Dement Neurocogn Disord.
2017 Mar;16(1):26-31. 10.12779/dnd.2017.16.1.26.
Cerebral Perfusion Changes after Acetyl-L-Carnitine Treatment in Early Alzheimer's Disease Using Single Photon Emission Computed Tomography
- Affiliations
-
- 1Department of Radiology, Incheon St. Mary's Hospital, The Catholic University of Korea, Incheon, Korea.
- 2Department of Neurology, Incheon St. Mary's Hospital, The Catholic University of Korea, Incheon, Korea. siuy@catholic.ac.kr
- 3Department of Neurology, Veterans Hospital, Seoul Medical Center, Seoul, Korea.
- KMID: 2442832
- DOI: http://doi.org/10.12779/dnd.2017.16.1.26
Abstract
- BACKGROUND AND PURPOSE
Although acetyl-L-carnitine (ALC) treatment may have beneficial effects on Alzheimer's disease (AD), its underlying neural correlates remain unclear. The purpose of this study was to investigate cerebral perfusion changes after ALC treatment in AD patients using technetium-99m hexamethylpropylene amine oxime single photon emission computed tomography (SPECT).
METHODS
A total of 18 patients with early AD were prospectively recruited and treated with ALC at 1.5 g/day for 1.4±0.3 years. At baseline and follow-up, brain SPECT, Mini-Mental State Examination (MMSE), Clinical Dementia Rating (CDR), Global Deterioration Scale (GDS), and Neuropsychiatric Inventory (NPI) were used to assess participants. After ALC administration, changes in brain perfusion, severity of dementia, cognitive performance, and neuropsychiatric disturbances were examined.
RESULTS
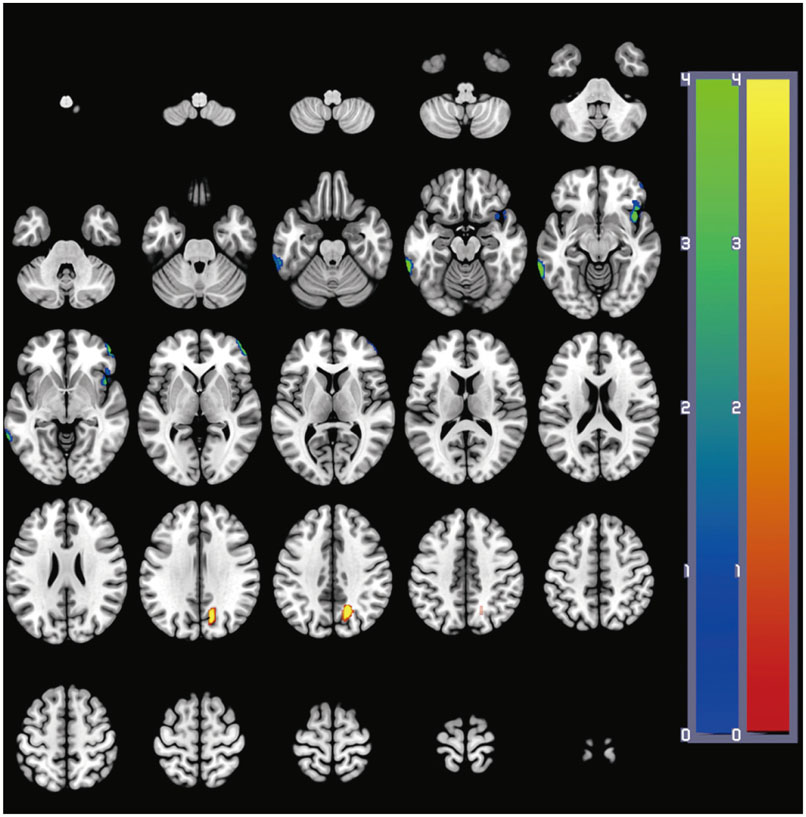
After ALC administration, changes in scores of MMSE, CDR, GDS, and NPI were not statistically significant (p>0.05). Voxel-wise whole-brain image analysis revealed that perfusion was significantly (p<0.001) increased in the right precuneus whereas perfusion was reduced in the left inferior temporal gyrus (p<0.001), the right middle frontal gyrus (p<0.001), and the right insular cortex (p=0.001) at follow-up.
CONCLUSIONS
Although previous studies have suggested that AD patients generally demonstrate progressive deterioration in brain perfusion and clinical symptoms, this study reveals that the perfusion of the precuneus is increased in AD patients after ALC administration and their cognitive and neuropsychiatric symptoms are not aggravated. Further studies are warranted to determine the potential association between perfusion increase in the precuneus and clinical symptoms after ALC treatment in AD patients.
Keyword
MeSH Terms
Figure
Reference
-
1. Helmes E, Østbye T. Beyond memory impairment: cognitive changes in Alzheimer's disease. Arch Clin Neuropsychol. 2002; 17:179–193.
Article2. Burlina AP, Sershen H, Debler EA, Lajtha A. Uptake of acetyl-L-carnitine in the brain. Neurochem Res. 1989; 14:489–493.
Article3. Pettegrew JW, Levine J, McClure RJ. Acetyl-L-carnitine physical-chemical, metabolic, and therapeutic properties: relevance for its mode of action in Alzheimer's disease and geriatric depression. Mol Psychiatry. 2000; 5:616–632.
Article4. Montgomery SA, Thal LJ, Amrein R. Meta-analysis of double blind randomized controlled clinical trials of acetyl-L-carnitine versus placebo in the treatment of mild cognitive impairment and mild Alzheimer's disease. Int Clin Psychopharmacol. 2003; 18:61–71.
Article5. Spagnoli A, Lucca U, Menasce G, Bandera L, Cizza G, Forloni G, et al. Long-term acetyl-L-carnitine treatment in Alzheimer's disease. Neurology. 1991; 41:1726–1732.
Article6. Pettegrew JW, Klunk WE, Panchalingam K, Kanfer JN, McClure RJ. Clinical and neurochemical effects of acetyl-L-carnitine in Alzheimer's disease. Neurobiol Aging. 1995; 16:1–4.
Article7. Henderson TA. The diagnosis and evaluation of dementia and mild cognitive impairment with emphasis on SPECT perfusion neuroimaging. CNS Spectr. 2012; 17:176–206.
Article8. Mazère J, Prunier C, Barret O, Guyot M, Hommet C, Guilloteau D, et al. In vivo SPECT imaging of vesicular acetylcholine transporter using [123 I]-IBVM in early Alzheimer's disease. Neuroimage. 2008; 40:280–288.
Article9. Kendziorra K, Wolf H, Meyer PM, Barthel H, Hesse S, Becker GA, et al. Decreased cerebral α4β2* nicotinic acetylcholine receptor availability in patients with mild cognitive impairment and Alzheimer's disease assessed with positron emission tomography. Eur J Nucl Med Mol Imaging. 2011; 38:515–525.
Article10. Minoshima S, Giordani B, Berent S, Frey KA, Foster NL, Kuhl DE. Metabolic reduction in the posterior cingulate cortex in very early Alzheimer's disease. Ann Neurol. 1997; 42:85–94.
Article11. American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th ed. Washington, DC: American Psychiatric Association;1994.12. McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology. 1984; 34:939–944.
Article13. Morris JC, Heyman A, Mohs RC, Hughes JP, van Belle G, Fillenbaum G, et al. The consortium to Establish a Registry for Alzheimer's disease (CERAD Clinical and neuropsychological assessment of Alzheimer's disease. Neurology. 1989; 39:1159–1165.14. Choi SH, Na DL, Lee BH, Hahm DS, Jeong JH, Jeong Y, et al. The validity of the Korean version of Global Deterioration Scale. J Korean Neurol Assoc. 2002; 20:612–617.15. Kang Y, Na DL, Hahn S. A validity study on the Korean Mini-Mental State Examination (K-MMSE) in dementia patients. J Korean Neurol Assoc. 1997; 15:300–308.16. Choi SH, Na DL, Kwon HM, Yoon SJ, Jeong JH, Ha CK. The Korean version of the neuropsychiatric inventory: a scoring tool for neuropsychiatric disturbance in dementia patients. J Korean Med Sci. 2000; 15:609–615.
Article17. Tzourio-Mazoyer N, Landeau B, Papathanassiou D, Crivello F, Etard O, Delcroix N, et al. Automated anatomical labeling of activations in SPM using a macroscopic anatomical parcellation of the MNI MRI single-subject brain. Neuroimage. 2002; 15:273–289.
Article18. Pickut BA, Dierckx RA, Dobbeleir A, Audenaert K, Van Laere K, Vervaet A, et al. Validation of the cerebellum as a reference region for SPECT quantification in patients suffering from dementia of the Alzheimer type. Psychiatry Res. 1999; 90:103–112.
Article19. Soonawala D, Amin T, Ebmeier KP, Steele JD, Dougall NJ, Best J, et al. Statistical parametric mapping of (99m)Tc-HMPAO-SPECT images for the diagnosis of Alzheimer's disease: normalizing to cerebellar tracer uptake. Neuroimage. 2002; 17:1193–1202.
Article20. Burns A, Jacoby R, Levy R. Progression of cognitive impairment in Alzheimer's disease. J Am Geriatr Soc. 1991; 39:39–45.
Article21. Benoit M, Robert PH, Staccini P, Brocker P, Guerin O, Lechowski L, et al. One-year longitudinal evaluation of neuropsychiatric symptoms in Alzheimer's disease. The REAL.FR study. J Nutr Health Aging. 2005; 9:95–99.22. Baron JC, Chételat G, Desgranges B, Perchey G, Landeau B, de la Sayette V, et al. In vivo mapping of gray matter loss with voxel-based morphometry in mild Alzheimer's disease. Neuroimage. 2001; 14:298–309.
Article23. Frisoni GB, Testa C, Zorzan A, Sabattoli F, Beltramello A, Soininen H, et al. Detection of grey matter loss in mild Alzheimer's disease with voxel based morphometry. J Neurol Neurosurg Psychiatry. 2002; 73:657–664.
Article24. Johnson NA, Jahng GH, Weiner MW, Miller BL, Chui HC, Jagust WJ, et al. Pattern of cerebral hypoperfusion in Alzheimer disease and mild cognitive impairment measured with arterial spin-labeling MR imaging: initial experience. Radiology. 2005; 234:851–859.
Article25. Mountjoy CQ, Roth M, Evans NJ, Evans HM. Cortical neuronal counts in normal elderly controls and demented patients. Neurobiol Aging. 1983; 4:1–11.
Article26. Royall DR. Insular Alzheimer disease pathology and the psychometric correlates of mortality. Cleve Clin J Med. 2008; 75:Suppl 2. S97–S99.
Article27. Scheff SW, Price DA, Schmitt FA, Scheff MA, Mufson EJ. Synaptic loss in the inferior temporal gyrus in mild cognitive impairment and Alzheimer's disease. J Alzheimers Dis. 2011; 24:547–557.
Article28. Cavanna AE, Trimble MR. The precuneus: a review of its functional anatomy and behavioural correlates. Brain. 2006; 129(Pt 3):564–583.
Article29. Fransson P, Marrelec G. The precuneus/posterior cingulate cortex plays a pivotal role in the default mode network: evidence from a partial correlation network analysis. Neuroimage. 2008; 42:1178–1184.
Article30. Rombouts SA, Barkhof F, Goekoop R, Stam CJ, Scheltens P. Altered resting state networks in mild cognitive impairment and mild Alzheimer's disease: an fMRI study. Hum Brain Mapp. 2005; 26:231–239.
Article31. Bradley KM, O'Sullivan VT, Soper ND, Nagy Z, King EM, Smith AD, et al. Cerebral perfusion SPET correlated with Braak pathological stage in Alzheimer's disease. Brain. 2002; 125(Pt 8):1772–1781.
Article32. Sakamoto S, Matsuda H, Asada T, Ohnishi T, Nakano S, Kanetaka H, et al. Apolipoprotein E genotype and early Alzheimer's disease: a longitudinal SPECT study. J Neuroimaging. 2003; 13:113–123.
Article33. Salmon E, Collette F, Degueldre C, Lemaire C, Franck G. Voxel-based analysis of confounding effects of age and dementia severity on cerebral metabolism in Alzheimer's disease. Hum Brain Mapp. 2000; 10:39–48.
Article34. Ryu SY, Kwon MJ, Lee SB, Yang DW, Kim TW, Song IU, et al. Measurement of precuneal and hippocampal volumes using magnetic resonance volumetry in Alzheimer's disease. J Clin Neurol. 2010; 6:196–203.
Article35. Sperling RA, Laviolette PS, O'Keefe K, O'Brien J, Rentz DM, Pihlajamaki M, et al. Amyloid deposition is associated with impaired default network function in older persons without dementia. Neuron. 2009; 63:178–188.
Article36. Petrella JR, Sheldon FC, Prince SE, Calhoun VD, Doraiswamy PM. Default mode network connectivity in stable vs progressive mild cognitive impairment. Neurology. 2011; 76:511–517.
Article37. Yun K, Song IU, Chung YA. Changes in cerebral glucose metabolism after 3 weeks of noninvasive electrical stimulation of mild cognitive impairment patients. Alzheimers Res Ther. 2016; 8:49.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- SPECT Findings on Psychiatric Symptoms in Alzheimer's Disease
- Differences of Regional Cerebral Blood Flow between Early Alzheimer's Disease and Parkinson's Disease Associated with Dementia on 99mTc-Hexamethyl Propylene Amine Oxime Perfusion Single Photon Emission Computed Tomography
- Specific SPECT Findings in Alzheimer's Dementia
- SPM Analysis of Changes in Cerebral Blood Flow in Alzheimer's Disease with Apathy
- Hyperglycemia-Induced Aphasia Presenting with Seizure-like Brain Perfusion Findings on Single Photon Emission Computed Tomography