J Breast Cancer.
2017 Sep;20(3):310-313. 10.4048/jbc.2017.20.3.310.
A Novel Germline Mutation in BRCA1 Causes Exon 20 Skipping in a Korean Family with a History of Breast Cancer
- Affiliations
-
- 1Department of Biochemistry, College of Veterinary Medicine, Konkuk University, Seoul, Korea.
- 2Genetic Counseling Clinic, National Cancer Center Hospital, National Cancer Center, Goyang, Korea.
- 3Department of System Cancer Science, Graduate School of Cancer Science and Policy, National Cancer Center, Goyang, Korea.
- 4Translational Epidemiology Branch, National Cancer Center Research Institute, National Cancer Center, Goyang, Korea.
- 5Department of Biomedical Sciences, Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea. suhwan.chang@amc.seoul.kr
- 6Center for Breast Cancer, National Cancer Center Hospital, National Cancer Center, Goyang, Korea.
- 7Precision Medicine Branch, National Cancer Center Research Institute, National Cancer Center, Goyang, Korea.
- KMID: 2438999
- DOI: http://doi.org/10.4048/jbc.2017.20.3.310
Abstract
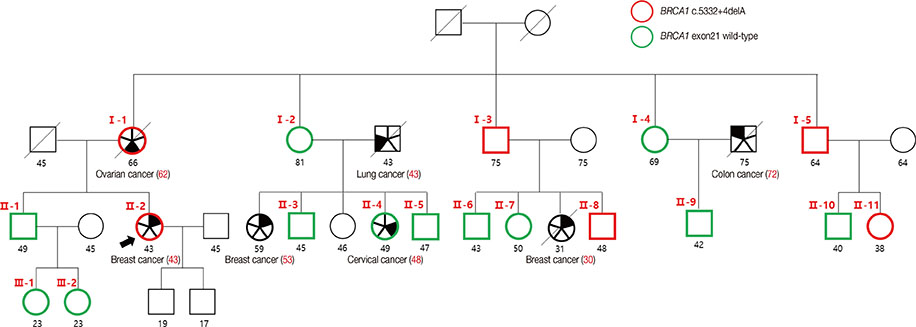
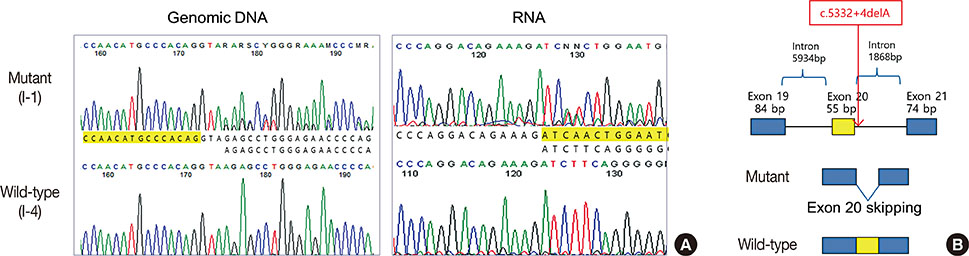
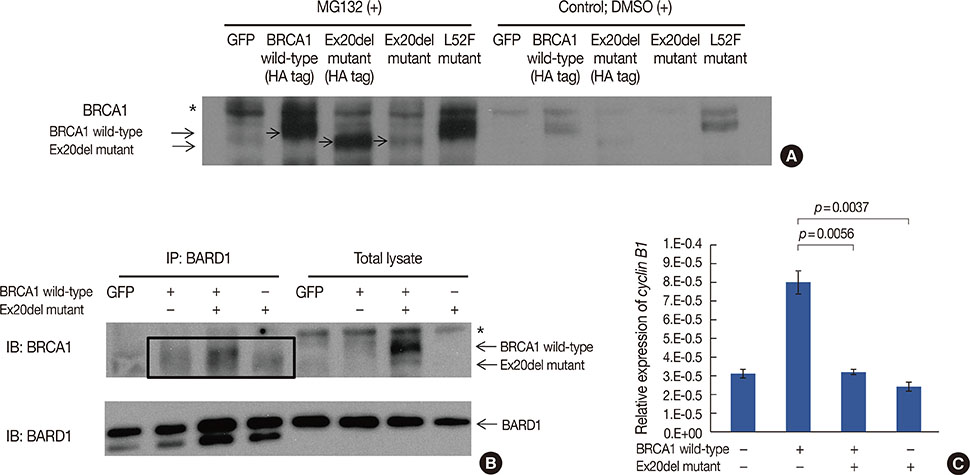
- Germline mutations in the BRCA1 and BRCA2 genes are strong genetic factors for predispositions to breast, ovarian, and other related cancers. This report describes a family with a history of breast and ovarian cancers that harbored a novel BRCA1 germline mutation. A single nucleotide deletion in intron 20, namely c.5332+4delA, was detected in a 43-year-old patient with breast cancer. This mutation led to the skipping of exon 20, which in turn resulted in the production of a truncated BRCA1 protein that was 1773 amino acids in length. The mother of the proband had died due to ovarian cancer and had harbored the same germline mutation. Ectopically expressed mutant BRCA1 protein interacted with the BARD1 protein, but showed a reduced transcriptional function, as demonstrated by the expression of cyclin B1. This novel germline mutation in the BRCA1 gene caused familial breast and ovarian cancers.
Keyword
MeSH Terms
Figure
Reference
-
1. Garber JE, Offit K. Hereditary cancer predisposition syndromes. J Clin Oncol. 2005; 23:276–292.
Article2. Lux MP, Fasching PA, Beckmann MW. Hereditary breast and ovarian cancer: review and future perspectives. J Mol Med (Berl). 2006; 84:16–28.
Article3. Hall MJ, Reid JE, Burbidge LA, Pruss D, Deffenbaugh AM, Frye C, et al. BRCA1 and BRCA2 mutations in women of different ethnicities undergoing testing for hereditary breast-ovarian cancer. Cancer. 2009; 115:2222–2233.
Article4. Narod SA. Modifiers of risk of hereditary breast cancer. Oncogene. 2006; 25:5832–5836.
Article5. Couch FJ, DeShano ML, Blackwood MA, Calzone K, Stopfer J, Campeau L, et al. BRCA1 mutations in women attending clinics that evaluate the risk of breast cancer. N Engl J Med. 1997; 336:1409–1415.
Article6. Ford D, Easton DF, Bishop DT, Narod SA, Goldgar DE. Risks of cancer in BRCA1-mutation carriers. Breast Cancer Linkage Consortium. Lancet. 1994; 343:692–695.7. Chen S, Parmigiani G. Meta-analysis of BRCA1 and BRCA2 penetrance. J Clin Oncol. 2007; 25:1329–1333.8. Langston AA, Malone KE, Thompson JD, Daling JR, Ostrander EA. BRCA1 mutations in a population-based sample of young women with breast cancer. N Engl J Med. 1996; 334:137–142.
Article9. Cartegni L, Chew SL, Krainer AR. Listening to silence and understanding nonsense: exonic mutations that affect splicing. Nat Rev Genet. 2002; 3:285–298.
Article10. Houdayer C, Caux-Moncoutier V, Krieger S, Barrois M, Bonnet F, Bourdon V, et al. Guidelines for splicing analysis in molecular diagnosis derived from a set of 327 combined in silico/in vitro studies on BRCA1 and BRCA2 variants. Hum Mutat. 2012; 33:1228–1238.
Article11. Liu HX, Cartegni L, Zhang MQ, Krainer AR. A mechanism for exon skipping caused by nonsense or missense mutations in BRCA1 and other genes. Nat Genet. 2001; 27:55–58.
Article12. Chang S, Biswas K, Martin BK, Stauffer S, Sharan SK. Expression of human BRCA1 variants in mouse ES cells allows functional analysis of BRCA1 mutations. J Clin Invest. 2009; 119:3160–3171.
Article13. Coquelle N, Green R, Glover JN. Impact of BRCA1 BRCT domain missense substitutions on phosphopeptide recognition. Biochemistry. 2011; 50:4579–4589.
Article14. Del Valle J, Campos O, Velasco A, Darder E, Menéndez M, Feliubadaló L, et al. Identification of a new complex rearrangement affecting exon 20 of BRCA1. Breast Cancer Res Treat. 2011; 130:341–344.
Article15. MacLachlan TK, Somasundaram K, Sgagias M, Shifman Y, Muschel RJ, Cowan KH, et al. BRCA1 effects on the cell cycle and the DNA damage response are linked to altered gene expression. J Biol Chem. 2000; 275:2777–2785.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- A Study for Germline Mutation of BRCA1 in Early Onset Breast Cancer Patients
- Frequency of BRCA1 and BRCA2 Germline Mutations Detected by Protein Truncation Test and Cumulative Risks of Breast and Ovarian Cancer among Mutation Carriers in Japanese Breast Cancer Families
- One Korean Patient with a Family History of BRCA1-associated Ovarian Cancer
- Novel Germline Mutations of BRCA1 and BRCA2 in Korean Familial Breast Cancer Patients
- A Study on BRCA1/2 Mutations, Hormone Status and HER-2 Status in Korean Women with Early-onset Breast Cancer