Yonsei Med J.
2019 Feb;60(2):119-125. 10.3349/ymj.2019.60.2.119.
Desensitization for Allergic Reactions to Chemotherapy
- Affiliations
-
- 1Division of Rheumatology, Immunology and Allergy, Brigham & Women's Hospital, Boston, MA, USA. dhong@bwh.harvard.edu
- KMID: 2431627
- DOI: http://doi.org/10.3349/ymj.2019.60.2.119
Abstract
- Drug desensitization is procedure by which patients can be tolerized to medications that have previously induced hypersensitivity reactions. Initially used in treating hypersensitivity reactions to antibiotics, desensitization is now frequently used in the setting of allergy to chemotherapy drugs and biologics, thus widening the clinical applicability of this procedure which has been proven to be quite safe and effective in improving clinical outcomes, mainly by allowing patients to remain on preferred first-line therapy. This paper reviews the history, application, and safety studies of drug desensitization for chemotherapy and biologics.
Keyword
MeSH Terms
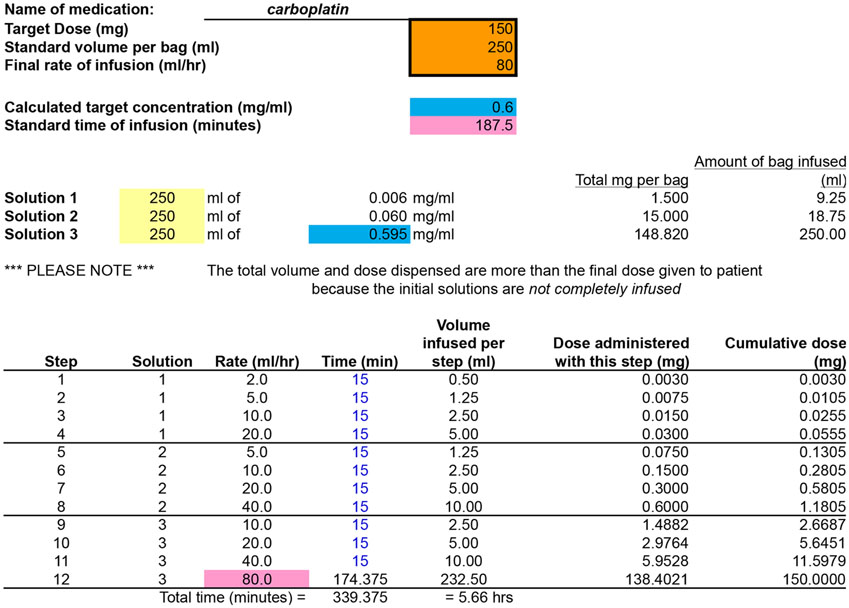
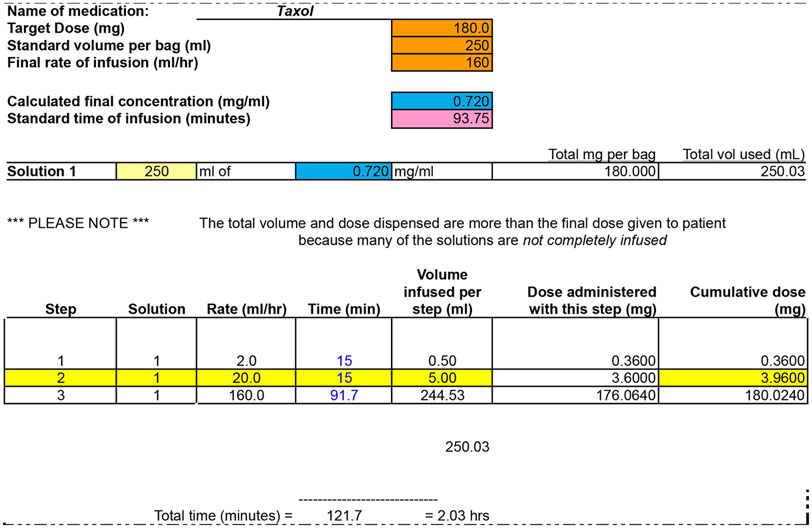
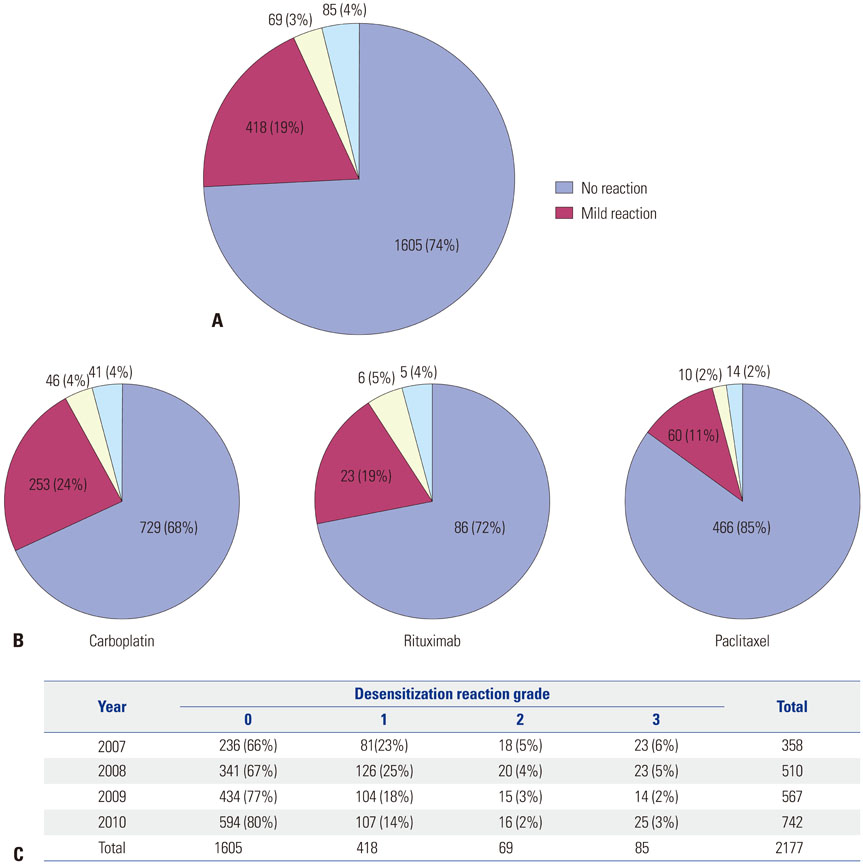
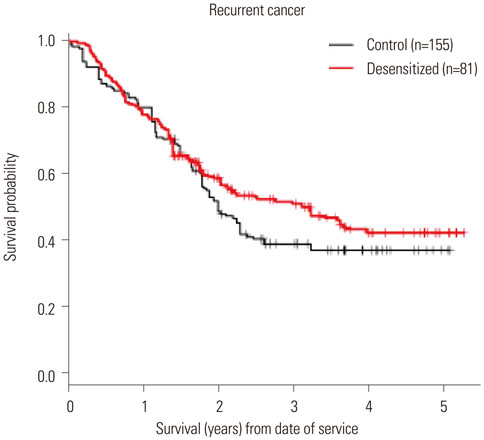
Figure
Reference
-
1. Brown SG, Stone SF, Fatovich DM, Burrows SA, Holdgate A, Celenza A, et al. Anaphylaxis: clinical patterns, mediator release, and severity. J Allergy Clin Immunol. 2013; 132:1141–1149.
Article2. Lenz HJ. Management and preparedness for infusion and hypersensitivity reactions. Oncologist. 2007; 12:601–609.
Article3. Brown SG. Clinical features and severity grading of anaphylaxis. J Allergy Clin Immunol. 2004; 114:371–376.
Article4. O’Donovan WJ, Klorfajn I. Sensitivity to penicillin; anaphylaxis and desensitisation. Lancet. 1946; 2:444–446.5. Peck SM, Siegal S, Bergamini R. Successful desensitization in penicillin sensitivity. J Am Med Assoc. 1947; 134:1546.
Article6. Gorman SK, Zed PJ, Dhingra VK, Ronco JJ. Rapid imipenem/cilastatin desensitization for multidrug-resistant Acinetobacter pneumonia. Ann Pharmacother. 2003; 37:513–516.
Article7. Papakonstantinou G, Bogner JR, Hofmeister F, Hehlmann R. Cefotaxime desensitization. Clin Investig. 1993; 71:165–167.
Article8. Lantner RR. Ciprofloxacin desensitization in a patient with cystic fibrosis. J Allergy Clin Immunol. 1995; 96(6 Pt 1):1001–1002.
Article9. Ozols RF, Bundy BN, Greer BE, Fowler JM, Clarke-Pearson D, Burger RA, et al. Phase III trial of carboplatin and paclitaxel compared with cisplatin and paclitaxel in patients with optimally resected stage III ovarian cancer: a Gynecologic Oncology Group study. J Clin Oncol. 2003; 21:3194–3200.
Article10. du Bois A, Lück HJ, Meier W, Adams HP, Möbus V, Costa S, et al. A randomized clinical trial of cisplatin/paclitaxel versus carboplatin/paclitaxel as first-line treatment of ovarian cancer. J Natl Cancer Inst. 2003; 95:1320–1329.
Article11. Lengyel E. Ovarian cancer development and metastasis. Am J Pathol. 2010; 177:1053–1064.
Article12. Markman M, Kennedy A, Webster K, Elson P, Peterson G, Kulp B, et al. Clinical features of hypersensitivity reactions to carboplatin. J Clin Oncol. 1999; 17:1141.
Article13. Markman M, Zanotti K, Peterson G, Kulp B, Webster K, Belinson J. Expanded experience with an intradermal skin test to predict for the presence or absence of carboplatin hypersensitivity. J Clin Oncol. 2003; 21:4611–4614.
Article14. Koshiba H, Hosokawa K, Kubo A, Miyagi Y, Oda T, Miyagi Y, et al. Incidence of carboplatin-related hypersensitivity reactions in Japanese patients with gynecologic malignancies. Int J Gynecol Cancer. 2009; 19:460–465.
Article15. Lax T, Long A, Banerji A. Skin testing in the evaluation and management of carboplatin-related hypersensitivity reactions. J Allergy Clin Immunol Pract. 2015; 3:856–862.
Article16. Weiss RB, Donehower RC, Wiernik PH, Ohnuma T, Gralla RJ, Trump DL, et al. Hypersensitivity reactions from taxol. J Clin Oncol. 1990; 8:1263–1268.
Article17. Markman M, Kennedy A, Webster K, Kulp B, Peterson G, Belinson J. Paclitaxel-associated hypersensitivity reactions: experience of the gynecologic oncology program of the Cleveland Clinic Cancer Center. J Clin Oncol. 2000; 18:102–105.
Article18. Lee CW, Matulonis UA, Castells MC. Carboplatin hypersensitivity: a 6-h 12-step protocol effective in 35 desensitizations in patients with gynecological malignancies and mast cell/IgE-mediated reactions. Gynecol Oncol. 2004; 95:370–376.
Article19. Castells MC, Tennant NM, Sloane DE, Hsu FI, Barrett NA, Hong DI, et al. Hypersensitivity reactions to chemotherapy: outcomes and safety of rapid desensitization in 413 cases. J Allergy Clin Immunol. 2008; 122:574–580.
Article20. Feldweg AM, Lee CW, Matulonis UA, Castells M. Rapid desensitization for hypersensitivity reactions to paclitaxel and docetaxel: a new standard protocol used in 77 successful treatments. Gynecol Oncol. 2005; 96:824–829.
Article21. Brennan PJ, Rodriguez Bouza T, Hsu FI, Sloane DE, Castells MC. Hypersensitivity reactions to mAbs: 105 desensitizations in 23 patients, from evaluation to treatment. J Allergy Clin Immunol. 2009; 124:1259–1266.
Article22. Dye D, Watkins J. Suspected anaphylactic reaction to Cremophor EL. Br Med J. 1980; 280:1353.
Article23. Lorenz W, Reimann HJ, Schmal A, Dormann P, Schwarz B, Neugebauer E, et al. Histamine release in dogs by Cremophor E1 and its derivatives: oxethylated oleic acid is the most effective constituent. Agents Actions. 1977; 7:63–67.
Article24. Weiszhár Z, Czúcz J, Révész C, Rosivall L, Szebeni J, Rozsnyay Z. Complement activation by polyethoxylated pharmaceutical surfactants: Cremophor-EL, Tween-80 and Tween-20. Eur J Pharm Sci. 2012; 45:492–498.
Article25. Hüttel MS, Schou Olesen A, Stoffersen E. Complement-mediated reactions to diazepam with Cremophor as solvent (Stesolid MR). Br J Anaesth. 1980; 52:77–79.
Article26. Fader AN, Rose PG. Abraxane for the treatment of gynecologic cancer patients with severe hypersensitivity reactions to paclitaxel. Int J Gynecol Cancer. 2009; 19:1281–1283.
Article27. Picard M, Pur L, Caiado J, Giavina-Bianchi P, Galvão VR, Berlin ST, et al. Risk stratification and skin testing to guide re-exposure in taxane-induced hypersensitivity reactions. J Allergy Clin Immunol. 2016; 137:1154–1164.
Article28. Ha E, Wang W, Wang YJ. Peroxide formation in polysorbate 80 and protein stability. J Pharm Sci. 2002; 91:2252–2264.
Article29. Commins SP, Platts-Mills TA. Anaphylaxis syndromes related to a new mammalian cross-reactive carbohydrate determinant. J Allergy Clin Immunol. 2009; 124:652–657.
Article30. Commins SP, James HR, Kelly LA, Pochan SL, Workman LJ, Perzanowski MS, et al. The relevance of tick bites to the production of IgE antibodies to the mammalian oligosaccharide galactose-α-1,3-galactose. J Allergy Clin Immunol. 2011; 127:1286–1293.
Article31. Chung CH, Mirakhur B, Chan E, Le QT, Berlin J, Morse M, et al. Cetuximab-induced anaphylaxis and IgE specific for galactose-alpha-1,3-galactose. N Engl J Med. 2008; 358:1109–1117.
Article32. Sloane D, Govindarajulu U, Harrow-Mortelliti J, Barry W, Hsu FI, Hong D, et al. Safety, costs, and efficacy of rapid drug desensitizations to chemotherapy and monoclonal antibodies. J Allergy Clin Immunol Pract. 2016; 4:497–504.
Article33. Gastaminza G, de la Borbolla JM, Goikoetxea MJ, Escudero R, Antón J, Espinós J, et al. A new rapid desensitization protocol for chemotherapy agents. J Investig Allergol Clin Immunol. 2011; 21:108–112.34. Madrigal-Burgaleta R, Berges-Gimeno MP, Angel-Pereira D, Ferreiro-Monteagudo R, Guillen-Ponce C, Pueyo C, et al. Hypersensitivity and desensitization to antineoplastic agents: outcomes of 189 procedures with a new short protocol and novel diagnostic tools assessment. Allergy. 2013; 68:853–861.
Article35. Madrigal-Burgaleta R, Bernal-Rubio L, Berges-Gimeno MP, Carpio-Escalona LV, Gehlhaar P, Alvarez-Cuesta E. A large single-hospital experience using drug provocation testing and rapid drug desensitization in hypersensitivity to antineoplastic and biological agents. J Allergy Clin Immunol Pract. 2018; 08. 08. [Epub]. DOI: 10.1016/j.jaip.2018.07.031.
Article36. Takase N, Matsumoto K, Onoe T, Kitao A, Tanioka M, Kikukawa Y, et al. 4-step 4-h carboplatin desensitization protocol for patients with gynecological malignancies showing platinum hypersensitivity: a retrospective study. Int J Clin Oncol. 2015; 20:566–573.
Article37. Altwerger G, Gressel GM, English DP, Nelson WK, Carusillo N, Silasi DA, et al. Platinum desensitization in patients with carboplatin hypersensitivity: a single-institution retrospective study. Gynecol Oncol. 2017; 144:77–82.
Article38. Lieberman P, Nicklas RA, Randolph C, Oppenheimer J, Bernstein D, Bernstein J, et al. Anaphylaxis--a practice parameter update 2015. Ann Allergy Asthma Immunol. 2015; 115:341–384.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Clinical applications of drug desensitization in the Asia-Pacific region
- Cyclosporine Desensitization in Patient with Multiple Hypersensitivity Reactions Immediately after Peripheral Blood Stem Cell Transplantation
- Updates on desensitization for hypersensitivity reactions related to chemotherapy
- A case of anaphylaxis to human recombinant insulin treated by desensitization
- Current Issues in Vaccine Allergy