J Cardiovasc Imaging.
2018 Sep;26(3):165-174. 10.4250/jcvi.2018.26.e19.
Early Experiences Using Cocoon Occluders for Closure of a Ventricular Septal Defect
- Affiliations
-
- 1Department of Pediatrics, Samsung Medical Center, Sungkyunkwan University School of Medicine, Seoul, Korea. amyjys@naver.com
- KMID: 2429858
- DOI: http://doi.org/10.4250/jcvi.2018.26.e19
Abstract
- BACKGROUND
Transcatheter device closure of ventricular septal defect (VSD) has become an attractive alternative to surgery. We report here on our early experiences of transcatheter closure of VSD using Cocoon devices, which are now available in Korea.
METHODS
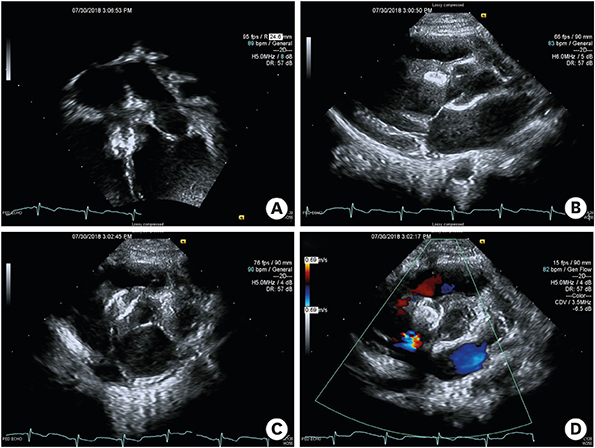
We reviewed the medical records and angiographic images of 13 patients who underwent transcatheter closure of VSD with a Cocoon occluder at Samsung Medical Center. The median patient age was 5.8 years, and the median patient weight was 20.3 kg.
RESULTS
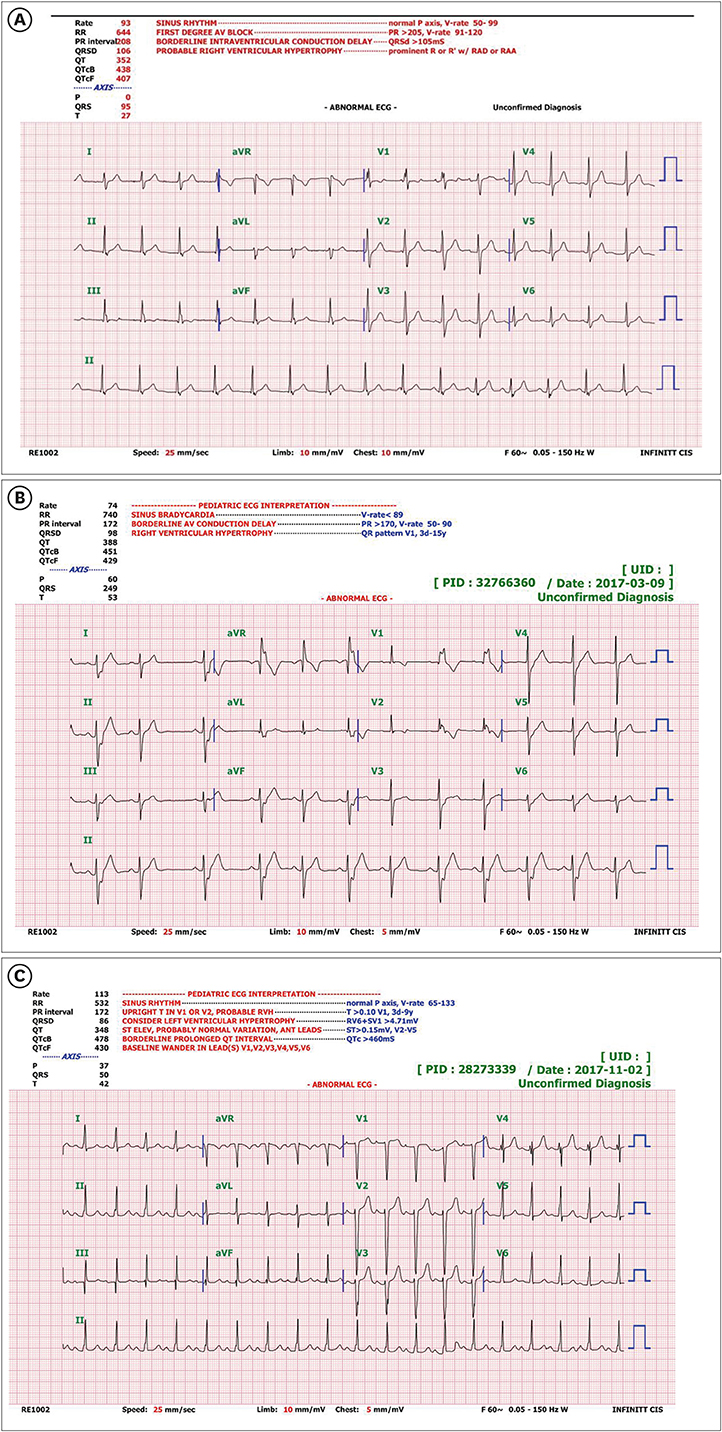
The device was successfully implanted in all patients. The follow-up period was 10 ± 6 months, and no mortality was observed. An immediate residual leak on the next day was detected in 7 patients (including 4 with perimembranous VSD). However, 6 months later, a residual leak was observed only in 2 patients with perimembranous VSD and 1 patient with muscular VSD. Early conduction abnormalities were observed in 3 patients, all of whom had perimembranous VSD, but no significant complete atrioventricular block was observed. One patient experienced newly developed significant aortic regurgitation that decreased spontaneously but still existed at the 6 month follow-up. No hemolysis or embolization was noted on the next day or during the follow-up period.
CONCLUSIONS
Cocoon devices can be used safely and effectively for VSD closure. However, residual leaks and conduction abnormalities may occur early after implantation, especially in patients with perimembranous VSD. Although normal conduction was recovered, long-term evaluation remains essential.
MeSH Terms
Figure
Reference
-
1. Lee SM, Song JY, Choi JY, et al. Transcatheter closure of perimembranous ventricular septal defect using Amplatzer ductal occluder. Catheter Cardiovasc Interv. 2013; 82:1141–1146.
Article2. Diab KA, Cao QL, Hijazi ZM. Device closure of congenital ventricular septal defects. Congenit Heart Dis. 2007; 2:92–103.
Article3. Butera G, Chessa M, Carminati M. Percutaneous closure of ventricular septal defects. Cardiol Young. 2007; 17:243–253.
Article4. Janorkar S, Goh T, Wilkinson J. Transcatheter closure of ventricular septal defects using the Rashkind device: initial experience. Catheter Cardiovasc Interv. 1999; 46:43–48.
Article5. Balzer D. Current status of percutaneous closure of ventricular septal defects. Pediatr Ther. 2012; 2:112.
Article6. Holzer R, Balzer D, Cao QL, Lock K, Hijazi ZM. Amplatzer Muscular Ventricular Septal Defect Investigators. Device closure of muscular ventricular septal defects using the Amplatzer muscular ventricular septal defect occluder: immediate and mid-term results of a U.S. registry. J Am Coll Cardiol. 2004; 43:1257–1263.
Article7. Fu YC. Transcatheter device closure of muscular ventricular septal defect. Pediatr Neonatol. 2011; 52:3–4.
Article8. Pedra CA, Pedra SR, Esteves CA, et al. Percutaneous closure of perimembranous ventricular septal defects with the Amplatzer device: technical and morphological considerations. Catheter Cardiovasc Interv. 2004; 61:403–410.
Article9. Zuo J, Xie J, Yi W, et al. Results of transcatheter closure of perimembranous ventricular septal defect. Am J Cardiol. 2010; 106:1034–1037.
Article10. Holzer R, de Giovanni J, Walsh KP, et al. Transcatheter closure of perimembranous ventricular septal defects using the amplatzer membranous VSD occluder: immediate and midterm results of an international registry. Catheter Cardiovasc Interv. 2006; 68:620–628.
Article11. Ghosh S, Sridhar A, Sivaprakasam M. Complete heart block following transcatheter closure of perimembranous VSD using amplatzer duct occluder II. Catheter Cardiovasc Interv. 2017; Jul. 14. [Epub ahead of print].
Article12. Ghaderian M, Merajie M, Mortezaeian H, Aarabi M, Mohammad Y, Shah Mohammadi A. Efficacy and safety of using amplatzer ductal occluder for transcatheter closure of perimembranous ventricular septal defect in pediatrics. Iran J Pediatr. 2015; 25:e386.
Article13. Lock JE, Block PC, McKay RG, Baim DS, Keane JF. Transcatheter closure of ventricular septal defects. Circulation. 1988; 78:361–368.
Article14. Hijazi ZM, Hakim F, Haweleh AA, et al. Catheter closure of perimembranous ventricular septal defects using the new Amplatzer membranous VSD occluder: initial clinical experience. Catheter Cardiovasc Interv. 2002; 56:508–515.
Article15. Yang L, Tai BC, Khin LW, Quek SC. A systematic review on the efficacy and safety of transcatheter device closure of ventricular septal defects (VSD). J Interv Cardiol. 2014; 27:260–272.
Article16. Hijazi ZM, Hakim F, Al-Fadley F, Abdelhamid J, Cao QL. Transcatheter closure of single muscular ventricular septal defects using the amplatzer muscular VSD occluder: initial results and technical considerations. Catheter Cardiovasc Interv. 2000; 49:167–172.
Article17. Kitagawa T, Durham LA 3rd, Mosca RS, Bove EL. Techniques and results in the management of multiple ventricular septal defects. J Thorac Cardiovasc Surg. 1998; 115:848–856.
Article18. Saurav A, Kaushik M, Mahesh Alla V, et al. Comparison of percutaneous device closure versus surgical closure of peri-membranous ventricular septal defects: A systematic review and meta-analysis. Catheter Cardiovasc Interv. 2015; 86:1048–1056.
Article19. Moodie DS. Technology Insight: transcatheter closure of ventricular septal defects. Nat Clin Pract Cardiovasc Med. 2005; 2:592–596.
Article20. Santhanam H, Yang L, Chen Z, Tai BC, Rajgor DD, Quek SC. A meta-analysis of transcatheter device closure of perimembranous ventricular septal defect. Int J Cardiol. 2018; 254:75–83.
Article21. Yang R, Kong XQ, Sheng YH, et al. Risk factors and outcomes of post-procedure heart blocks after transcatheter device closure of perimembranous ventricular septal defect. JACC Cardiovasc Interv. 2012; 5:422–427.
Article22. Li P, Zhao XX, Zheng X, Qin YW. Arrhythmias after transcatheter closure of perimembranous ventricular septal defects with a modified double-disk occluder: early and long-term results. Heart Vessels. 2012; 27:405–410.
Article23. Zhao LJ, Han B, Zhang JJ, Yi YC, Jiang DD, Lyu JL. Postprocedural outcomes and risk factors for arrhythmias following transcatheter closure of congenital perimembranous ventricular septal defect: a single-center retrospective study. Chin Med J (Engl). 2017; 130:516–521.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Erratum: Early Experiences Using Cocoon Occluders for Closure of a Ventricular Septal Defect
- Reoperation of Postinfarction VSD
- Cinical Study of Spontaneous Closure in Simple Ventricular Septal Defects
- The muscular ventricular septal defect and multiple ventricular septal defects in some congenital heart diseases
- A Case of Traumatic Ventricular Septal Defect Secondary to Nonpenetrating Chest Trauma