL-4/IL-13 pathway in atopic diseases. Expert Rev Clin Immunol. 2017; 13:425–437.
Article33. Jonstam K, Swanson BN, Mannent LP, Cardell LO, Tian N, Wang Y, et al. Dupilumab reduces local type 2 pro-inflammatory biomarkers in chronic rhinosinusitis with nasal polyposis. Allergy. 2019; 74:743–752.
Article34. Furue K, Ito T, Tsuji G, Ulzii D, Vu YH, Kido-Nakahara M, et al. The IL-13-OVOL1-FLG axis in atopic dermatitis. Immunology. Forthcoming. 2019.35. Callewaert C, Nakatsuji T, Knight R, Kosciolek T, Vrbanac A, Kotol P, et al. IL-4Rα blockade by dupilumab decreases Staphylococcus aureus colonization and increases microbial diversity in atopic dermatitis. J Invest Dermatol. Forthcoming. 2019.36. Guttman-Yassky E, Bissonnette R, Ungar B, Suárez-Fariñas M, Ardeleanu M, Esaki H, et al. Dupilumab progressively improves systemic and cutaneous abnormalities in patients with atopic dermatitis. J Allergy Clin Immunol. 2019; 143:155–172.
Article37. León B, Lund FE. Compartmentalization of dendritic cell and T-cell interactions in the lymph node: anatomy of T-cell fate decisions. Immunol Rev. 2019; 289:84–100.
Article38. Prout MS, Kyle RL, Ronchese F, Le Gros G. IL-4 Is a key requirement for IL-4- and IL-4/IL-13-expressing CD4 Th2 subsets in lung and skin. Front Immunol. 2018; 9:1211.
Article39. Kim JE, Jung K, Kim JA, Kim SH, Park HS, Kim YS. Engineering of anti-human interleukin-4 receptor alpha antibodies with potent antagonistic activity. Sci Rep. 2019; 9:7772.
Article40. Goh YP, Henderson NC, Heredia JE, Red Eagle A, Odegaard JI, Lehwald N, et al. Eosinophils secrete IL-4 to facilitate liver regeneration. Proc Natl Acad Sci U S A. 2013; 110:9914–9919.
Article41. Weng SY, Wang X, Vijayan S, Tang Y, Kim YO, Padberg K, et al. IL-4 Receptor alpha signaling through macrophages differentially regulates liver fibrosis progression and reversal. EBioMedicine. 2018; 29:92–103.
Article42. Mastroianni CM, Lichtner M, Citton R, Del Borgo C, Rago A, Martini H, et al. Current trends in management of hepatitis B virus reactivation in the biologic therapy era. World J Gastroenterol. 2011; 17:3881–3887.
Article43. Palomares Ó, Sánchez-Ramón S, Dávila I, Prieto L, Pérez de Llano L, Lleonart M, et al. dIvergEnt: how IgE axis contributes to the continuum of allergic asthma and anti-IgE therapies. Int J Mol Sci. 2017; 18:E1328.
Article44. Anto JM, Bousquet J, Akdis M, Auffray C, Keil T, Momas I, et al. Mechanisms of the Development of Allergy (MeDALL): introducing novel concepts in allergy phenotypes. J Allergy Clin Immunol. 2017; 139:388–399.45. Wilcock LK, Francis JN, Durham SR. IgE-facilitated antigen presentation: role in allergy and the influence of allergen immunotherapy. Immunol Allergy Clin North Am. 2006; 26:333–347.
Article46. Gill MA, Liu AH, Calatroni A, Krouse RZ, Shao B, Schiltz A, et al. Enhanced plasmacytoid dendritic cell antiviral responses after omalizumab. J Allergy Clin Immunol. 2018; 141:1735–1743.e9.
Article47. Teach SJ, Gill MA, Togias A, Sorkness CA, Arbes SJ Jr, Calatroni A, et al. Preseasonal treatment with either omalizumab or an inhaled corticosteroid boost to prevent fall asthma exacerbations. J Allergy Clin Immunol. 2015; 136:1476–1485.
Article48. Martinez-Gonzalez I, Steer CA, Takei F. Lung ILC2s link innate and adaptive responses in allergic inflammation. Trends Immunol. 2015; 36:189–195.
Article49. Roan F, Obata-Ninomiya K, Ziegler SF. Epithelial cell-derived cytokines: more than just signaling the alarm. J Clin Invest. 2019; 129:1441–1451.
Article50. Morita H, Moro K, Koyasu S. Innate lymphoid cells in allergic and nonallergic inflammation. J Allergy Clin Immunol. 2016; 138:1253–1264.
Article51. Kim HJ, Lee SH, Jeong S, Hong SJ. Protease-activated receptors 2-antagonist suppresses asthma by inhibiting reactive oxygen species-thymic stromal lymphopoietin inflammation and epithelial tight junction degradation. Allergy Asthma Immunol Res. 2019; 11:560–571.
Article52. Varricchi G, Pecoraro A, Marone G, Criscuolo G, Spadaro G, Genovese A, et al. Thymic stromal lymphopoietin isoforms, inflammatory disorders, and cancer. Front Immunol. 2018; 9:1595.
Article53. Anderson EL, Kobayashi T, Iijima K, Bartemes KR, Chen CC, Kita H. IL-33 mediates reactive eosinophilopoiesis in response to airborne allergen exposure. Allergy. 2016; 71:977–988.
Article54. Johnston LK, Hsu CL, Krier-Burris RA, Chhiba KD, Chien KB, McKenzie A, et al. IL-33 precedes IL-5 in regulating eosinophil commitment and is required for eosinophil homeostasis. J Immunol. 2016; 197:3445–3453.
Article55. Angulo EL, McKernan EM, Fichtinger PS, Mathur SK. Comparison of IL-33 and IL-5 family mediated activation of human eosinophils. PLoS One. 2019; 14:e0217807.
Article56. Altman MC, Lai Y, Nolin JD, Long S, Chen CC, Piliponsky AM, et al. Airway epithelium-shifted mast cell infiltration regulates asthmatic inflammation via IL-33 signaling. J Clin Invest. 2019; 129:4979–4991.
Article57. Corren J, Parnes JR, Wang L, Mo M, Roseti SL, Griffiths JM, et al. Tezepelumab in adults with uncontrolled asthma. N Engl J Med. 2017; 377:936–946.
Article58. Højen JF, Kristensen ML, McKee AS, Wade MT, Azam T, Lunding LP, et al. IL-1R3 blockade broadly attenuates the functions of six members of the IL-1 family, revealing their contribution to models of disease. Nat Immunol. 2019; 20:1138–1149.
Article59. Zak M, Hanan EJ, Lupardus P, Brown DG, Robinson C, Siu M, et al. Discovery of a class of highly potent Janus kinase 1/2 (JAK1/2) inhibitors demonstrating effective cell-based blockade of IL-13 signaling. Bioorg Med Chem Lett. 2019; 29:1522–1531.
Article60. Fragoulis GE, McInnes IB, Siebert S. JAK-inhibitors. New players in the field of immune-mediated diseases, beyond rheumatoid arthritis. Rheumatology (Oxford). 2019; 58:i43–i54.
Article61. Pavel AB, Song T, Kim HJ, Del Duca E, Krueger JG, Dubin C, et al. Oral Janus kinase/SYK inhibition (ASN002) suppresses inflammation and improves epidermal barrier markers in patients with atopic dermatitis. J Allergy Clin Immunol. 2019; 144:1011–1024.
Article62. Calbet M, Ramis I, Calama E, Carreño C, Paris S, Maldonado M, et al. Novel inhaled pan-JAK inhibitor, LAS194046, reduces allergen-induced airway inflammation, late asthmatic response, and pSTAT activation in brown Norway rats. J Pharmacol Exp Ther. 2019; 370:137–147.
Article63. Wenzel S, Ford L, Pearlman D, Spector S, Sher L, Skobieranda F, et al. Dupilumab in persistent asthma with elevated eosinophil levels. N Engl J Med. 2013; 368:2455–2466.
Article64. Katsaounou P, Buhl R, Brusselle G, Pfister P, Martínez R, Wahn U, et al. Omalizumab as alternative to chronic use of oral corticosteroids in severe asthma. Respir Med. 2019; 150:51–62.
Article65. Chipps BE, Newbold P, Hirsch I, Trudo F, Goldman M. Benralizumab efficacy by atopy status and serum immunoglobulin E for patients with severe, uncontrolled asthma. Ann Allergy Asthma Immunol. 2018; 120:504–511.e4.
Article66. Corren J, Castro M, O'Riordan T, Hanania NA, Pavord ID, Quirce S, et al. Dupilumab efficacy in patients with uncontrolled, moderate-to-severe allergic asthma. J Allergy Clin Immunol Pract. Forthcoming. 2019.67. Weinstein SF, Katial R, Jayawardena S, Pirozzi G, Staudinger H, Eckert L, et al. Efficacy and safety of dupilumab in perennial allergic rhinitis and comorbid asthma. J Allergy Clin Immunol. 2018; 142:171–177.e1.
Article68. Vignola AM, Humbert M, Bousquet J, Boulet LP, Hedgecock S, Blogg M, et al. Efficacy and tolerability of anti-immunoglobulin E therapy with omalizumab in patients with concomitant allergic asthma and persistent allergic rhinitis: SOLAR. Allergy. 2004; 59:709–717.
Article69. Fiocchi A, Artesani MC, Riccardi C, Mennini M, Pecora V, Fierro V, et al. Impact of omalizumab on food allergy in patients treated for asthma: a real-life study. J Allergy Clin Immunol Pract. 2019; 7:1901–1909.e5.
Article70. Bousquet J, Rao S, Manga V. Global evaluation of treatment effectiveness (GETE) is an accurate predictor of response to omalizumab in patients with severe allergic asthma: a pooled analysis. Eur Respir J. 2014; 44:3483.71. Bateman ED, Djukanović R, Castro M, Canvin J, Germinaro M, Noble R, et al. Predicting responders to reslizumab after 16 weeks of treatment using an algorithm derived from clinical studies of patients with severe eosinophilic asthma. Am J Respir Crit Care Med. 2019; 199:489–495.
Article72. Bachert C, Hellings PW, Mullol J, Hamilos DL, Gevaert P, Naclerio RM, et al. Dupilumab improves health-related quality of life in patients with chronic rhinosinusitis with nasal polyposis. Allergy. Forthcoming. 2019.
Article73. Bachert C, Sousa AR, Lund VJ, Scadding GK, Gevaert P, Nasser S, et al. Reduced need for surgery in severe nasal polyposis with mepolizumab: randomized trial. J Allergy Clin Immunol. 2017; 140:1024–1031.e14.
Article74. Bachert C, Mannent L, Naclerio RM, Mullol J, Ferguson BJ, Gevaert P, et al. Effect of subcutaneous dupilumab on nasal polyp burden in patients with chronic sinusitis and nasal polyposis: a randomized clinical trial. JAMA. 2016; 315:469–479.75. Gevaert P, Calus L, Van Zele T, Blomme K, De Ruyck N, Bauters W, et al. Omalizumab is effective in allergic and nonallergic patients with nasal polyps and asthma. J Allergy Clin Immunol. 2013; 131:110–116.e1.
Article76. Kartush AG, Schumacher JK, Shah R, Patadia MO. Biologic agents for the treatment of chronic rhinosinusitis with nasal polyps. Am J Rhinol Allergy. 2019; 33:203–211.
Article77. Tsetsos N, Goudakos JK, Daskalakis D, Konstantinidis I, Markou K. Monoclonal antibodies for the treatment of chronic rhinosinusitis with nasal polyposis: a systematic review. Rhinology. 2018; 56:11–21.
Article78. Jung HJ, Zhang YL, Kim DK, Rhee CS, Kim DY. The role of NF-κB in chronic rhinosinusitis with nasal polyps. Allergy Asthma Immunol Res. 2019; 11:806–817.
Article79. Seger EW, Wechter T, Strowd L, Feldman SR. Relative efficacy of systemic treatments for atopic dermatitis. J Am Acad Dermatol. 2019; 80:411–416.e4.
Article80. Thaçi D, L Simpson E, Deleuran M, Kataoka Y, Chen Z, Gadkari A, et al. Efficacy and safety of dupilumab monotherapy in adults with moderate-to-severe atopic dermatitis: a pooled analysis of two phase 3 randomized trials (LIBERTY AD SOLO 1 and LIBERTY AD SOLO 2). J Dermatol Sci. 2019; 94:266–275.
Article81. Simpson EL, Bieber T, Guttman-Yassky E, Beck LA, Blauvelt A, Cork MJ, et al. Two phase 3 trials of dupilumab versus placebo in atopic dermatitis. N Engl J Med. 2016; 375:2335–2348.
Article82. Blauvelt A, Rosmarin D, Bieber T, Simpson EL, Bagel J, Worm M, et al. Improvement of atopic dermatitis with dupilumab occurs equally well across different anatomical regions: data from phase III clinical trials. Br J Dermatol. 2019; 181:196–197.83. Alexis AF, Rendon M, Silverberg JI, Pariser DM, Lockshin B, Griffiths CE, et al. Efficacy of dupilumab in different racial subgroups of adults with moderate-to-severe atopic dermatitis in three randomized, placebo-controlled phase 3 trials. J Drugs Dermatol. 2019; 18:804–813.84. Wollenberg A, Beck LA, Blauvelt A, Simpson EL, Chen Z, Chen Q, et al. Laboratory safety of dupilumab in moderate-to-severe atopic dermatitis: results from three phase III trials (LIBERTY AD SOLO 1, LIBERTY AD SOLO 2, LIBERTY AD CHRONOS). Br J Dermatol. Forthcoming. 2019.
Article85. Simpson EL, Flohr C, Eichenfield LF, Bieber T, Sofen H, Taïeb A, et al. Efficacy and safety of lebrikizumab (an anti-IL-13 monoclonal antibody) in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical corticosteroids: A randomized, placebo-controlled phase II trial (TREBLE). J Am Acad Dermatol. 2018; 78:863–871.e11.
Article86. Wollenberg A, Howell MD, Guttman-Yassky E, Silverberg JI, Kell C, Ranade K, et al. Treatment of atopic dermatitis with tralokinumab, an anti-IL-13 mAb. J Allergy Clin Immunol. 2019; 143:135–141.
Article87. Simpson EL, Parnes JR, She D, Crouch S, Rees W, Mo M, et al. Tezepelumab, an anti-thymic stromal lymphopoietin monoclonal antibody, in the treatment of moderate to severe atopic dermatitis: a randomized phase 2a clinical trial. J Am Acad Dermatol. 2019; 80:1013–1021.
Article88. Bosma AL, Spuls PI, Garcia-Doval I, Naldi L, Prieto-Merino D, Tesch F, et al. TREatment of ATopic eczema (TREAT) Registry Taskforce: protocol for a European safety study of dupilumab and other systemic therapies in patients with atopic eczema. Br J Dermatol. Forthcoming. 2019.
Article89. Ertas R, Ozyurt K, Atasoy M, Hawro T, Maurer M. The clinical response to omalizumab in chronic spontaneous urticaria patients is linked to and predicted by IgE levels and their change. Allergy. 2018; 73:705–712.
Article90. Weller K, Ohanyan T, Hawro T, Ellrich A, Sussman G, Koplowitz J, et al. Total IgE levels are linked to the response of chronic spontaneous urticaria patients to omalizumab. Allergy. 2018; 73:2406–2408.
Article91. Ertaş R, Hawro T, Altrichter S, Özyurt K, Erol K, Ketenci Ertaş Ş, et al. Antinuclear antibodies are common and linked to poor response to omalizumab treatment in patients with CSU. Allergy. Forthcoming. 2019.
Article92. Metz M, Torene R, Kaiser S, Beste MT, Staubach P, Bauer A, et al. Omalizumab normalizes the gene expression signature of lesional skin in patients with chronic spontaneous urticaria: A randomized, double-blind, placebo-controlled study. Allergy. 2019; 74:141–151.
Article93. Min TK, Saini SS. Emerging therapies in chronic spontaneous urticaria. Allergy Asthma Immunol Res. 2019; 11:470–481.
Article94. Wood RA, Kim JS, Lindblad R, Nadeau K, Henning AK, Dawson P, et al. A randomized, double-blind, placebo-controlled study of omalizumab combined with oral immunotherapy for the treatment of cow's milk allergy. J Allergy Clin Immunol. 2016; 137:1103–1110.e11.
Article95. MacGinnitie AJ, Rachid R, Gragg H, Little SV, Lakin P, Cianferoni A, et al. Omalizumab facilitates rapid oral desensitization for peanut allergy. J Allergy Clin Immunol. 2017; 139:873–881.e8.
Article96. Yee CS, Albuhairi S, Noh E, El-Khoury K, Rezaei S, Abdel-Gadir A, et al. Long-term outcome of peanut oral immunotherapy facilitated initially by omalizumab. J Allergy Clin Immunol Pract. 2019; 7:451–461.e7.
Article97. Loizou D, Enav B, Komlodi-Pasztor E, Hider P, Kim-Chang J, Noonan L, et al. A pilot study of omalizumab in eosinophilic esophagitis. PLoS One. 2015; 10:e0113483.
Article98. Markowitz JE, Jobe L, Miller M, Frost C, Laney Z, Eke R. Safety and efficacy of reslizumab for children and adolescents with eosinophilic esophagitis treated for 9 years. J Pediatr Gastroenterol Nutr. 2018; 66:893–897.
Article99. Otani IM, Anilkumar AA, Newbury RO, Bhagat M, Beppu LY, Dohil R, et al. Anti-IL-5 therapy reduces mast cell and IL-9 cell numbers in pediatric patients with eosinophilic esophagitis. J Allergy Clin Immunol. 2013; 131:1576–1582.
Article100. Rothenberg ME, Wen T, Greenberg A, Alpan O, Enav B, Hirano I, et al. Intravenous anti-IL-13 mAb QAX576 for the treatment of eosinophilic esophagitis. J Allergy Clin Immunol. 2015; 135:500–507.
Article101. Hirano I, Collins MH, Assouline-Dayan Y, Evans L, Gupta S, Schoepfer AM, et al. RPC4046, a monoclonal antibody against IL13, reduces histologic and endoscopic activity in patients with eosinophilic esophagitis. Gastroenterology. 2019; 156:592–603.e10.102. Nhu QM, Chiao H, Moawad FJ, Bao F, Konijeti GG. The anti-α4β7 integrin therapeutic antibody for inflammatory bowel disease, vedolizumab, ameliorates eosinophilic esophagitis: a novel clinical observation. Am J Gastroenterol. 2018; 113:1261–1263.
Article103. Straumann A, Bussmann C, Conus S, Beglinger C, Simon HU. Anti-TNF-alpha (infliximab) therapy for severe adult eosinophilic esophagitis. J Allergy Clin Immunol. 2008; 122:425–427.104. Tomizawa Y, Melek J, Komaki Y, Kavitt RT, Sakuraba A. Efficacy of pharmacologic therapy for eosinophilic esophagitis: a systematic review and network meta-analysis. J Clin Gastroenterol. 2018; 52:596–606.105. Youngblood BA, Brock EC, Leung J, Falahati R, Bochner BS, Rasmussen HS, et al. Siglec-8 antibody reduces eosinophils and mast cells in a transgenic mouse model of eosinophilic gastroenteritis. JCI Insight. 2019; 4:126219.
Article106. Noti M, Wojno ED, Kim BS, Siracusa MC, Giacomin PR, Nair MG, et al. Thymic stromal lymphopoietin-elicited basophil responses promote eosinophilic esophagitis. Nat Med. 2013; 19:1005–1013.107. Vicari AP, Schoepfer AM, Meresse B, Goffin L, Léger O, Josserand S, et al. Discovery and characterization of a novel humanized anti-IL-15 antibody and its relevance for the treatment of refractory celiac disease and eosinophilic esophagitis. MAbs. 2017; 9:927–944.
Article108. Roufosse FE, Kahn JE, Gleich GJ, Schwartz LB, Singh AD, Rosenwasser LJ, et al. Long-term safety of mepolizumab for the treatment of hypereosinophilic syndromes. J Allergy Clin Immunol. 2013; 131:461–647.e1-5.
Article109. Kuang FL, Legrand F, Makiya M, Ware J, Wetzler L, Brown T, et al. Benralizumab for PDGFRA-negative hypereosinophilic syndrome. N Engl J Med. 2019; 380:1336–1346.110. Kuang FL, Fay MP, Ware J, Wetzler L, Holland-Thomas N, Brown T, et al. Long-term clinical outcomes of high-dose mepolizumab treatment for hypereosinophilic syndrome. J Allergy Clin Immunol Pract. 2018; 6:1518–1527.e5.
Article111. Roufosse F, de Lavareille A, Schandené L, Cogan E, Georgelas A, Wagner L, et al. Mepolizumab as a corticosteroid-sparing agent in lymphocytic variant hypereosinophilic syndrome. J Allergy Clin Immunol. 2010; 126:828–835.e3.
Article112. Wechsler ME, Akuthota P, Jayne D, Khoury P, Klion A, Langford CA, et al. Mepolizumab or placebo for eosinophilic granulomatosis with polyangiitis. N Engl J Med. 2017; 376:1921–1932.
Article113. Kahn JE, Grandpeix-Guyodo C, Marroun I, Catherinot E, Mellot F, Roufosse F, et al. Sustained response to mepolizumab in refractory Churg-Strauss syndrome. J Allergy Clin Immunol. 2010; 125:267–270.
Article114. Soeda S, To M, Kono Y, Yamawaki S, Tsuzuki R, Katsube O, et al. Case series of allergic bronchopulmonary aspergillosis treated successfully and safely with long-term mepolizumab. Allergol Int. 2019; 68:377–379.
Article115. Soeda S, Kono Y, Tsuzuki R, Yamawaki S, Katsube O, To M, et al. Allergic bronchopulmonary aspergillosis successfully treated with benralizumab. J Allergy Clin Immunol Pract. 2019; 7:1633–1635.
Article116. Ange N, Alley S, Fernando SL, Coyle L, Yun J. Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) syndrome successfully treated with mepolizumab. J Allergy Clin Immunol Pract. 2018; 6:1059–1060.
Article117. Matucci A, Liotta F, Vivarelli E, Dies L, Annunziato F, Piccinni MP, et al. Efficacy and safety of mepolizumab (anti-interleukin-5) treatment in Gleich's syndrome. Front Immunol. 2018; 9:1198.
Article118. Taillé C, Pison C, Nocent C, Devouassoux G, Prud’homme A, Gruber A, et al. Patients in the IDEAL cohort: a snapshot of severe asthma in France. Rev Mal Respir. 2019; 36:179–190.119. Jeffery MM, Shah ND, Karaca-Mandic P, Ross JS, Rank MA. Trends in Omalizumab Utilization for Asthma: Evidence of Suboptimal Patient Selection. J Allergy Clin Immunol Pract. 2018; 6:1568–1577.e4.
Article120. Llanos JP, Bell CF, Packnett E, Thiel E, Irwin DE, Hahn B, et al. Real-world characteristics and disease burden of patients with asthma prior to treatment initiation with mepolizumab or omalizumab: a retrospective cohort database study. J Asthma Allergy. 2019; 12:43–58.121. Inselman JW, Jeffery MM, Maddux JT, Shah ND, Rank MA. Trends and disparities in asthma biologic use in the United States. J Allergy Clin Immunol Pract. Forthcoming. 2019.
Article122. Singh H, Peters JI, Kaur Y, Maselli DJ, Diaz JD. Long-term evaluation of response to omalizumab therapy in real life by a novel multimodular approach: the Real-life Effectiveness of Omalizumab Therapy (REALITY) study. Ann Allergy Asthma Immunol. 2019; 123:476–482.e1.123. Sullivan PW, Li Q, Bilir SP, Dang J, Kavati A, Yang M, et al. Cost-effectiveness of omalizumab for the treatment of moderate-to-severe uncontrolled allergic asthma in the United States. Curr Med Res Opin. Forthcoming. 2019.
Article124. Busse W, Chupp G, Nagase H, Albers FC, Doyle S, Shen Q, et al. Anti-IL-5 treatments in patients with severe asthma by blood eosinophil thresholds: Indirect treatment comparison. J Allergy Clin Immunol. 2019; 143:190–200.e20.
Article125. Casale TB, Pacou M, Mesana L, Farge G, Sun SX, Castro M. Reslizumab compared with benralizumab in patients with eosinophilic asthma: a systematic literature review and network meta-analysis. J Allergy Clin Immunol Pract. 2019; 7:122–130.e1.
Article126. Menzella F, Biava M, Bagnasco D, Galeone C, Simonazzi A, Ruggiero P, et al. Efficacy and steroid-sparing effect of benralizumab: has it an advantage over its competitors? Drugs Context. 2019; 8:212580.
Article127. Signorovitch JE, Sikirica V, Erder MH, Xie J, Lu M, Hodgkins PS, et al. Matching-adjusted indirect comparisons: a new tool for timely comparative effectiveness research. Value Health. 2012; 15:940–947.
Article128. Phillippo D, Ades T, Dias S, Palmer S, Abrams KR, Welton N. NICE DSU Technical Support Document 18: Methods for population-adjusted indirect comparisons in submissions to NICE. Sheffield: Decision Support Unit, ScHARR, University of Sheffield;2016.129. Bourdin A, Husereau D, Molinari N, Golam S, Siddiqui MK, Lindner L, et al. Matching-adjusted indirect comparison of benralizumab versus interleukin-5 inhibitors for the treatment of severe asthma: a systematic review. Eur Respir J. 2018; 52:180.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Omalizumab and unmet needs in severe asthma and allergic comorbidities in Japanese children
- Unraveling the Genetic Basis of Asthma and Allergic Diseases
- New Therapeutic Modalities for Asthma: Biologicals from a Practical Point of View
- Allergic rhinitis, sinusitis and asthma: evidence for respiratory system integration
- Epigenetics in Allergic Diseases