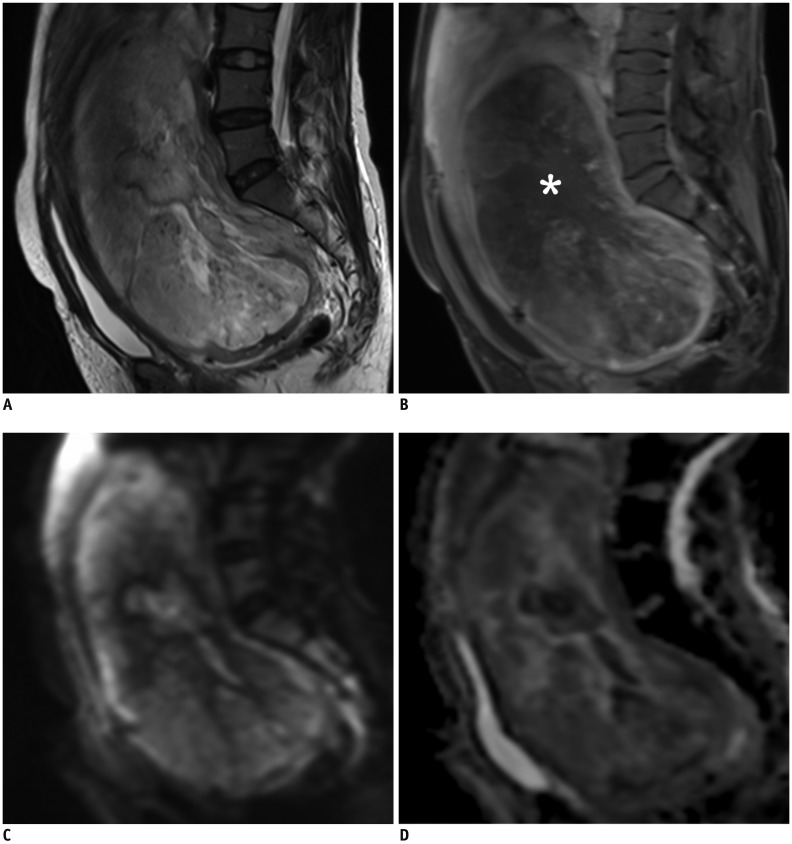
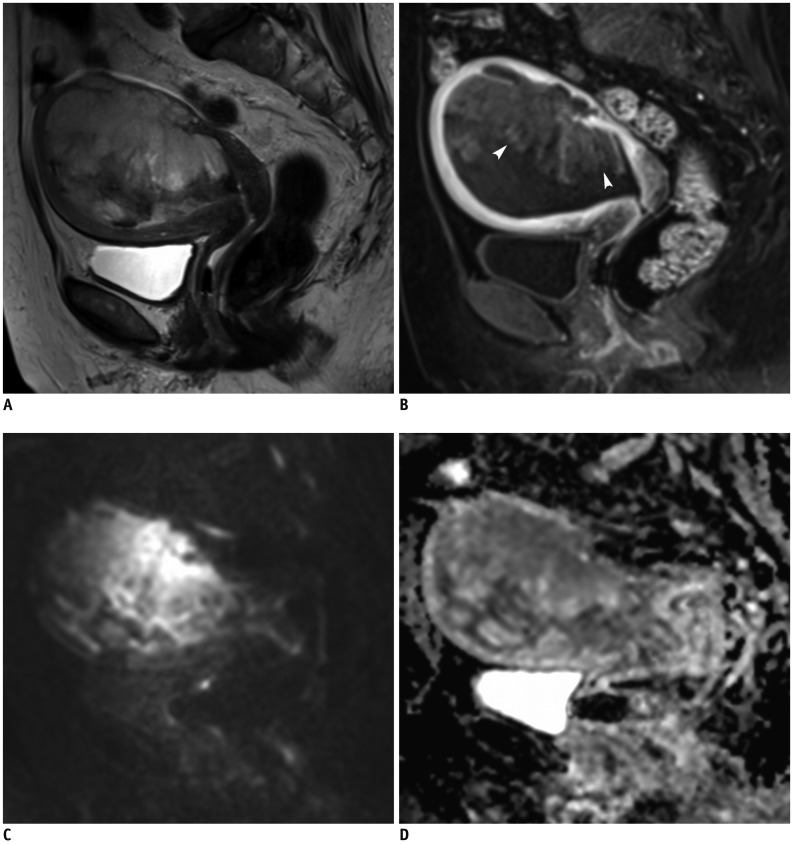
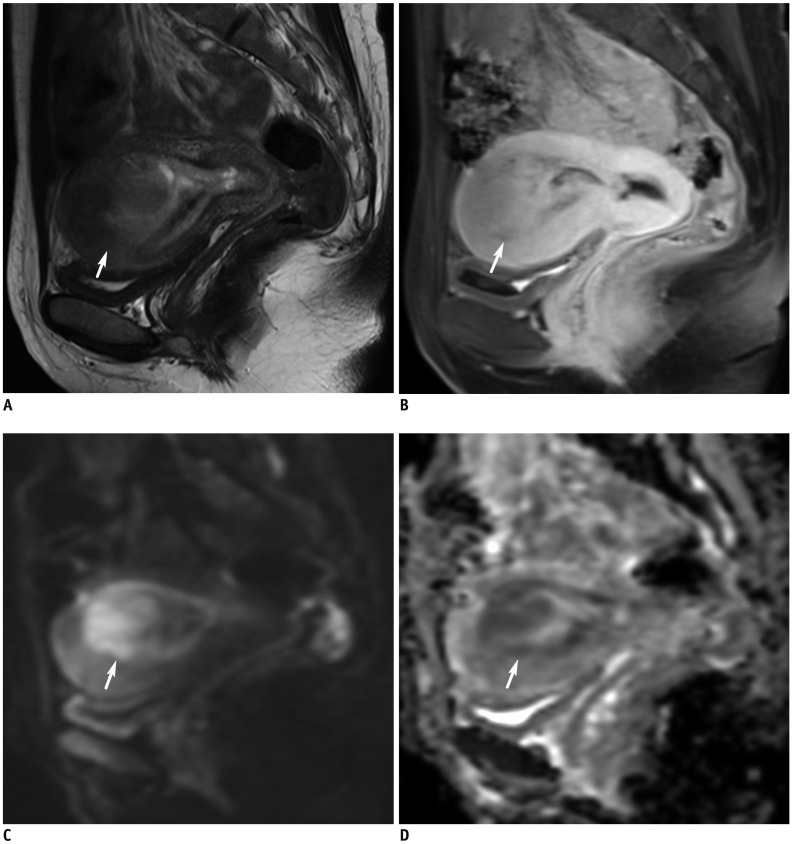
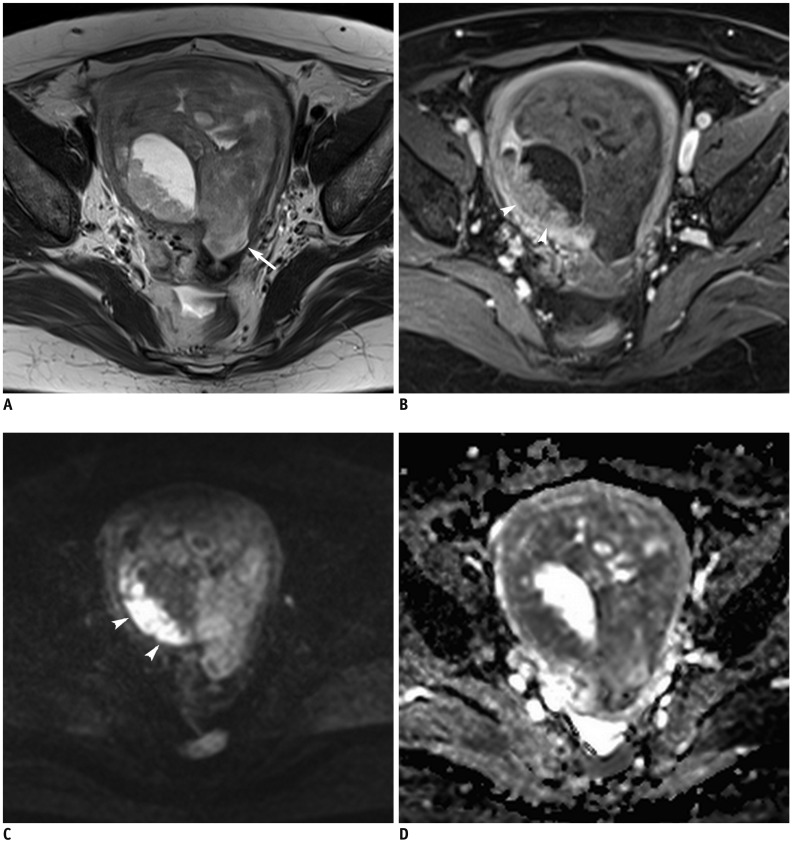
Korean J Radiol.
2019 Jan;20(1):18-33. 10.3348/kjr.2018.0090.
Current Status of Magnetic Resonance Imaging in Patients with Malignant Uterine Neoplasms: A Review
- Affiliations
-
- 1Department of Medical Imaging and Intervention, Imaging Core Laboratory, Institute for Radiological Research, Chang Gung Memorial Hospital at Linkou, Taoyuan, Taiwan. giginlin@cgmh.org.tw
- 2Department of Diagnostic Radiology, Chang Gung Memorial Hospital at Keelung, Keelung, Taiwan.
- 3Clinical Metabolomic Core Laboratory, Chang Gung Memorial Hospital at Linkou, Chang Gung University, Taoyuan, Taiwan.
- 4Gynecologic Cancer Research Center, Chang Gung Memorial Hospital, Chang Gung University College of Medicine, Taoyuan, Taiwan.
- KMID: 2429917
- DOI: http://doi.org/10.3348/kjr.2018.0090
Abstract
- In this study, we summarize the clinical role of magnetic resonance imaging (MRI) in the diagnosis of patients with malignant uterine neoplasms, including leiomyosarcoma, endometrial stromal sarcoma, adenosarcoma, uterine carcinosarcoma, and endometrial cancer, with emphasis on the challenges and disadvantages. MRI plays an essential role in patients with uterine malignancy, for the purpose of tumor detection, primary staging, and treatment planning. MRI has advanced in scope beyond the visualization of the many aspects of anatomical structures, including diffusion-weighted imaging, dynamic contrast enhancement-MRI, and magnetic resonance spectroscopy. Emerging technologies coupled with the use of artificial intelligence in MRI are expected to lead to progressive improvement in case management of malignant uterine neoplasms.
MeSH Terms
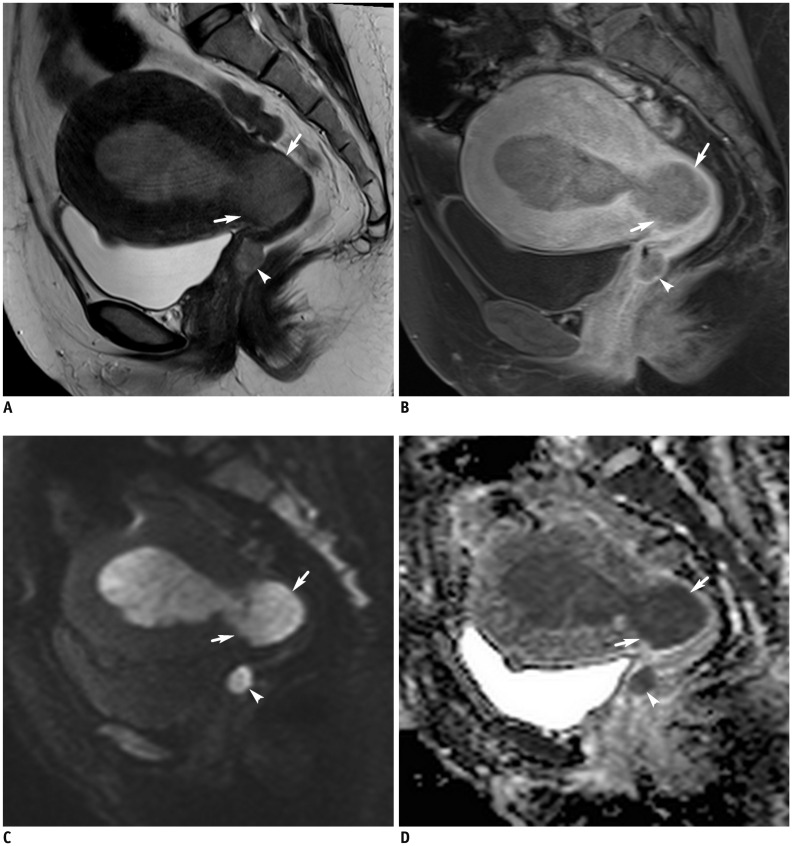
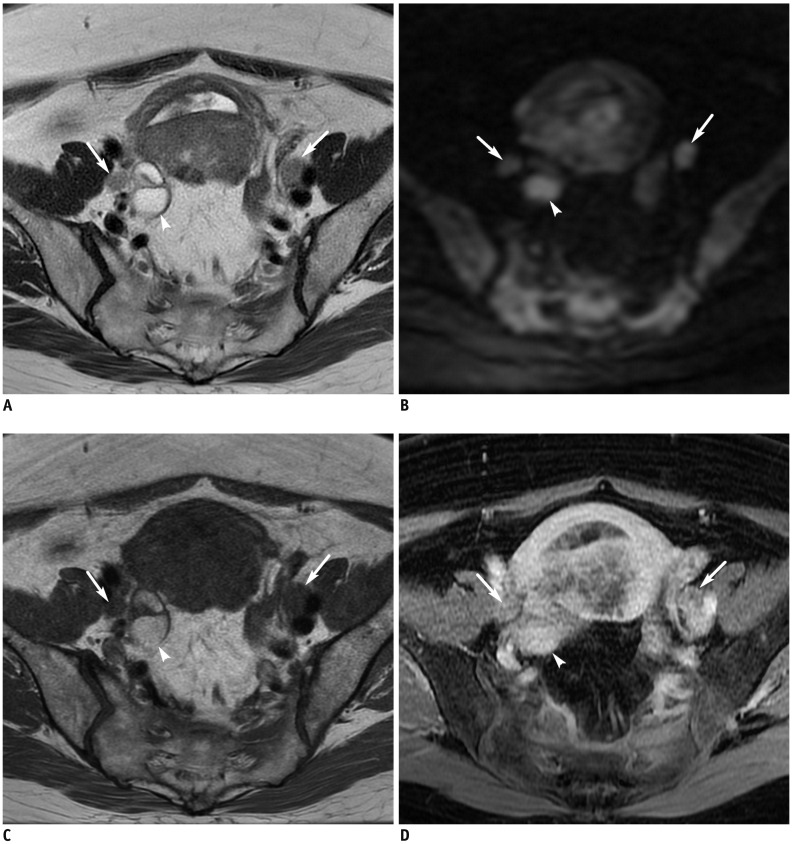
Figure
Reference
-
1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin. 2018; 68:7–30. PMID: 29313949.
Article2. Global Cancer Observatory. International Agency for Research on Cancer (IARC) web site. 2014. Accessed May 11, 2018. http://globocan.iarc.fr/.3. Kurman RJ, Carcangiu ML, Herrington CS, Young RH. WHO classification of tumours of female reproductive organs, Volume 6. 4th ed. Geneva: World Health Organization;2014. p. 141–145.4. D'Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol. 2010; 116:131–139. PMID: 19853898.5. Koh WJ, Abu-Rustum NR, Bean S, Bradley K, Campos SM, Cho KR, et al. Uterine neoplasms, Version 1.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2018; 16:170–199. PMID: 29439178.
Article6. Patel-Lippmann K, Robbins JB, Barroilhet L, Anderson B, Sadowski EA, Boyum J. MR imaging of cervical cancer. Magn Reson Imaging Clin N Am. 2017; 25:635–649. PMID: 28668164.
Article7. Sala E, Rockall AG, Freeman SJ, Mitchell DG, Reinhold C. The added role of MR imaging in treatment stratification of patients with gynecologic malignancies: what the radiologist needs to know. Radiology. 2013; 266:717–740. PMID: 23431227.
Article8. Padhani AR, Koh DM, Collins DJ. Whole-body diffusion-weighted MR imaging in cancer: current status and research directions. Radiology. 2011; 261:700–718. PMID: 22095994.
Article9. Leach MO, Morgan B, Tofts PS, Buckley DL, Huang W, Horsfield MA, et al. Experimental Cancer Medicine Centres Imaging Network Steering Committee. Experimental Cancer Medicine Centres Imaging Network Steering Committee. Imaging vascular function for early stage clinical trials using dynamic contrast-enhanced magnetic resonance imaging. Eur Radiol. 2012; 22:1451–1464. PMID: 22562143.10. Goto A, Takeuchi S, Sugimura K, Maruo T. Usefulness of Gd-DTPA contrast-enhanced dynamic MRI and serum determination of LDH and its isozymes in the differential diagnosis of leiomyosarcoma from degenerated leiomyoma of the uterus. Int J Gynecol Cancer. 2002; 12:354–361. PMID: 12144683.
Article11. Lin G, Lin YC, Wu RC, Yang LY, Lu HY, Tsai SY, et al. Developing and validating a multivariable prediction model to improve the diagnostic accuracy in determination of cervical versus endometrial origin of uterine adenocarcinomas: a prospective MR study combining diffusion-weighted imaging and spectroscopy. J Magn Reson Imaging. 2018; 47:1654–1666. PMID: 29178414.
Article12. Cornfeld D, Israel G, Martel M, Weinreb J, Schwartz P, McCarthy S. MRI appearance of mesenchymal tumors of the uterus. Eur J Radiol. 2010; 74:241–249. PMID: 19349135.
Article13. Parker WH, Fu YS, Berek JS. Uterine sarcoma in patients operated on for presumed leiomyoma and rapidly growing leiomyoma. Obstet Gynecol. 1994; 83:414–418. PMID: 8127535.14. Giuntoli RL 2nd, Metzinger DS, DiMarco CS, Cha SS, Sloan JA, Keeney GL, et al. Retrospective review of 208 patients with leiomyosarcoma of the uterus: prognostic indicators, surgical management, and adjuvant therapy. Gynecol Oncol. 2003; 89:460–469. PMID: 12798712.
Article15. Wu TI, Yen TC, Lai CH. Clinical presentation and diagnosis of uterine sarcoma, including imaging. Best Pract Res Clin Obstet Gynaecol. 2011; 25:681–689. PMID: 21816678.
Article16. Lakhman Y, Veeraraghavan H, Chaim J, Feier D, Goldman DA, Moskowitz CS, et al. Differentiation of uterine leiomyosarcoma from atypical leiomyoma: diagnostic accuracy of qualitative MR imaging features and feasibility of texture analysis. Eur Radiol. 2017; 27:2903–2915. PMID: 27921159.
Article17. Shah SH, Jagannathan JP, Krajewski K, O'Regan KN, George S, Ramaiya NH. Uterine sarcomas: then and now. AJR Am J Roentgenol. 2012; 199:213–223. PMID: 22733915.
Article18. Tanaka YO, Nishida M, Tsunoda H, Okamoto Y, Yoshikawa H. Smooth muscle tumors of uncertain malignant potential and leiomyosarcomas of the uterus: MR findings. J Magn Reson Imaging. 2004; 20:998–1007. PMID: 15558559.
Article19. Thomassin-Naggara I, Dechoux S, Bonneau C, Morel A, Rouzier R, Carette MF, et al. How to differentiate benign from malignant myometrial tumours using MR imaging. Eur Radiol. 2013; 23:2306–2314. PMID: 23563602.
Article20. Tamai K, Koyama T, Saga T, Morisawa N, Fujimoto K, Mikami Y, et al. The utility of diffusion-weighted MR imaging for differentiating uterine sarcomas from benign leiomyomas. Eur Radiol. 2008; 18:723–730. PMID: 17929022.
Article21. Namimoto T, Yamashita Y, Awai K, Nakaura T, Yanaga Y, Hirai T, et al. Combined use of T2-weighted and diffusion-weighted 3-T MR imaging for differentiating uterine sarcomas from benign leiomyomas. Eur Radiol. 2009; 19:2756–2764. PMID: 19504102.
Article22. Ueda H, Togashi K, Konishi I, Kataoka ML, Koyama T, Fujiwara T, et al. Unusual appearances of uterine leiomyomas: MR imaging findings and their histopathologic backgrounds. Radiographics. 1999; 19 Spec No:S131–S145. PMID: 10517450.
Article23. Hricak H, Tscholakoff D, Heinrichs L, Fisher MR, Dooms GC, Reinhold C, et al. Uterine leiomyomas: correlation of MR, histopathologic findings, and symptoms. Radiology. 1986; 158:385–391. PMID: 3753623.
Article24. Lin G, Yang LY, Huang YT, Ng KK, Ng SH, Ueng SH, et al. Comparison of the diagnostic accuracy of contrast-enhanced MRI and diffusion-weighted MRI in the differentiation between uterine leiomyosarcoma / smooth muscle tumor with uncertain malignant potential and benign leiomyoma. J Magn Reson Imaging. 2016; 43:333–342. PMID: 26383110.
Article25. Li HM, Liu J, Qiang JW, Zhang H, Zhang GF, Ma F. Diffusion-weighted imaging for differentiating uterine leiomyosarcoma from degenerated leiomyoma. J Comput Assist Tomogr. 2017; 41:599–606. PMID: 27997438.
Article26. Sato K, Yuasa N, Fujita M, Fukushima Y. Clinical application of diffusion-weighted imaging for preoperative differentiation between uterine leiomyoma and leiomyosarcoma. Am J Obstet Gynecol. 2014; 210:368.e1–368.e8. PMID: 24368137.
Article27. Tropé CG, Abeler VM, Kristensen GB. Diagnosis and treatment of sarcoma of the uterus. A review. Acta Oncol. 2012; 51:694–705. PMID: 22793037.
Article28. Tse KY, Crawford R, Ngan HY. Staging of uterine sarcomas. Best Pract Res Clin Obstet Gynaecol. 2011; 25:733–749. PMID: 21752716.
Article29. Gadducci A, Cosio S, Romanini A, Genazzani AR. The management of patients with uterine sarcoma: a debated clinical challenge. Crit Rev Oncol Hematol. 2008; 65:129–142. PMID: 17706430.
Article30. Tirumani SH, Ojili V, Shanbhogue AK, Fasih N, Ryan JG, Reinhold C. Current concepts in the imaging of uterine sarcoma. Abdom Imaging. 2013; 38:397–411. PMID: 22699695.
Article31. Furukawa R, Akahane M, Yamada H, Kiryu S, Sato J, Komatsu S, et al. Endometrial stromal sarcoma located in the myometrium with a low-intensity rim on T2-weighted images: report of three cases and literature review. J Magn Reson Imaging. 2010; 31:975–979. PMID: 20373443.
Article32. Koyama T, Togashi K, Konishi I, Kobayashi H, Ueda H, Kataoka ML, et al. MR imaging of endometrial stromal sarcoma: correlation with pathologic findings. AJR Am J Roentgenol. 1999; 173:767–772. PMID: 10470920.
Article33. Ueda M, Otsuka M, Hatakenaka M, Sakai S, Ono M, Yoshimitsu K, et al. MR imaging findings of uterine endometrial stromal sarcoma: differentiation from endometrial carcinoma. Eur Radiol. 2001; 11:28–33. PMID: 11194912.
Article34. Boskovic V, Bozanovic T, Ljubic A, Likic-Ladjevic I, Janjic T, Milicevic S. Endometrial stromal sarcoma with intracaval extension at initial presentation. Eur J Gynaecol Oncol. 2013; 34:280–281. PMID: 23967567.35. Huang YL, Huang YT, Ng KK, Lai CH, Lin G. Differentiating low-grade endometrial stromal sarcoma and undifferentiated endometrial sarcoma by using magnetic resonance imaging, Abstract No. ESGO7-0441. In : Proceeding of International Meeting of the European Society of Gynaecological Oncology; 2017 November 4-7; Vienna, Austria.36. Santos P, Cunha TM. Uterine sarcomas: clinical presentation and MRI features. Diagn Interv Radiol. 2015; 21:4–9. PMID: 25347940.
Article37. Arend R, Bagaria M, Lewin SN, Sun X, Deutsch I, Burke WM, et al. Long-term outcome and natural history of uterine adenosarcomas. Gynecol Oncol. 2010; 119:305–308. PMID: 20688363.
Article38. Takeuchi M, Matsuzaki K, Yoshida S, Kudo E, Bando Y, Hasebe H, et al. Adenosarcoma of the uterus: magnetic resonance imaging characteristics. Clin Imaging. 2009; 33:244–247. PMID: 19411035.
Article39. Yoshizako T, Wada A, Kitagaki H, Ishikawa N, Miyazaki K. MR imaging of uterine adenosarcoma: case report and literature review. Magn Reson Med Sci. 2011; 10:251–254. PMID: 22214910.
Article40. Barral M, Placé V, Dautry R, Bendavid S, Cornelis F, Foucher R, et al. Magnetic resonance imaging features of uterine sarcoma and mimickers. Abdom Radiol (NY). 2017; 42:1762–1772. PMID: 28246921.
Article41. Artioli G, Wabersich J, Ludwig K, Gardiman MP, Borgato L, Garbin F. Rare uterine cancer: carcinosarcomas. Review from histology to treatment. Crit Rev Oncol Hematol. 2015; 94:98–104. PMID: 25468677.
Article42. Arend R, Doneza JA, Wright JD. Uterine carcinosarcoma. Curr Opin Oncol. 2011; 23:531–536. PMID: 21743326.
Article43. Menczer J. Review of recommended treatment of uterine carcinosarcoma. Curr Treat Options Oncol. 2015; 16:53. PMID: 26374341.
Article44. Lewin SN. Revised FIGO staging system for endometrial cancer. Clin Obstet Gynecol. 2011; 54:215–218. PMID: 21508690.
Article45. Beddy P, Moyle P, Kataoka M, Yamamoto AK, Joubert I, Lomas D, et al. Evaluation of depth of myometrial invasion and overall staging in endometrial cancer: comparison of diffusion-weighted and dynamic contrast-enhanced MR imaging. Radiology. 2012; 262:530–537. PMID: 22114239.
Article46. Lin G, Ng KK, Chang CJ, Wang JJ, Ho KC, Yen TC, et al. Myometrial invasion in endometrial cancer: diagnostic accuracy of diffusion-weighted 3.0-T MR imaging--initial experience. Radiology. 2009; 250:784–792. PMID: 19244045.
Article47. Bharwani N, Newland A, Tunariu N, Babar S, Sahdev A, Rockall AG, et al. MRI appearances of uterine malignant mixed müllerian tumors. AJR Am J Roentgenol. 2010; 195:1268–1275. PMID: 20966339.
Article48. Genever AV, Abdi S. Can MRI predict the diagnosis of endometrial carcinosarcoma? Clin Radiol. 2011; 66:621–624. PMID: 21507390.
Article49. Tanaka YO, Tsunoda H, Minami R, Yoshikawa H, Minami M. Carcinosarcoma of the uterus: MR findings. J Magn Reson Imaging. 2008; 28:434–439. PMID: 18666176.
Article50. Takeuchi M, Matsuzaki K, Harada M. Carcinosarcoma of the uterus: MRI findings including diffusion-weighted imaging and MR spectroscopy. Acta Radiol. 2016; 57:1277–1284. PMID: 26787673.
Article51. Kato H, Kanematsu M, Furui T, Imai A, Hirose Y, Kondo H, et al. Carcinosarcoma of the uterus: radiologic-pathologic correlations with magnetic resonance imaging including diffusion-weighted imaging. Magn Reson Imaging. 2008; 26:1446–1450. PMID: 18501546.
Article52. Hernández Mateo P, Méndez Fernández R, Serrano Tamayo E. Uterine sarcoma vs adenocarcinoma: can MRI distinguish between them. Radiologia. 2016; 58:199–206. PMID: 26617259.53. Huang YT, Chang CB, Yeh CJ, Lin G, Huang HJ, Wang CC, et al. Diagnostic accuracy of 3.0T diffusion-weighted MRI for patients with uterine carcinosarcoma: assessment of tumor extent and lymphatic metastasis. J Magn Reson Imaging. 2018; 48:622–631.
Article54. Smith-Bindman R, Kerlikowske K, Feldstein VA, Subak L, Scheidler J, Segal M, et al. Endovaginal ultrasound to exclude endometrial cancer and other endometrial abnormalities. JAMA. 1998; 280:1510–1517. PMID: 9809732.
Article55. Felix AS, Weissfeld JL, Stone RA, Bowser R, Chivukula M, Edwards RP, et al. Factors associated with Type I and Type II endometrial cancer. Cancer Causes Control. 2010; 21:1851–1856. PMID: 20628804.
Article56. Amant F, Mirza MR, Koskas M, Creutzberg CL. Cancer of the corpus uteri. Int J Gynaecol Obstet. 2015; 131(Suppl 2):S96–S104. PMID: 26433681.
Article57. Creasman W. Revised FIGO staging for carcinoma of the endometrium. Int J Gynaecol Obstet. 2009; 105:109. PMID: 19345353.
Article58. Kinkel K, Kaji Y, Yu KK, Segal MR, Lu Y, Powell CB, et al. Radiologic staging in patients with endometrial cancer: a meta-analysis. Radiology. 1999; 212:711–718. PMID: 10478237.
Article59. Fujii S, Kido A, Baba T, Fujimoto K, Daido S, Matsumura N, et al. Subendometrial enhancement and peritumoral enhancement for assessing endometrial cancer on dynamic contrast enhanced MR imaging. Eur J Radiol. 2015; 84:581–589. PMID: 25636389.
Article60. Andreano A, Rechichi G, Rebora P, Sironi S, Valsecchi MG, Galimberti S. MR diffusion imaging for preoperative staging of myometrial invasion in patients with endometrial cancer: a systematic review and meta-analysis. Eur Radiol. 2014; 24:1327–1338. PMID: 24668009.
Article61. Ota T, Hori M, Onishi H, Sakane M, Tsuboyama T, Tatsumi M, et al. Preoperative staging of endometrial cancer using reduced field-of-view diffusion-weighted imaging: a preliminary study. Eur Radiol. 2017; 27:5225–5235. PMID: 28674966.
Article62. Takeuchi M, Matsuzaki K, Harada M. Evaluating myometrial invasion in endometrial cancer: comparison of reduced field-of-view diffusion-weighted imaging and dynamic contrast-enhanced MR imaging. Magn Reson Med Sci. 2018; 17:28–34. PMID: 28515411.
Article63. Bhosale P, Ma J, Iyer R, Ramalingam P, Wei W, Soliman P, et al. Feasibility of a reduced field-of-view diffusion-weighted (rFOV) sequence in assessment of myometrial invasion in patients with clinical FIGO stage I endometrial cancer. J Magn Reson Imaging. 2016; 43:316–324. PMID: 26185031.
Article64. Eskander RN, Randall LM, Berman ML, Tewari KS, Disaia PJ, Bristow RE. Fertility preserving options in patients with gynecologic malignancies. Am J Obstet Gynecol. 2011; 205:103–110. PMID: 21411052.
Article65. Lin G, Huang YT, Chao A, Ng KK, Yang LY, Ng SH, et al. Influence of menopausal status on diagnostic accuracy of myometrial invasion in endometrial cancer: diffusion-weighted and dynamic contrast-enhanced MRI at 3 T. Clin Radiol. 2015; 70:1260–1268. PMID: 26233681.
Article66. Turan T, Hızlı D, Yılmaz SS, Gundogdu B, Boran N, Tulunay G, et al. What is the impact of cervical invasion on lymph node metastasis in patients with stage IIIC endometrial cancer? Arch Gynecol Obstet. 2012; 285:1119–1124. PMID: 21898081.
Article67. Haldorsen IS, Berg A, Werner HM, Magnussen IJ, Helland H, Salvesen OO, et al. Magnetic resonance imaging performs better than endocervical curettage for preoperative prediction of cervical stromal invasion in endometrial carcinomas. Gynecol Oncol. 2012; 126:413–418. PMID: 22609110.
Article68. Luomaranta A, Leminen A, Loukovaara M. Magnetic resonance imaging in the assessment of high-risk features of endometrial carcinoma: a meta-analysis. Int J Gynecol Cancer. 2015; 25:837–842. PMID: 25010040.69. Freeman SJ, Aly AM, Kataoka MY, Addley HC, Reinhold C, Sala E. The revised FIGO staging system for uterine malignancies: implications for MR imaging. Radiographics. 2012; 32:1805–1827. PMID: 23065170.
Article70. Lin G, Huang YT, Chao A, Lin YC, Yang LY, Wu RC, et al. Endometrial cancer with cervical stromal invasion: diagnostic accuracy of diffusion-weighted and dynamic contrast enhanced MR imaging at 3T. Eur Radiol. 2017; 27:1867–1876. PMID: 27631107.
Article71. Tamai K, Koyama T, Saga T, Umeoka S, Mikami Y, Fujii S, et al. Diffusion-weighted MR imaging of uterine endometrial cancer. J Magn Reson Imaging. 2007; 26:682–687. PMID: 17729360.
Article72. Nougaret S, Reinhold C, Alsharif SS, Addley H, Arceneau J, Molinari N, et al. Endometrial cancer: combined MR volumetry and diffusion-weighted imaging for assessment of myometrial and lymphovascular invasion and tumor grade. Radiology. 2015; 276:797–808. PMID: 25928157.
Article73. Cao K, Gao M, Sun YS, Li YL, Sun Y, Gao YN, et al. Apparent diffusion coefficient of diffusion weighted MRI in endometrial carcinoma-Relationship with local invasiveness. Eur J Radiol. 2012; 81:1926–1930. PMID: 21592709.
Article74. Shih IL, Yen RF, Chen CA, Chen BB, Wei SY, Chang WC, et al. Standardized uptake value and apparent diffusion coefficient of endometrial cancer evaluated with integrated whole-body PET/MR: correlation with pathological prognostic factors. J Magn Reson Imaging. 2015; 42:1723–1732. PMID: 25919115.
Article75. Zhang J, Cai S, Li C, Sun X, Han X, Yang C, et al. Can magnetic resonance spectroscopy differentiate endometrial cancer? Eur Radiol. 2014; 24:2552–2560. PMID: 24903228.
Article76. Han X, Kang J, Zhang J, Xiu J, Huang Z, Yang C, et al. Can the signal-to-noise ratio of choline in magnetic resonance spectroscopy reflect the aggressiveness of endometrial cancer? Acad Radiol. 2015; 22:453–459. PMID: 25641318.
Article77. Ueno Y, Forghani B, Forghani R, Dohan A, Zeng XZ, Chamming's F, et al. Endometrial carcinoma: MR imaging-based texture model for preoperative risk stratification-A preliminary analysis. Radiology. 2017; 284:748–757. PMID: 28493790.78. Brown AP, Gaffney DK, Dodson MK, Soisson AP, Belnap TW, Alleman K, et al. Survival analysis of endometrial cancer patients with positive lymph nodes. Int J Gynecol Cancer. 2013; 23:861–868. PMID: 23598890.
Article79. Creasman WT, Odicino F, Maisonneuve P, Quinn MA, Beller U, Benedet JL, et al. Carcinoma of the corpus uteri. FIGO 26th annual report on the results of treatment in gynecological cancer. Int J Gynaecol Obstet. 2006; 95(Suppl 1):S105–S143.80. ASTEC study group. Kitchener H, Swart AM, Qian Q, Amos C, Parmar MK. Efficacy of systematic pelvic lymphadenectomy in endometrial cancer (MRC ASTEC trial): a randomised study. Lancet. 2009; 373:125–136. PMID: 19070889.81. Benedetti Panici P, Basile S, Maneschi F, Alberto Lissoni A, Signorelli M, Scambia G, et al. Systematic pelvic lymphadenectomy vs. no lymphadenectomy in early-stage endometrial carcinoma: randomized clinical trial. J Natl Cancer Inst. 2008; 100:1707–1716. PMID: 19033573.82. Todo Y, Kato H, Kaneuchi M, Watari H, Takeda M, Sakuragi N. Survival effect of para-aortic lymphadenectomy in endometrial cancer (SEPAL study): a retrospective cohort analysis. Lancet. 2010; 375:1165–1172. PMID: 20188410.
Article83. Eggemann H, Ignatov T, Kaiser K, Burger E, Costa SD, Ignatov A. Survival advantage of lymphadenectomy in endometrial cancer. J Cancer Res Clin Oncol. 2016; 142:1051–1060. PMID: 26746654.
Article84. Lin G, Ho KC, Wang JJ, Ng KK, Wai YY, Chen YT, et al. Detection of lymph node metastasis in cervical and uterine cancers by diffusion-weighted magnetic resonance imaging at 3T. J Magn Reson Imaging. 2008; 28:128–135. PMID: 18581404.
Article85. Park JY, Kim EN, Kim DY, Suh DS, Kim JH, Kim YM, et al. Comparison of the validity of magnetic resonance imaging and positron emission tomography/computed tomography in the preoperative evaluation of patients with uterine corpus cancer. Gynecol Oncol. 2008; 108:486–492. PMID: 18201753.
Article86. Lai CH, Lin G, Yen TC, Liu FY. Molecular imaging in the management of gynecologic malignancies. Gynecol Oncol. 2014; 135:156–162. PMID: 25065896.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Extranodal Malignant Lymphoma Concurrent Involving Female Urethra and Cervix: a Case Report
- Evaluation of malignant intraductal papillary mucinous neoplasms of the pancreas on computed tomography and magnetic resonance imaging
- MRI Findings and Differential Diagnosis of Benign and Malignant Tumors of the Uterine Corpus
- Uterine Lipoma: A Case Report
- Modern Brain Tumor Imaging