Korean J Radiol.
2019 Jun;20(6):939-946. 10.3348/kjr.2018.0803.
Percutaneous Sacroplasty for Painful Sacral Metastases Involving Multiple Sacral Vertebral Bodies: Initial Experience with an Interpedicular Approach
- Affiliations
-
- 1Department of Diagnostic and Interventional Radiology, Shanghai Sixth People's Hospital East Affiliated to Shanghai University of Medicine & Health Sciences, Shanghai, China. 649514608@qq.com
- KMID: 2447072
- DOI: http://doi.org/10.3348/kjr.2018.0803
Abstract
OBJECTIVE
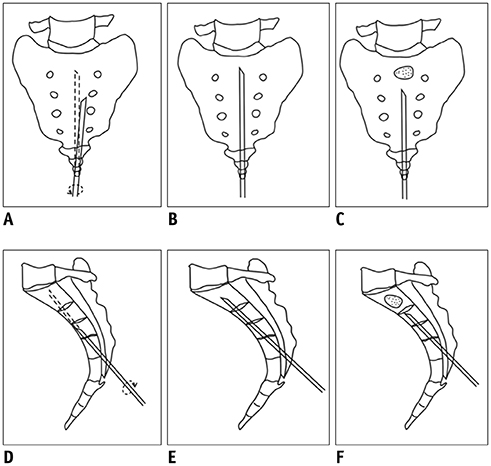
To report our initial experience of percutaneous sacroplasty (PSP) with an interpedicular approach for treating painful sacral metastases involving multiple sacral vertebral bodies.
MATERIALS AND METHODS
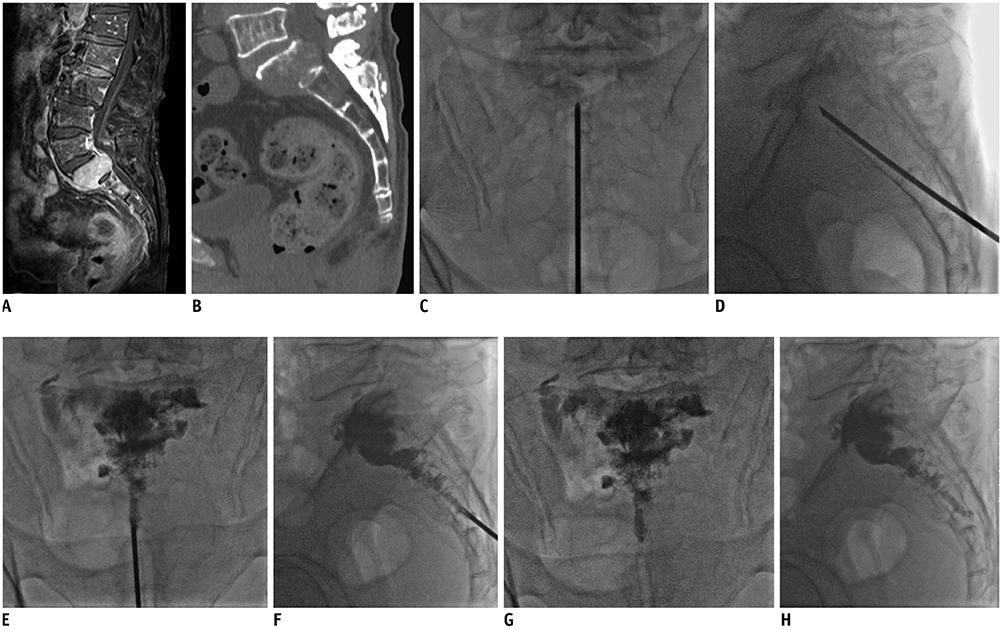
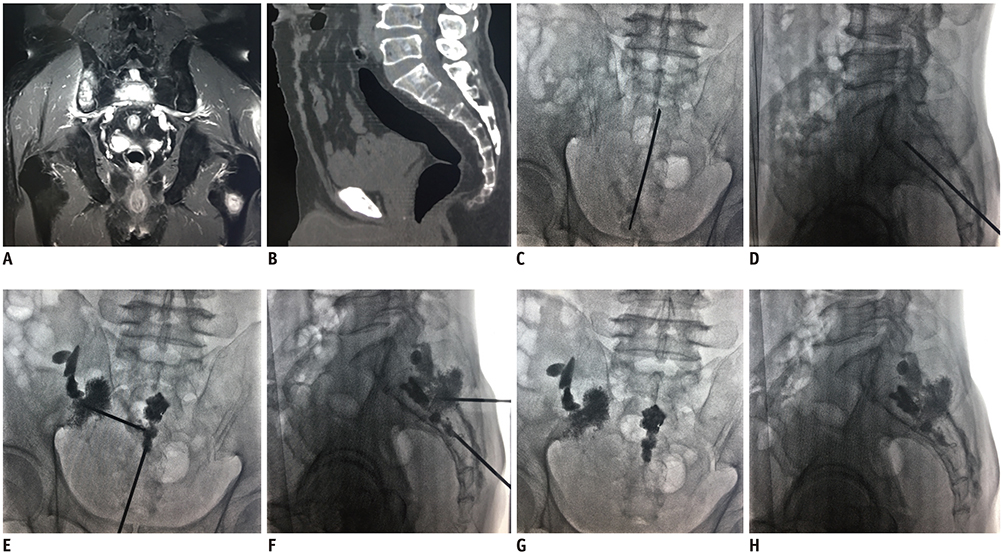
This study prospectively enrolled 10 consecutive patients (six men and four women; mean age, 56.3 ± 13.8 years) who underwent PSP for painful sacral metastases involving multiple sacral vertebral bodies from March 2017 to September 2018. Visual analogue scale (VAS) scores, Oswestry disability index (ODI) values, and the number of opioids prescribed to the patients were assessed before and after PSP. The procedure duration, length of hospitalization, and complications were also recorded.
RESULTS
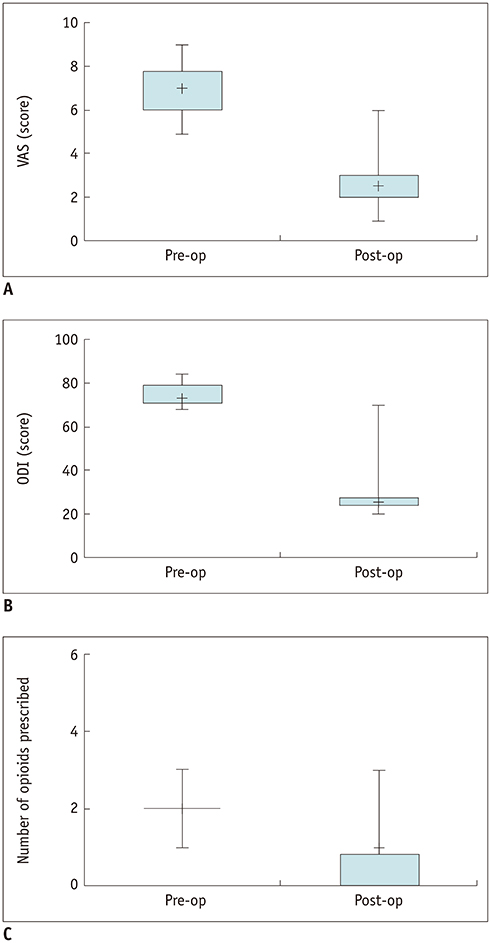
Mean VAS and ODI declined significantly from 6.90 ± 1.20 and 74.40 ± 5.48 before the procedure to 2.70 ± 1.34 and 29.60 ± 14.57 after the procedure, respectively (p < 0.01). The median number of opioids prescribed per patient decreased from 2 (interquartile range [IQR] 1-3) pre-procedure to 1 (IQR 0-3) post-procedure (p < 0.01). Nine of the 10 patients showed no or decreased opioid usage, and only 1 patient showed unchanged usage. The mean procedure duration was 48.5 ± 3.0 minutes. The average length of hospitalization was 4.7 ± 1.7 days. Extraosseous cement leakage occurred in three cases without causing any clinical complications.
CONCLUSION
PSP with an interpedicular approach is a safe and effective treatment in patients with painful sacral metastases involving multiple sacral vertebral bodies and can relieve pain and improve mobility.
MeSH Terms
Figure
Reference
-
1. Kortman K, Ortiz O, Miller T, Brook A, Tutton S, Mathis J, et al. Multicenter study to assess the efficacy and safety of sacroplasty in patients with osteoporotic sacral insufficiency fractures or pathologic sacral lesions. J Neurointerv Surg. 2013; 5:461–466.
Article2. Pereira LP, Clarençon F, Cormier E, Rose M, Jean B, Le Jean L, et al. Safety and effectiveness of percutaneous sacroplasty: a single-centre experience in 58 consecutive patients with tumors or osteoporotic insufficient fractures treated under fluoroscopic guidance. Eur Radiol. 2013; 23:2764–2772.3. Gupta AC, Chandra RV, Yoo AJ, Leslie-Mazwi TM, Bell DL, Mehta BP, et al. Safety and effectiveness of sacroplasty: a large single-center experience. Am J Neuroradiol. 2014; 35:2202–2206.
Article4. Dehdashti AR, Martin JB, Jean B, Rüfenacht DA. PMMA cementoplasty in symptomatic metastatic lesions of the S1 vertebral body. Cardiovasc Intervent Radiol. 2000; 23:235–237.
Article5. Talmadge J, Smith K, Dykes T, Mittleider D. Clinical impact of sacroplasty on patient mobility. J Vasc Interv Radiol. 2014; 25:911–915.
Article6. Frey ME, Warner C, Thomas SM, Johar K, Singh H, Mohammad MS, et al. Sacroplasty: a ten-year analysis of prospective patients treated with percutaneous sacroplasty: literature review and technical considerations. Pain Physician. 2017; 20:E1063–E1072.7. Yang SC, Tsai TT, Chen HS, Fang CJ, Kao YH, Tu YK. Comparison of sacroplasty with or without balloon assistance for the treatment of sacral insufficiency fractures. J Orthop Surg (Hong Kong). 2018; 26:2309499018782575.
Article8. Basile A, Tsetis D, Cavalli M, Fiumara P, Di Raimondo F, Coppolino F, et al. Sacroplasty for local or massive localization of multiple myeloma. Cardiovasc Intervent Radiol. 2010; 33:1270–1277.
Article9. Sun G, Jin P, Li M, Liu XW, Li FD. Three-dimensional C-arm computed tomography reformation combined with fluoroscopic-guided sacroplasty for sacral metastases. Support Care Cancer. 2012; 20:2083–2088.
Article10. Agarwal V, Sreedher G, Weiss KR, Hughes MA. Sacroplasty for symptomatic sacral hemangioma: a novel treatment approach. A case report. Interv Neuroradiol. 2013; 19:245–249.11. Andresen R, Radmer S, Lüdtke CW, Kamusella P, Wissgott C, Schober HC. Balloon sacroplasty as a palliative pain treatment in patients with metastasis-induced bone destruction and pathological fractures. Rofo. 2014; 186:881–886.
Article12. Moussazadeh N, Laufer I, Werner T, Krol G, Boland P, Bilsky MH, et al. Sacroplasty for cancer-associated insufficiency fractures. Neurosurgery. 2015; 76:446–450. discussion 450.
Article13. Nas OF, Kaçar E, Buyukkaya R, Şanal B, Erdogan C, Hakyemez B. Treatment of sacral aneurysmal bone cyst with percutaneous sacroplasty. Spine J. 2016; 16:e1–e2.
Article14. Burgard CA, Dinkel J, Strobl F, Paprottka PM, Schramm N, Reiser M, et al. CT fluoroscopy-guided percutaneous osteoplasty with or without radiofrequency ablation in the treatment of painful extraspinal and spinal bone metastases: technical outcome and complications in 29 patients. Diagn Interv Radiol. 2018; 24:158–165.
Article15. Zhang J, Wu CG, Gu YF, Li MH. Percutaneous sacroplasty for sacral metastatic tumors under fluoroscopic guidance only. Korean J Radiol. 2008; 9:572–576.
Article16. Firat AK, Gümüş B, Kaya E, Kuku I, Harma A. Interpedicular approach in percutaneous sacroplasty for treatment of sacral vertebral body pathologic fractures. Cardiovasc Intervent Radiol. 2011; 34:Suppl 2. S282–S287.
Article17. Dmytriw AA, Talla K, Smith R. Percutaneous sacroplasty for the management of painful pathologic fracture in a multiple myeloma patient: case report and review of the literature. Neuroradiol J. 2017; 30:80–83.
Article18. Miller JW, Diani A, Docsa S, Ashton K, Sciamanna M. Sacroplasty procedural extravasation with high viscosity bone cement: comparing the intraoperative long-axis versus short-axis techniques in osteoporotic cadavers. J Neurointerv Surg. 2017; 9:899–904.
Article19. Fairbank JC, Pynsent PB. The Oswestry disability index. Spine. 2000; 25:2940–2953.
Article20. Zanoli G, Strömqvist B, Jönsson B. Visual analog scales for interpretation of back and leg pain intensity in patients operated for degenerative lumbar spine disorders. Spine (Phila Pa 1976). 2001; 26:2375–2380.
Article21. Farrar JT, Young JP Jr, LaMoreaux L, Werth JL, Poole RM. Clinical importance of changes in chronic pain intensity measured on an 11-point numerical pain rating scale. Pain. 2001; 94:149–158.
Article22. Farrar JT, Berlin JA, Strom BL. Clinically important changes in acute pain outcome measures: a validation study. J Pain Symptom Manage. 2003; 25:406–411.23. Iannessi A, Amoretti N, Marcy PY, Sedat J. Percutaneous cementoplasty for the treatment of extraspinal painful bone lesion, a prospective study. Diagn Interv Imaging. 2012; 93:859–870.
Article24. Yin JJ, Pollock CB, Kelly K. Mechanisms of cancer metastasis to the bone. Cell Res. 2005; 15:57–62.
Article25. Nader R, Rhines LD, Mendel E. Metastatic sacral tumors. Neurosurg Clin N Am. 2004; 15:453–457.
Article26. Perrin RG, Laxton AW. Metastatic spine disease: epidemiology, pathophysiology, and evaluation of patients. Neurosurg Clin N Am. 2004; 15:365–373.
Article27. Finiels H, Finiels PJ, Jacquot JM, Strubel D. [Fractures of the sacrum caused by bone insufficiency. Meta-analysis of 508 cases]. Presse Med. 1997; 26:1568–1157.28. Douis H, James SL. CT-guided sacroplasty for the treatment of zone II sacral insuffciency fractures. Clin Radiol. 2009; 64:1037–1040.29. Hierholzer J, Anselmetti G, Fuchs H, Depriester C, Koch K, Pappert D. Percutaneous osteoplasty as a treatment for painful malignant bone lesions of the pelvis and femur. J Vasc Interv Radiol. 2003; 14:773–777.
Article30. Uemura A, Matsusako M, Numaguchi Y, Oka M, Kobayashi N, Niinami C, et al. Percutaneous sacroplasty for haemorrhagic metastases for hepatocellular carcinoma. AJNR Am J Neuroradiol. 2005; 26:493–495.31. Jayaraman MV, Chang H, Ahn SH. An easily identifable anatomic landmark for fluoroscopically guided sacroplasty: anatomic description and validation with treatment in 13 patients. AJNR Am J Neuroradiol. 2009; 30:1070–1073.32. Denis F. The three column spine and its significance in the classification of acute thoracolumbar spinal injuries. Spine (Phila Pa 1976). 1983; 8:817–831.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Interpedicular approach in percutaneous sacroplasty for treating pain due to direct invasion of rectal cancer into the S3 body: a case report
- Transiliac sacroplasty for Denis 3 fracture: Two cases report
- Percutaneous Sacroplasty for the Sacral Insufficiency Fracture Caused by Metastasis
- Therapeutic Considerations of Percutaneous Sacroplasty for the Sacral Insufficiency Fracture
- Percutaneous Sacroplasty for Sacral Metastatic Tumors Under Fluoroscopic Guidance Only