Korean J Radiol.
2019 Sep;20(9):1381-1389. 10.3348/kjr.2018.0814.
Radiomics MRI Phenotyping with Machine Learning to Predict the Grade of Lower-Grade Gliomas: A Study Focused on Nonenhancing Tumors
- Affiliations
-
- 1Department of Radiology, Ewha Womans University College of Medicine, Seoul, Korea.
- 2Department of Radiology and Research Institute of Radiological Science, Yonsei University College of Medicine, Seoul, Korea. yoonseong.choi07@gmail.com
- 3Department of Neurosurgery, Yonsei University College of Medicine, Seoul, Korea.
- 4Department of Pathology, Yonsei University College of Medicine, Seoul, Korea.
- KMID: 2455766
- DOI: http://doi.org/10.3348/kjr.2018.0814
Abstract
OBJECTIVE
To assess whether radiomics features derived from multiparametric MRI can predict the tumor grade of lower-grade gliomas (LGGs; World Health Organization grade II and grade III) and the nonenhancing LGG subgroup.
MATERIALS AND METHODS
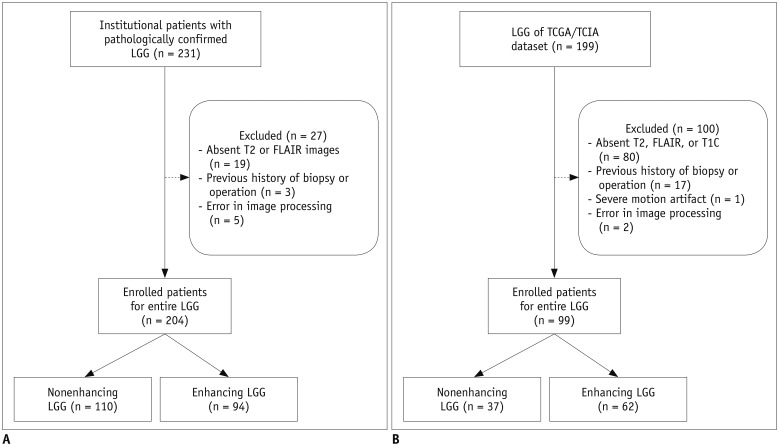
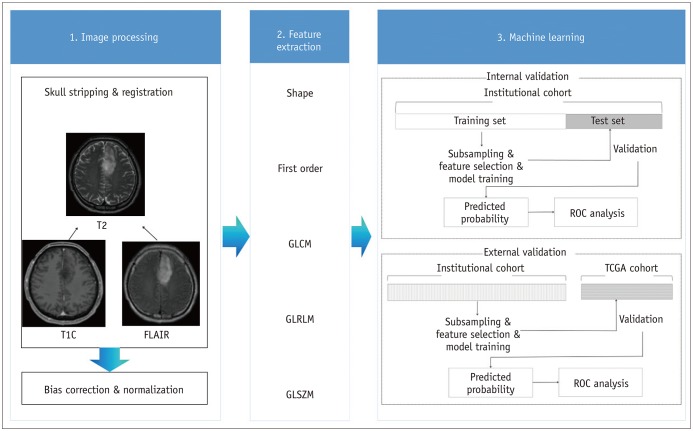
Two-hundred four patients with LGGs from our institutional cohort were allocated to training (n = 136) and test (n = 68) sets. Postcontrast T1-weighted images, T2-weighted images, and fluid-attenuated inversion recovery images were analyzed to extract 250 radiomics features. Various machine learning classifiers were trained using the radiomics features to predict the glioma grade. The trained classifiers were internally validated on the institutional test set and externally validated on a separate cohort (n = 99) from The Cancer Genome Atlas (TCGA). Classifier performance was assessed by determining the area under the curve (AUC) from receiver operating characteristic curve analysis. An identical process was performed in the nonenhancing LGG subgroup (institutional training set, n = 73; institutional test set, n = 37; and TCGA cohort, n = 37) to predict the glioma grade.
RESULTS
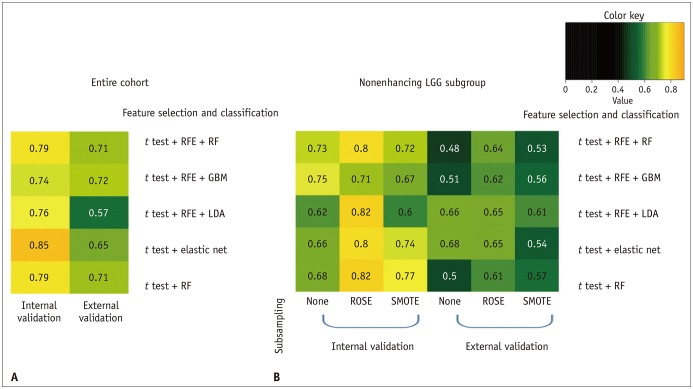
The performance of the best classifier was good in the internal validation set (AUC, 0.85) and fair in the external validation set (AUC, 0.72) to predict the LGG grade. For the nonenhancing LGG subgroup, the performance of the best classifier was good in the internal validation set (AUC, 0.82), but poor in the external validation set (AUC, 0.68).
CONCLUSION
Radiomics feature-based classifiers may be useful to predict LGG grades. However, radiomics classifiers may have a limited value when applied to the nonenhancing LGG subgroup in a TCGA cohort.
MeSH Terms
Figure
Cited by 1 articles
-
Multiomics Approach to Acromegaly: Unveiling Translational Insights for Precision Medicine
Kyungwon Kim, Cheol Ryong Ku, Eun Jig Lee
Endocrinol Metab. 2023;38(5):463-471. doi: 10.3803/EnM.2023.1820.
Reference
-
1. Cancer Genome Atlas Research Network. Brat DJ, Verhaak RG, Aldape KD, Yung WK, Salama SR, Cooper LA, et al. Comprehensive, integrative genomic analysis of diffuse lower-grade gliomas. N Engl J Med. 2015; 372:2481–2498. PMID: 26061751.2. Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, et al. The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol. 2016; 131:803–820. PMID: 27157931.
Article3. Rollin N, Guyotat J, Streichenberger N, Honnorat J, Tran Minh VA, Cotton F. Clinical relevance of diffusion and perfusion magnetic resonance imaging in assessing intra-axial brain tumors. Neuroradiology. 2006; 48:150–159. PMID: 16470375.
Article4. Lee EJ, Lee SK, Agid R, Bae JM, Keller A, Terbrugge K. Preoperative grading of presumptive low-grade astrocytomas on MR imaging: diagnostic value of minimum apparent diffusion coefficient. AJNR Am J Neuroradiol. 2008; 29:1872–1877. PMID: 18719036.
Article5. Olar A, Wani KM, Alfaro-Munoz KD, Heathcock LE, van Thuijl HF, Gilbert MR, et al. IDH mutation status and role of WHO grade and mitotic index in overall survival in grade II–III diffuse gliomas. Acta Neuropathol. 2015; 129:585–596. PMID: 25701198.
Article6. Reuss DE, Mamatjan Y, Schrimpf D, Capper D, Hovestadt V, Kratz A, et al. IDH mutant diffuse and anaplastic astrocytomas have similar age at presentation and little difference in survival: a grading problem for WHO. Acta Neuropathol. 2015; 129:867–873. PMID: 25962792.
Article7. Killela PJ, Pirozzi CJ, Healy P, Reitman ZJ, Lipp E, Rasheed BA, et al. Mutations in IDH1, IDH2, and in the TERT promoter define clinically distinct subgroups of adult malignant gliomas. Oncotarget. 2014; 5:1515–1525. PMID: 24722048.
Article8. Wong JC, Provenzale JM, Petrella JR. Perfusion MR imaging of brain neoplasms. AJR Am J Roentgenol. 2000; 174:1147–1157. PMID: 10749268.
Article9. Cho SK, Na DG, Ryoo JW, Roh HG, Moon CH, Byun HS, et al. Perfusion MR imaging: clinical utility for the differential diagnosis of various brain tumors. Korean J Radiol. 2002; 3:171–179. PMID: 12271162.
Article10. Feiden W, Steude U, Bise K, Gündisch O. Accuracy of stereotactic brain tumor biopsy: comparison of the histologic findings in biopsy cylinders and resected tumor tissue. Neurosurg Rev. 1991; 14:51–56. PMID: 2030827.11. Yu X, Liu Z, Tian Z, Li S, Huang H, Xiu B, et al. Stereotactic biopsy for intracranial space-occupying lesions: clinical analysis of 550 cases. Stereotact Funct Neurosurg. 2000; 75:103–108. PMID: 11740177.
Article12. Field M, Witham TF, Flickinger JC, Kondziolka D, Lunsford LD. Comprehensive assessment of hemorrhage risks and outcomes after stereotactic brain biopsy. J Neurosurg. 2001; 94:545–551. PMID: 11302651.
Article13. Takano K, Kinoshita M, Arita H, Okita Y, Chiba Y, Kagawa N, et al. Diagnostic and prognostic value of 11C-methionine PET for nonenhancing gliomas. AJNR Am J Neuroradiol. 2016; 37:44–50. PMID: 26381556.14. Tynninen O, Aronen HJ, Ruhala M, Paetau A, Von Boguslawski K, Salonen O, et al. MRI enhancement and microvascular density in gliomas. Correlation with tumor cell proliferation. Invest Radiol. 1999; 34:427–443. PMID: 10353036.15. Lüdemann L, Grieger W, Wurm R, Budzisch M, Hamm B, Zimmer C. Comparison of dynamic contrast-enhanced MRI with WHO tumor grading for gliomas. Eur Radiol. 2001; 11:1231–1241. PMID: 11471617.
Article16. Butler AR, Horii SC, Kricheff II, Shannon MB, Budzilovich GN. Computed tomography in astrocytomas. A statistical analysis of the parameters of malignancy and the positive contrast-enhanced CT scan. Radiology. 1978; 129:433–443. PMID: 212778.17. White ML, Zhang Y, Kirby P, Ryken TC. Can tumor contrast enhancement be used as a criterion for differentiating tumor grades of oligodendrogliomas? AJNR Am J Neuroradiol. 2005; 26:784–790. PMID: 15814921.18. Maia AC Jr, Malheiros SM, da Rocha AJ, da Silva CJ, Gabbai AA, Ferraz FA, et al. MR cerebral blood volume maps correlated with vascular endothelial growth factor expression and tumor grade in nonenhancing gliomas. AJNR Am J Neuroradiol. 2005; 26:777–783. PMID: 15814920.19. Scott JN, Brasher PM, Sevick RJ, Rewcastle NB, Forsyth PA. How often are nonenhancing supratentorial gliomas malignant? A population study. Neurology. 2002; 59:947–994. PMID: 12297589.
Article20. Barker FG 2nd, Chang SM, Huhn SL, Davis RL, Gutin PH, McDermott MW, et al. Age and the risk of anaplasia in magnetic resonance-nonenhancing supratentorial cerebral tumors. Cancer. 1997; 80:936–941. PMID: 9307194.
Article21. Liu X, Tian W, Kolar B, Yeaney GA, Qiu X, Johnson MD, et al. MR diffusion tensor and perfusion-weighted imaging in preoperative grading of supratentorial nonenhancing gliomas. Neuro Oncol. 2011; 13:447–455. PMID: 21297125.
Article22. Davnall F, Yip CS, Ljungqvist G, Selmi M, Ng F, Sanghera B, et al. Assessment of tumor heterogeneity: an emerging imaging tool for clinical practice? Insights Imaging. 2012; 3:573–589. PMID: 23093486.
Article23. Zhou H, Vallières M, Bai HX, Su C, Tang H, Oldridge D, et al. MRI features predict survival and molecular markers in diffuse lower-grade gliomas. Neuro Oncol. 2017; 19:862–870. PMID: 28339588.
Article24. Kickingereder P, Burth S, Wick A, Götz M, Eidel O, Schlemmer HP, et al. Radiomic profiling of glioblastoma: identifying an imaging predictor of patient survival with improved performance over established clinical and radiologic risk models. Radiology. 2016; 280:880–889. PMID: 27326665.
Article25. Ceccarelli M, Barthel FP, Malta TM, Sabedot TS, Salama SR, Murray BA, et al. Molecular profiling reveals biologically discrete subsets and pathways of progression in diffuse glioma. Cell. 2016; 164:550–563. PMID: 26824661.26. Bakas S, Akbari H, Sotiras A, Bilello M, Rozycki M, Kirby JS, et al. Advancing The Cancer Genome Atlas glioma MRI collections with expert segmentation labels and radiomic features. Sci Data. 2017; 4:170117. PMID: 28872634.
Article27. Shinohara RT, Sweeney EM, Goldsmith J, Shiee N, Mateen FJ, Calabresi PA, et al. Australian Imaging Biomarkers Lifestyle Flagship Study of Ageing. Alzheimer's Disease Neuroimaging Initiative. Statistical normalization techniques for magnetic resonance imaging. Neuroimage Clin. 2014; 6:9–19. PMID: 25379412.
Article28. van Griethuysen JJM, Fedorov A, Parmar C, Hosny A, Aucoin N, Narayan V, et al. Computational radiomics system to decode the radiographic phenotype. Cancer Res. 2017; 77:e104–e107. PMID: 29092951.
Article29. Chawla NV, Bowyer KW, Hall LO, Kegelmeyer WP. SMOTE: synthetic minority over-sampling technique. J Artif Intell Res. 2002; 16:321–357.
Article30. Kuhn M. Building predictive models in R using the caret package. J Stat Softw. 2008; 28:1–26. PMID: 27774042.31. Hilario A, Ramos A, Perez-Nuñez A, Salvador E, Millan JM, Lagares A, et al. The added value of apparent diffusion coefficient to cerebral blood volume in the preoperative grading of diffuse gliomas. AJNR Am J Neuroradiol. 2012; 33:701–707. PMID: 22207304.
Article32. Stadlbauer A, Gruber S, Nimsky C, Fahlbusch R, Hammen T, Buslei R, et al. Preoperative grading of gliomas by using metabolite quantification with high-spatial-resolution proton MR spectroscopic imaging. Radiology. 2006; 238:958–969. PMID: 16424238.
Article33. Yu J, Shi Z, Lian Y, Li Z, Liu T, Gao Y, et al. Noninvasive IDH1 mutation estimation based on a quantitative radiomics approach for grade II glioma. Eur Radiol. 2017; 27:3509–3522. PMID: 28004160.
Article34. Park YW, Han K, Ahn SS, Choi YS, Chang JH, Kim SH, et al. Whole-tumor histogram and texture analyses of DTI for evaluation of IDH1-mutation and 1p/19q-codeletion status in World Health Organization grade II gliomas. AJNR Am J Neuroradiol. 2018; 39:693–698. PMID: 29519794.35. Mihara F, Numaguchi Y, Rothman M, Kristt D, Fiandaca M, Swallow L. Non-enhancing supratentorial malignant astrocytomas: MR features and possible mechanisms. Radiat Med. 1995; 13:11–17. PMID: 7597198.36. van den Bent MJ. Interobserver variation of the histopathological diagnosis in clinical trials on glioma: a clinician's perspective. Acta Neuropathol. 2010; 120:297–304. PMID: 20644945.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Radiomics and machine learning analysis of liver magnetic resonance imaging for prediction and early detection of tumor response in colorectal liver metastases
- Comparison of Normalization Techniques for Radiomics Features From Magnetic Resonance Imaging in Predicting Histologic Grade of Meningiomas
- Diffusion- and Perfusion-Weighted MRI Radiomics for Survival Prediction in Patients with Lower-Grade Gliomas
- Clinical and Pathophysiological Characteristics of Cerebral Gliomas without Enhancement on Magnetic Resonance Images
- Expression of Trans forming Growth Factor-a and Proliferating Cell Nuclear Antigen in Human Gliomas