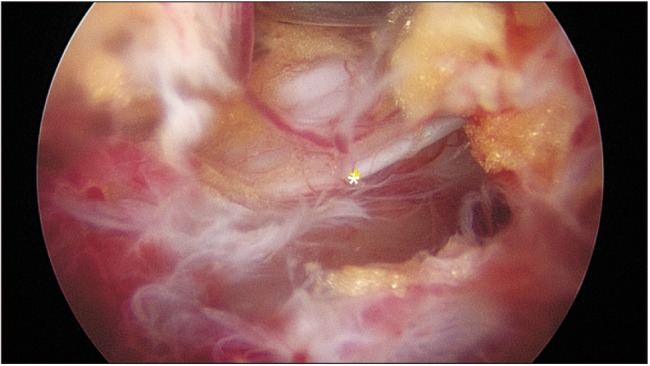
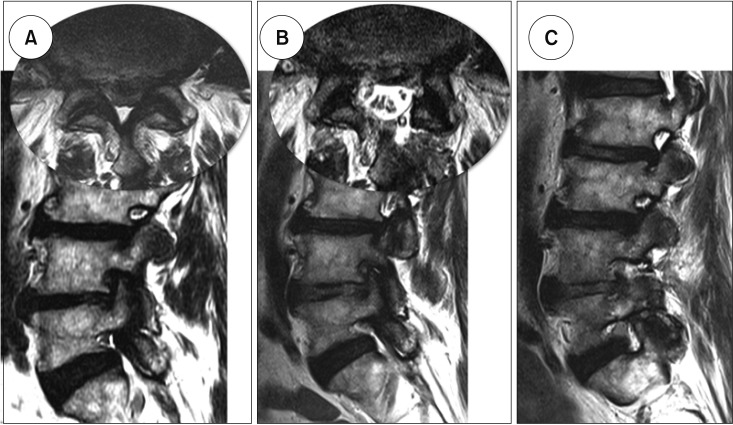
Clin Orthop Surg.
2019 Mar;11(1):82-88. 10.4055/cios.2019.11.1.82.
Efficacy of Biportal Endoscopic Spine Surgery for Lumbar Spinal Stenosis
- Affiliations
-
- 1Spine Center, Barun Hospital, Jinju, Korea. djchoi9@hanmail.net
- 2Department of Orthopedic Surgery, Andong Hospital, Andong, Korea.
- KMID: 2438337
- DOI: http://doi.org/10.4055/cios.2019.11.1.82
Abstract
- BACKGROUND
Biportal endoscopic spine surgery (BESS) is a recent addition to minimally invasive spine surgery treatments. It boasts excellent magnification and fine discrimination of neural structures. Selective decompression with preservation of facet joints for structural stability is also feasible owing to access to the spinal canal and foramen deeper inside. This study has a purpose to investigate clinical benefits of BESS for spinal stenosis in comparison to the other common surgical treatments such as microscopic decompression-only (DO) and fusion and instrumentation (FI).
METHODS
From December 2013 to March 2015, 30 cases of DO, 48 cases of FI, and 66 consecutive cases of BESS for lumbar spinal stenosis (LSS) were enrolled to evaluate the relative clinical efficacy of BESS. Visual analog scale (VAS) for back pain and leg pain, postoperative hemoglobin, C-reactive protein (CRP) changes, transfusion, and postoperative complications were examined.
RESULTS
All the patients were followed up until 6 months, and 98 patients (86.7%) for 2 years. At the 6-month follow-up, VAS for back pain improved from 6.8 to 2.8, 6.8 to 3.2, and 6.8 to 2.8 (p = 0.078) for BESS, DO, and FI, respectively; VAS for leg pain improved from 6.3 to 2.2, 7.0 to 2.5, and 7.2 to 2.5 (p = 0.291), respectively. Two cases in the BESS group underwent additional foraminal decompression, but no fusion surgery was performed. Postoperative hemoglobin changes for BESS, DO, and FI were −2.5, −2.4, and −1.3 mL, respectively. The BESS group had no transfusion cases, whereas 10 cases (33.3%) in DO and 41 cases (85.4%) in FI had transfusion (p = 0.000). CRP changes for BESS, DO, and FI were 0.32, 6.53, and 6.00, respectively, at day 2 postoperatively (p = 0.000); the complication rate for each group was 8.6% (two dural tears and one root injury), 6.7% (two dural tears), and 8.3% (two dural tears and two wound infections), respectively.
CONCLUSIONS
BESS for LSS showed clinical results not inferior to those of the other open surgery methods in the short-term. Stable hemodynamic changes with no need for blood transfusion and minimal changes in CRP were thought to cause less injury to the back muscles with minimal bleeding. Foraminal stenosis decompression should be simultaneously conducted with central decompression to avoid an additional surgery.
MeSH Terms
-
Back Muscles
Back Pain
Blood Transfusion
C-Reactive Protein
Constriction, Pathologic
Decompression
Discrimination (Psychology)
Endoscopy
Follow-Up Studies
Hemodynamics
Hemorrhage
Humans
Leg
Lumbar Vertebrae
Minimally Invasive Surgical Procedures
Pain, Postoperative
Postoperative Complications
Spinal Canal
Spinal Stenosis*
Spine*
Tears
Treatment Outcome
Visual Analog Scale
Wounds and Injuries
Zygapophyseal Joint
C-Reactive Protein
Figure
Reference
-
1. Polikandriotis JA, Hudak EM, Perry MW. Minimally invasive surgery through endoscopic laminotomy and foraminotomy for the treatment of lumbar spinal stenosis. J Orthop. 2013; 10(1):13–16. PMID: 24403742.
Article2. Wong AP, Smith ZA, Lall RR, Bresnahan LE, Fessler RG. The microendoscopic decompression of lumbar stenosis: a review of the current literature and clinical results. Minim Invasive Surg. Minim Invasive Surg. 2012; 2012:325095. PMID: 22900163.3. Birkenmaier C, Komp M, Leu HF, Wegener B, Ruetten S. The current state of endoscopic disc surgery: review of controlled studies comparing full-endoscopic procedures for disc herniations to standard procedures. Pain Physician. 2013; 16(4):335–344. PMID: 23877449.
Article4. Hu ZJ, Fang XQ, Zhou ZJ, Wang JY, Zhao FD, Fan SW. Effect and possible mechanism of muscle-splitting approach on multifidus muscle injury and atrophy after posterior lumbar spine surgery. J Bone Joint Surg Am. 2013; 95(24):e192(1-9). PMID: 24352778.
Article5. Huang YH, Ou CY. Significant blood loss in lumbar fusion surgery for degenerative spine. World Neurosurg. 2015; 84(3):780–785. PMID: 25986203.
Article6. Choi DJ, Jung JT, Lee SJ, Kim YS, Jang HJ, Yoo B. Biportal endoscopic spinal surgery for recurrent lumbar disc herniations. Clin Orthop Surg. 2016; 8(3):325–329. PMID: 27583117.
Article7. Choi DJ, Choi CM, Jung JT, Lee SJ, Kim YS. Learning curve associated with complications in biportal endoscopic spinal surgery: challenges and strategies. Asian Spine J. 2016; 10(4):624–629. PMID: 27559440.
Article8. Scholler K, Alimi M, Cong GT, Christos P, Hartl R. Lumbar spinal stenosis associated with degenerative lumbar spondylolisthesis: a systematic review and meta-analysis of secondary fusion rates following open vs minimally invasive decompression. Neurosurgery. 2017; 80(3):355–367. PMID: 28362963.9. Forsth P, Olafsson G, Carlsson T, et al. A randomized, controlled trial of fusion surgery for lumbar spinal stenosis. N Engl J Med. 2016; 374(15):1413–1423. PMID: 27074066.
Article10. Tai CL, Hsieh PH, Chen WP, Chen LH, Chen WJ, Lai PL. Biomechanical comparison of lumbar spine instability between laminectomy and bilateral laminotomy for spinal stenosis syndrome: an experimental study in porcine model. BMC Musculoskelet Disord. 2008; 9:84. PMID: 18547409.
Article11. Hartmann F, Janssen C, Bohm S, Hely H, Rommens PM, Gercek E. Biomechanical effect of graded minimal-invasive decompression procedures on lumbar spinal stability. Arch Orthop Trauma Surg. 2012; 132(9):1233–1239. PMID: 22592915.
Article12. Pao JL, Chen WC, Chen PQ. Clinical outcomes of microendoscopic decompressive laminotomy for degenerative lumbar spinal stenosis. Eur Spine J. 2009; 18(5):672–678. PMID: 19238459.
Article13. Takaso M, Nakazawa T, Imura T, et al. Less invasive and less technically demanding decompressive procedure for lumbar spinal stenosis: appropriate for general orthopaedic surgeons. Int Orthop. 2011; 35(1):67–73. PMID: 20229149.14. Mikami Y, Nagae M, Ikeda T, Tonomura H, Fujiwara H, Kubo T. Tubular surgery with the assistance of endoscopic surgery via midline approach for lumbar spinal canal stenosis: a technical note. Eur Spine J. 2013; 22(9):2105–2112. PMID: 23649957.
Article15. Gewurz H, Mold C, Siegel J, Fiedel B. C-reactive protein and the acute phase response. Adv Intern Med. 1982; 27:345–372. PMID: 7041546.16. Sproston NR, Ashworth JJ. Role of C-reactive protein at sites of inflammation and infection. Front Immunol. 2018; 9:754. PMID: 29706967.
Article17. Turner JA, Ersek M, Herron L, Deyo R. Surgery for lumbar spinal stenosis: attempted meta-analysis of the literature. Spine (Phila Pa 1976). 1992; 17(1):1–8. PMID: 1531550.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Current Status of Biportal Endoscopic Decompression for Lumbar Central Stenosis
- A Narrative Review of Biportal Endoscopic Spine Surgery in the Cervical and Thoracic Spine: Insights into Its Capabilities, Limitations, and Possibilities
- Complications and Management of Endoscopic Spinal Surgery
- Overview and Prevention of Complications During Biportal Endoscopic Lumbar Spine Surgery
- Biportal Endoscopic Transforaminal Lumbar Interbody Fusion with Arthroscopy