Ann Pediatr Endocrinol Metab.
2019 Sep;24(3):149-157. 10.6065/apem.2019.24.3.149.
Evaluation and management of amenorrhea related to congenital sex hormonal disorders
- Affiliations
-
- 1Department of Pediatrics, Pusan National University Children's Hospital, Yangsan, Korea. chongkun@pusan.ac.kr
- KMID: 2460761
- DOI: http://doi.org/10.6065/apem.2019.24.3.149
Abstract
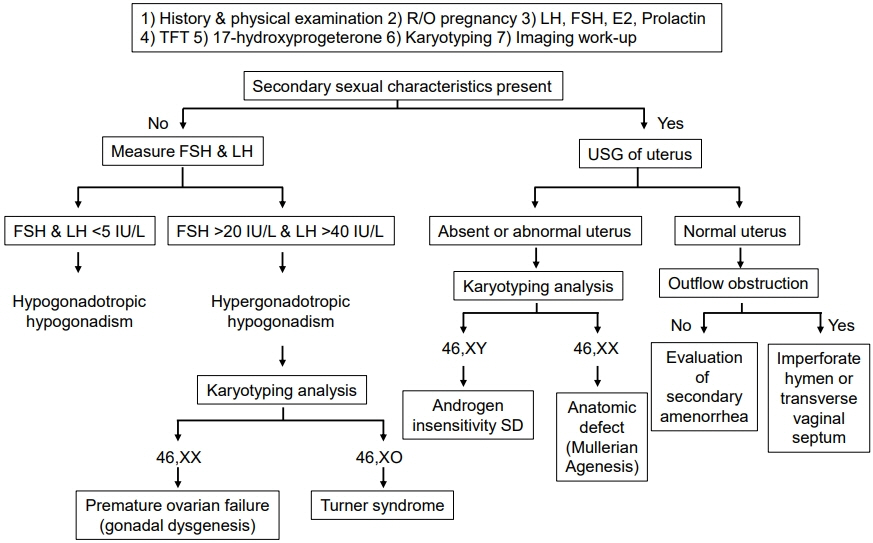
- Primary amenorrhea is a symptom with a substantial list of underlying etiologies which presents in adolescence, although some conditions are diagnosed in childhood. Primary amenorrhea is defined as not having menarche until 15 years of age (or 13 years with secondary sex characteristics). Various etiologies of primary amenorrhea include outflow tract obstructions, gonadal dysgenesis, abnormalities of the central nervous system, various endocrine diseases, chronic illnesses, psychologic problems, and constitutional delay of puberty. The management of primary amenorrhea may vary considerably depending on the patient and the specific diagnosis. In this article, the various causes, evaluation, and management of primary amenorrhea are reviewed with special emphasis on congenital sex hormonal disorders.
Keyword
MeSH Terms
Figure
Reference
-
References
1. ACOG Committee Opinion No. 651: Menstruation in girls and adolescents: using the menstrual cycle as a vital sign. Obstet Gynecol. 2015; 126:e143–6.2. Klein DA, Poth MA. Amenorrhea: an approach to diagnosis and management. Am Fam Physician. 2013; 87:781–8.3. Timmreck LS, Reindollar RH. Contemporary issues in primary amenorrhea. Obstet Gynecol Clin North Am. 2003; 30:287–302.4. Pettersson F, Fries H, Nillius SJ. Epidemiology of secondary amenorrhea. I. Incidence and prevalence rates. Am J Obstet Gynecol. 1973; 117:80–6.5. Klein DA, Paradise SL, Reeder RM. Amenorrhea: a systematic approach to diagnosis and management. Am Fam Physician. 2019; 100:39–48.6. Reindollar RH, Byrd JR, McDonough PG. Delayed sexual development: a study of 252 patients. Am J Obstet Gynecol. 1981; 140:371–80.7. Molitch ME. Diagnosis and treatment of pituitary adenomas: a review. JAMA. 2017; 317:516–24.8. Practice Committee of American Society for Reproductive Medicine. Current evaluation of amenorrhea. Fertil Steril. 2008; 90(5 Suppl):S219–25.9. Well CK, Barbieri RL. Evaluation and management of primary amenorrhea [Internet]. UpToDate;2019. [updated 2019 Mar 21; cited 2019 Sep 8]. Available from: https://www.uptodate.com/contents/evaluation-and-management-of-primary-amenorrhea.10. Gordon CM, Ackerman KE, Berga SL, Kaplan JR, Mastorakos G, Misra M, et al. Functional hypothalamic amenorrhea: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2017; 102:1413–39.11. Baxter RM, Arboleda VA, Lee H, Barseghyan H, Adam MP, Fechner PY, et al. Exome sequencing for the diagnosis of 46,XY disorders of sex development. J Clin Endocrinol Metab. 2015; 100:E333–44.12. Dong Y, Yi Y, Yao H, Yang Z, Hu H, Liu J, et al. Targeted next-generation sequencing identification of mutations in patients with disorders of sex development. BMC Med Genet. 2016; 17:23.13. Marc A, Fritz MD, Leon S. Clinical gynecologic endocrinology and infertility. 8th ed. Philadelphia (PA): Lippincott Williams & Wilkins;2013.14. Rock JA, Zacur HA, Dlugi AM, Jones HW Jr, TeLinde RW. Pregnancy success following surgical correction of imperforate hymen and complete transverse vaginal septum. Obstet Gynecol. 1982; 59:448–51.15. Marsh CA, Grimstad FW. Primary amenorrhea: diagnosis and management. Obstet Gynecol Surv. 2014; 69:603–12.16. Kwon SK, Chae HD, Lee KH, Kim SH, Kim CH, Kang BM. Causes of amenorrhea in Korea: experience of a single large center. Clin Exp Reprod Med. 2014; 41:29–32.17. Hoffman BL, Schorge JO, Bradshaw KD, Halvorson LM, Schaffer JI, Corton MM. Williams gynecology. New York: McGraw-Hill Medical;2012.18. Oktay K, Bedoschi G, Berkowitz K, Bronson R, Kashani B, McGovern P, et al. Fertility preservation in women with Turner syndrome: a comprehensive review and practical guidelines. J Pediatr Adolesc Gynecol. 2016; 29:409–16.19. Bryman I, Sylvén L, Berntorp K, Innala E, Bergström I, Hanson C, et al. Pregnancy rate and outcome in Swedish women with Turner syndrome. Fertil Steril. 2011; 95:2507–10.20. Lucaccioni L, Wong SC, Smyth A, Lyall H, Dominiczak A, Ahmed SF, et al. Turner syndrome--issues to consider for transition to adulthood. Br Med Bull. 2015; 113:45–58.21. Oktay K, Bedoschi G. Fertility preservation in girls with Turner syndrome: limitations, current success and future prospects. Fertil Steril. 2019; 111:1124–6.22. Emans SJ, Goldstein DP, Laufer MR. Pediatric and adolescent gynecology. 5th ed. Philadelphia (PA): Lippincott Williams & Wilkins;2005.23. Michala L, Goswami D, Creighton SM, Conway GS. Swyer syndrome: presentation and outcomes. BJOG. 2008; 115:737–41.24. Larijani B, Bastanhagh MH, Pajouhi M, Kargar Shadab F, Vasigh A, Aghakhani S. Presentation and outcome of 93 cases of craniopharyngioma. Eur J Cancer Care (Engl). 2004; 13:11–5.25. Balasubramanian R, Crowley WF Jr. Isolated gonadotropinreleasing hormone (GnRH) deficiency. In : Margaret PA, Holly HA, editors. GeneReviews. Seattle: University of Washington;1993.26. Chaudhry S, Tadokoro-Cuccaro R, Hannema SE, Acerini CL, Hughes IA. Frequency of gonadal tumours in complete androgen insensitivity syndrome (CAIS): a retrospective case-series analysis. J Pediatr Urol. 2017; 13:498. e1-498.e6.27. Deligeoroglou E, Athanasopoulos N, Tsimaris P, Dimopoulos KD, Vrachnis N, Creatsas G. Evaluation and management of adolescent amenorrhea. Ann N Y Acad Sci. 2010; 1205:23–32.28. Houk CP, Hughes IA, Ahmed SF, Lee PA; Writing Committee for the International Intersex Consensus Conference Participants. Summary of consensus statement on intersex disorders and their management. International Intersex Consensus Conference. Pediatrics. 2006; 118:753–7.29. Cheon CK. Practical approach to steroid 5alpha-reductase type 2 deficiency. Eur J Pediatr. 2011; 170:1–8.30. Well CK, Barbieri RL. Causes of primary amenorrhea [Internet]. UpToDate;2019. [updated 2017 Dec 6; cited 2019 Sep 8]. Available from: https://www.uptodate.com/contents/causes-of-primary-amenorrhea.31. Mieszczak J, Houk CP, Lee PA. Assignment of the sex of rearing in the neonate with a disorder of sex development. Curr Opin Pediatr. 2009; 21:541–7.32. Kojima Y, Mizuno K, Nakane A, Kato T, Kohri K, Hayashi Y. Long-term physical, hormonal, and sexual outcome of males with disorders of sex development. J Pediatr Surg. 2009; 44:1491–6.33. Sultan C, Paris F, Terouanne B, Balaguer P, Georget V, Poujol N, et al. Disorders linked to insufficient androgen action in male children. Hum Reprod Update. 2001; 7:314–22.34. Mendonca BB, Domenice S, Arnhold IJ, Costa EM. 46,XY disorders of sex development (DSD). Clin Endocrinol (Oxf). 2009; 70:173–87.35. Doherty E, Pakarinen P, Tiitinen A, Kiilavuori A, Huhtaniemi I, Forrest S, et al. A Novel mutation in the FSH receptor inhibiting signal transduction and causing primary ovarian failure. J Clin Endocrinol Metab. 2002; 87:1151–5.36. Channing CP, Schaerf FW, Anderson LD, Tsafriri A. Ovarian follicular and luteal physiology. Int Rev Physiol. 1980; 22:117–201.37. Jiang X, Dias JA, He X. Structural biology of glycoprotein hormones and their receptors: insights to signaling. Mol Cell Endocrinol. 2014; 382:424–51.38. Bramble MS, Goldstein EH, Lipson A, Ngun T, Eskin A, Gosschalk JE, et al. A novel follicle-stimulating hormone receptor mutation causing primary ovarian failure: a fertility application of whole exome sequencing. Hum Reprod. 2016; 31:905–14.39. Persani L, Rossetti R, Cacciatore C. Genes involved in human premature ovarian failure. J Mol Endocrinol. 2010; 45:257–79.40. Desai SS, Roy BS, Mahale SD. Mutations and polymorphisms in FSH receptor: functional implications in human reproduction. Reproduction. 2013; 146:R235–48.41. Laven JSE. Follicle stimulating hormone receptor (FSHR) polymorphisms and polycystic ovary syndrome (PCOS). Front Endocrinol (Lausanne). 2019; 10:23.42. McAllister JM, Legro RS, Modi BP, Strauss JF 3rd. Functional genomics of PCOS: from GWAS to molecular mechanisms. Trends Endocrinol Metab. 2015; 26:118–24.43. Practice Committee of the American Society for Reproductive Medicine. Obesity and reproduction: a committee opinion. Fertil Steril. 2015; 104:1116–26.44. ACOG Practice Bulletin No. 194: Polycystic Ovary Syndrome. Obstet Gynecol. 2018; 131:e157–71.45. Melmed S, Casanueva FF, Hoffman AR, Kleinberg DL, Montori VM, Schlechte JA, et al. Diagnosis and treatment of hyperprolactinemia: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2011; 96:273–88.46. Carmina E, Dewailly D, Escobar-Morreale HF, Kelestimur F, Moran C, Oberfield S, et al. Non-classic congenital adrenal hyperplasia due to 21-hydroxylase deficiency revisited: an update with a special focus on adolescent and adult women. Hum Reprod Update. 2017; 23:580–99.47. Moran C, Azziz R, Carmina E, Dewailly D, Fruzzetti F, Ibañez L, et al. 21-Hydroxylase-deficient nonclassic adrenal hyperplasia is a progressive disorder: a multicenter study. Am J Obstet Gynecol. 2000; 183:1468–74.48. Moran C, Azziz R, Weintrob N, Witchel SF, Rohmer V, Dewailly D, et al. Reproductive outcome of women with 21-hydroxylase-deficient nonclassic adrenal hyperplasia. J Clin Endocrinol Metab. 2006; 91:3451–6.49. Bidet M, Bellanné-Chantelot C, Galand-Portier MB, Golmard JL, Tardy V, Morel Y, et al. Fertility in women with nonclassical congenital adrenal hyperplasia due to 21-hydroxylase deficiency. J Clin Endocrinol Metab. 2010; 95:1182–90.50. Reichman DE, White PC, New MI, Rosenwaks Z. Fertility in patients with congenital adrenal hyperplasia. Fertil Steril. 2014; 101:301–9.51. Miller WL, Auchus RJ. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr Rev. 2011; 32:81–151.52. Griffing GT, Wilson TE, Holbrook MM, Dale SL, Jackson TK, Ullrich I, et al. Plasma and urinary 19-nordeoxycorticosterone in 17 alpha-hydroxylase deficiency syndrome. J Clin Endocrinol Metab. 1984; 59:1011–5.53. Martin RM, Lin CJ, Costa EM, de Oliveira ML, Carrilho A, Villar H, et al. P450c17 deficiency in Brazilian patients: biochemical diagnosis through progesterone levels confirmed by CYP17 genotyping. J Clin Endocrinol Metab. 2003; 88:5739–46.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Causes and Clinical Characteristics of Patients with Abnormal Sex Differentiation and Development
- A case of primary amenorrhea with hypertension due to 17alpha-hydroxylase deficiency
- Diagnostic approach of amenorrhea
- A case of male Pseudohermaphroditism due to 17alpha-Hydroxylase Deficiency
- A Cytogenetic Study of Amenorrhea