Korean J Radiol.
2016 Oct;17(5):684-694. 10.3348/kjr.2016.17.5.684.
Diagnosing Lung Nodules on Oncologic MR/PET Imaging: Comparison of Fast T1-Weighted Sequences and Influence of Image Acquisition in Inspiration and Expiration Breath-Hold
- Affiliations
-
- 1Department of Diagnostic and Interventional Radiology, University Hospital of Tuebingen, Tuebingen 72076, Germany. christina.schraml@med.uni-tuebingen.de
- 2Department of Diagnostic and Interventional Neuroradiology, University Hospital of Tuebingen, Tuebingen 72076, Germany.
- 3Department of Nuclear Medicine, University Hospital of Tuebingen, Tuebingen 72076, Germany.
- KMID: 2458060
- DOI: http://doi.org/10.3348/kjr.2016.17.5.684
Abstract
OBJECTIVE
First, to investigate the diagnostic performance of fast T1-weighted sequences for lung nodule evaluation in oncologic magnetic resonance (MR)/positron emission tomography (PET). Second, to evaluate the influence of image acquisition in inspiration and expiration breath-hold on diagnostic performance.
MATERIALS AND METHODS
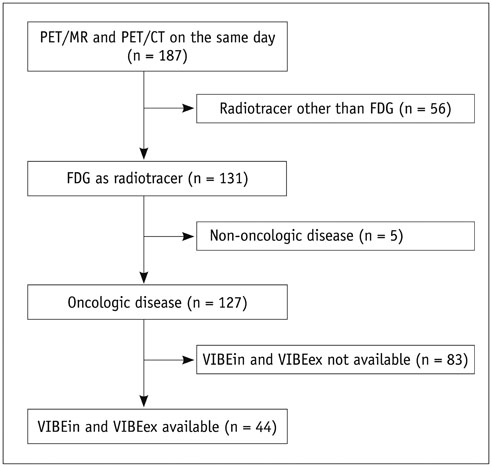
The study was approved by the local Institutional Review Board. PET/CT and MR/PET of 44 cancer patients were evaluated by 2 readers. PET/CT included lung computed tomography (CT) scans in inspiration and expiration (CTin, CTex). MR/PET included Dixon sequence for attenuation correction and fast T1-weighted volumetric interpolated breath-hold examination (VIBE) sequences (volume interpolated breath-hold examination acquired in inspiration [VIBEin], volume interpolated breath-hold examination acquired in expiration [VIBEex]). Diagnostic performance was analyzed for lesion-, lobe-, and size-dependence. Diagnostic confidence was evaluated (4-point Likert-scale; 1 = high). Jackknife alternative free-response receiver-operating characteristic (JAFROC) analysis was performed.
RESULTS
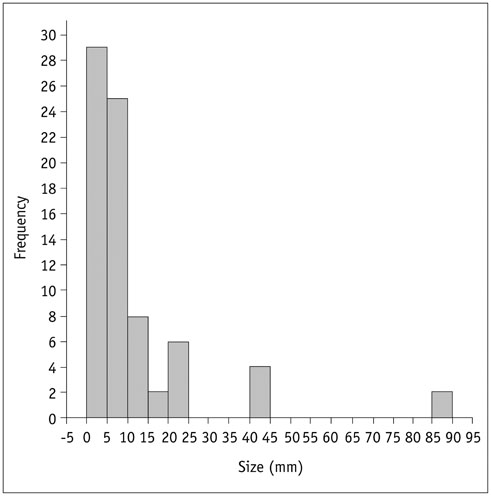
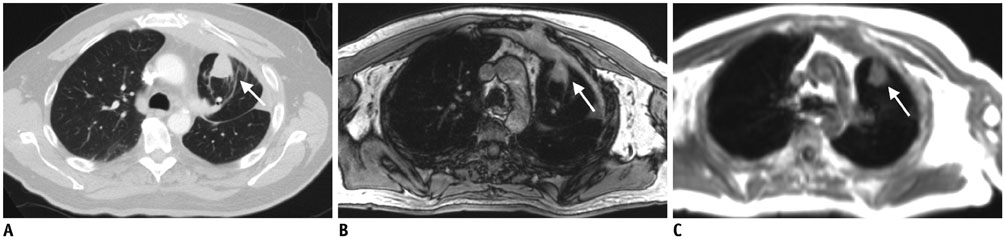
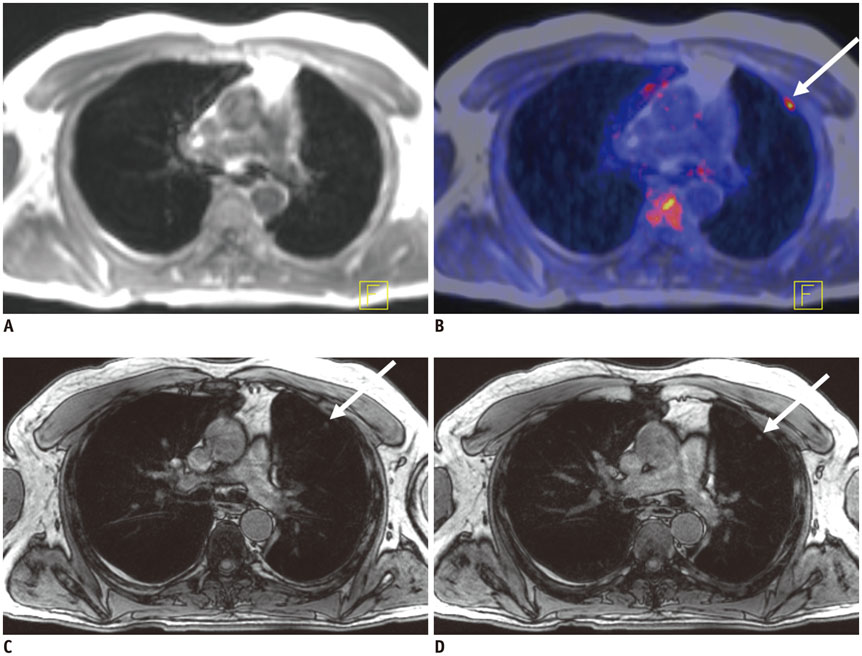
Seventy-six pulmonary lesions were evaluated. Lesion-based detection rates were: CTex, 77.6%; VIBEin, 53.3%; VIBEex, 51.3%; and Dixon, 22.4%. Lobe-based detection rates were: CTex, 89.6%; VIBEin, 58.3%; VIBEex, 60.4%; and Dixon, 31.3%. In contrast to CT, inspiration versus expiration did not alter diagnostic performance in VIBE sequences. Diagnostic confidence was best for VIBEin and CTex and decreased in VIBEex and Dixon (1.2 ± 0.6; 1.2 ± 0.7; 1.5 ± 0.9; 1.7 ± 1.1, respectively). The JAFROC figure-of-merit of Dixon was significantly lower. All patients with malignant lesions were identified by CTex, VIBEin, and VIBEex, while 3 patients were false-negative in Dixon.
CONCLUSION
Fast T1-weighted VIBE sequences allow for identification of patients with malignant pulmonary lesions. The Dixon sequence is not recommended for lung nodule evaluation in oncologic MR/PET patients. In contrast to CT, inspiration versus expiratory breath-hold in VIBE sequences was less crucial for lung nodule evaluation but was important for diagnostic confidence.
Keyword
MeSH Terms
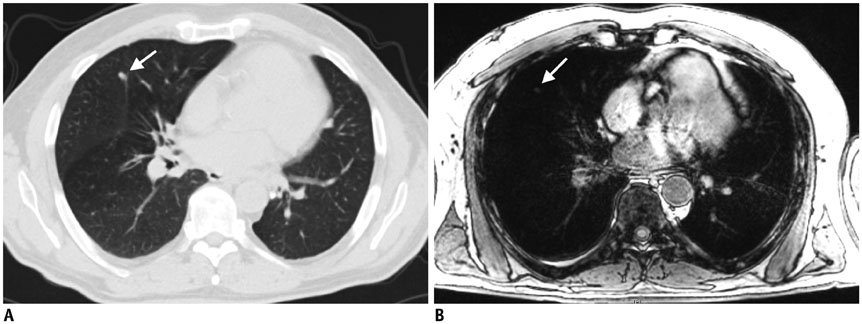
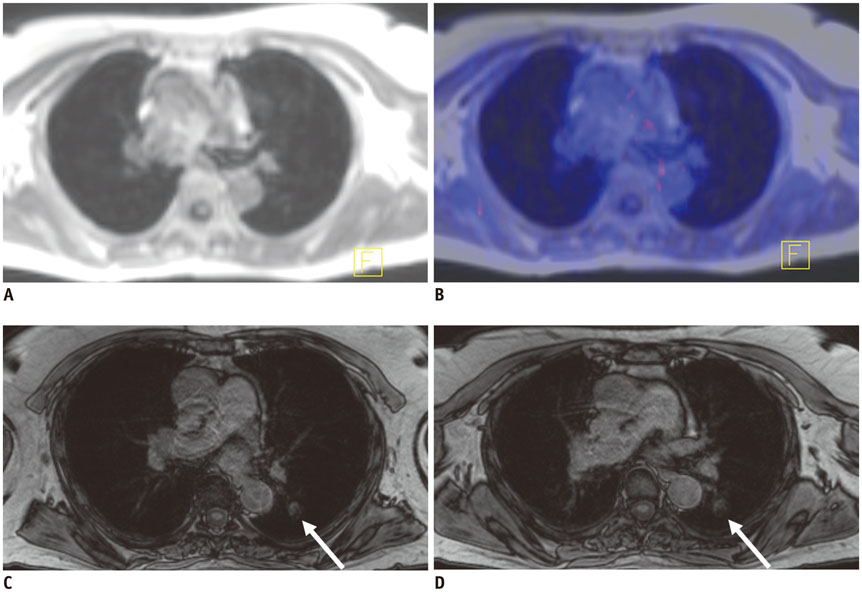
Figure
Cited by 1 articles
-
Morphologic Evaluation of Primary Non-Small Cell Lung Cancer by 3 Tesla MRI with Free-Breathing Ultrashort Echo Time and Radial T1-Weighted Gradient Echo Sequences: A Comparison with CT Analysis
Hyunji Lee, EunHee Choi, Myoung Kyu Lee, Yu Zhang, Woocheol Kwon
J Korean Soc Radiol. 2019;80(3):466-476. doi: 10.3348/jksr.2019.80.3.466.
Reference
-
1. Yoo HJ, Lee JS, Lee JM. Integrated whole body MR/PET: where are we? Korean J Radiol. 2015; 16:32–49.2. Catalano OA, Rosen BR, Sahani DV, Hahn PF, Guimaraes AR, Vangel MG, et al. Clinical impact of PET/MR imaging in patients with cancer undergoing same-day PET/CT: initial experience in 134 patients--a hypothesis-generating exploratory study. Radiology. 2013; 269:857–869.3. Boss A, Weiger M, Wiesinger F. Future image acquisition trends for PET/MRI. Semin Nucl Med. 2015; 45:201–211.4. Gatidis S, Schmidt H, Gückel B, Bezrukov I, Seitz G, Ebinger M, et al. Comprehensive oncologic imaging in infants and preschool children with substantially reduced radiation exposure using combined simultaneous 18F-fluorodeoxyglucose positron emission tomography/magnetic resonance imaging: a direct comparison to 18F-fluorodeoxyglucose positron emission tomography/computed tomography. Invest Radiol. 2016; 51:7–14.5. Grueneisen J, Schaarschmidt BM, Heubner M, Suntharalingam S, Milk I, Kinner S, et al. Implementation of FAST-PET/MRI for whole-body staging of female patients with recurrent pelvic malignancies: a comparison to PET/CT. Eur J Radiol. 2015; 84:2097–2102.6. Manber R, Thielemans K, Hutton BF, Barnes A, Ourselin S, Arridge S, et al. Practical PET respiratory motion correction in clinical PET/MR. J Nucl Med. 2015; 56:890–896.7. Würslin C, Schmidt H, Martirosian P, Brendle C, Boss A, Schwenzer NF, et al. Respiratory motion correction in oncologic PET using T1-weighted MR imaging on a simultaneous whole-body PET/MR system. J Nucl Med. 2013; 54:464–471.8. Biederer J, Beer M, Hirsch W, Wild J, Fabel M, Puderbach M, et al. MRI of the lung (2/3). Why … when … how? Insights Imaging. 2012; 3:355–371.9. Wild JM, Marshall H, Bock M, Schad LR, Jakob PM, Puderbach M, et al. MRI of the lung (1/3): methods. Insights Imaging. 2012; 3:345–353.10. Chandarana H, Block TK, Rosenkrantz AB, Lim RP, Kim D, Mossa DJ, et al. Free-breathing radial 3D fat-suppressed T1-weighted gradient echo sequence: a viable alternative for contrast-enhanced liver imaging in patients unable to suspend respiration. Invest Radiol. 2011; 46:648–653.11. Raad RA, Friedman KP, Heacock L, Ponzo F, Melsaether A, Chandarana H. Outcome of small lung nodules missed on hybrid PET/MRI in patients with primary malignancy. J Magn Reson Imaging. 2016; 43:504–511.12. Bader TR, Semelka RC, Pedro MS, Armao DM, Brown MA, Molina PL. Magnetic resonance imaging of pulmonary parenchymal disease using a modified breath-hold 3D gradient-echo technique: initial observations. J Magn Reson Imaging. 2002; 15:31–38.13. Biederer J, Schoene A, Freitag S, Reuter M, Heller M. Simulated pulmonary nodules implanted in a dedicated porcine chest phantom: sensitivity of MR imaging for detection. Radiology. 2003; 227:475–483.14. Fink C, Puderbach M, Biederer J, Fabel M, Dietrich O, Kauczor HU, et al. Lung MRI at 1.5 and 3 Tesla: observer preference study and lesion contrast using five different pulse sequences. Invest Radiol. 2007; 42:377–383.15. Eiber M, Martinez-Möller A, Souvatzoglou M, Holzapfel K, Pickhard A, Löffelbein D, et al. Value of a Dixon-based MR/PET attenuation correction sequence for the localization and evaluation of PET-positive lesions. Eur J Nucl Med Mol Imaging. 2011; 38:1691–1701.16. Gilman MD, Fischman AJ, Krishnasetty V, Halpern EF, Aquino SL. Optimal CT breathing protocol for combined thoracic PET/CT. AJR Am J Roentgenol. 2006; 187:1357–1360.17. Juergens KU, Weckesser M, Stegger L, Franzius C, Beetz M, Schober O, et al. Tumor staging using whole-body high-resolution 16-channel PET-CT: does additional low-dose chest CT in inspiration improve the detection of solitary pulmonary nodules? Eur Radiol. 2006; 16:1131–1137.18. de Juan R, Seifert B, Berthold T, von Schulthess GK, Goerres GW. Clinical evaluation of a breathing protocol for PET/CT. Eur Radiol. 2004; 14:1118–1123.19. Goerres GW, Kamel E, Heidelberg TN, Schwitter MR, Burger C, von Schulthess GK. PET-CT image co-registration in the thorax: influence of respiration. Eur J Nucl Med Mol Imaging. 2002; 29:351–360.20. MacMahon H, Austin JH, Gamsu G, Herold CJ, Jett JR, Naidich DP, et al. Guidelines for management of small pulmonary nodules detected on CT scans: a statement from the Fleischner Society. Radiology. 2005; 237:395–400.21. Chakraborty DP. Analysis of location specific observer performance data: validated extensions of the jackknife free-response (JAFROC) method. Acad Radiol. 2006; 13:1187–1193.22. Chakraborty DP, Berbaum KS. Observer studies involving detection and localization: modeling, analysis, and validation. Med Phys. 2004; 31:2313–2330.23. Vogel MN, Brechtel K, Klein MD, Aschoff P, Horger M, Eschmann S, et al. [Evaluation of different breathing and contrast-protocols concerning quality and alignment in 18F-FDG PET/CT]. Rofo. 2007; 179:72–79.24. Allen-Auerbach M, Yeom K, Park J, Phelps M, Czernin J. Standard PET/CT of the chest during shallow breathing is inadequate for comprehensive staging of lung cancer. J Nucl Med. 2006; 47:298–301.25. Aquino SL, Kuester LB, Muse VV, Halpern EF, Fischman AJ. Accuracy of transmission CT and FDG-PET in the detection of small pulmonary nodules with integrated PET/CT. Eur J Nucl Med Mol Imaging. 2006; 33:692–696.26. Chandarana H, Heacock L, Rakheja R, DeMello LR, Bonavita J, Block TK, et al. Pulmonary nodules in patients with primary malignancy: comparison of hybrid PET/MR and PET/CT imaging. Radiology. 2013; 268:874–881.27. Rauscher I, Eiber M, Fürst S, Souvatzoglou M, Nekolla SG, Ziegler SI, et al. PET/MR imaging in the detection and characterization of pulmonary lesions: technical and diagnostic evaluation in comparison to PET/CT. J Nucl Med. 2014; 55:724–729.28. Heye T, Ley S, Heussel CP, Dienemann H, Kauczor HU, Hosch W, et al. Detection and size of pulmonary lesions: how accurate is MRI? A prospective comparison of CT and MRI. Acta Radiol. 2012; 53:153–160.29. Goo JM, Kim KG, Gierada DS, Castro M, Bae KT. Volumetric measurements of lung nodules with multi-detector row CT: effect of changes in lung volume. Korean J Radiol. 2006; 7:243–248.30. Drzezga A, Souvatzoglou M, Eiber M, Beer AJ, Fürst S, Martinez-Möller A, et al. First clinical experience with integrated whole-body PET/MR: comparison to PET/CT in patients with oncologic diagnoses. J Nucl Med. 2012; 53:845–855.31. Stolzmann P, Veit-Haibach P, Chuck N, Rossi C, Frauenfelder T, Alkadhi H, et al. Detection rate, location, and size of pulmonary nodules in trimodality PET/CT-MR: comparison of low-dose CT and Dixon-based MR imaging. Invest Radiol. 2013; 48:241–246.32. Schaarschmidt B, Buchbender C, Gomez B, Rubbert C, Hild F, Köhler J, et al. Thoracic staging of non-small-cell lung cancer using integrated (18)F-FDG PET/MR imaging: diagnostic value of different MR sequences. Eur J Nucl Med Mol Imaging. 2015; 42:1257–1267.33. Sommer G, Tremper J, Koenigkam-Santos M, Delorme S, Becker N, Biederer J, et al. Lung nodule detection in a high-risk population: comparison of magnetic resonance imaging and low-dose computed tomography. Eur J Radiol. 2014; 83:600–605.34. Fraioli F, Screaton NJ, Janes SM, Win T, Menezes L, Kayani I, et al. Non-small-cell lung cancer resectability: diagnostic value of PET/MR. Eur J Nucl Med Mol Imaging. 2015; 42:49–55.35. Munden RF, Erasmus JJ, Wahba H, Fineberg NS. Follow-up of small (4 mm or less) incidentally detected nodules by computed tomography in oncology patients: a retrospective review. J Thorac Oncol. 2010; 5:1958–1962.36. Lee KH, Park CM, Lee SM, Lee JM, Cho JY, Paeng JC, et al. Pulmonary nodule detection in patients with a primary malignancy using hybrid PET/MRI: is there value in adding contrast-enhanced MR imaging? PLoS One. 2015; 10:e0129660.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Optimal MR Pulse Sequences for Hepatic Hemangiomas: Comparison of T2-Weighted Turbo-Spin-Echo, T2-Weighted Breath-hold Turbo-Spin-Echo, and T1-Weighted FLASH Dynamic Imaging
- Comparison of Non-Breath-Hold T2-weighted Turbo Spin-Echo and Three Breath-Hold T2-weighted MR Images for Detection of Focal Hepatic Lesion
- SPIO-enhanced MR Imaging for HCC Detection in Cirrhotic Patient: Comparison of Various Techniques for Optimal Sequence Selection
- Comparison of In-Phase and Opposed-Phase FMPSPGR Images in Breath-hold T1-weighted MR Imaging of Liver
- Chemical Saturation Breath-hold Fast MR Imaging for Characterization of Regional Fatty Changes in Liver