Korean J Radiol.
2017 Oct;18(5):786-798. 10.3348/kjr.2017.18.5.786.
CT Features of Vasculitides Based on the 2012 International Chapel Hill Consensus Conference Revised Classification
- Affiliations
-
- 1Department of Radiology, Seoul National University Bundang Hospital, Seongnam 13620, Korea. drejchun@hanmail.net
- 2Department of Radiology, Kangbuk Samsung Hospital, Sungkyunkwan University School of Medicine, Seoul 03181, Korea.
- 3Department of Radiology, Seoul National University Hospital, Seoul 03080, Korea.
- KMID: 2427215
- DOI: http://doi.org/10.3348/kjr.2017.18.5.786
Abstract
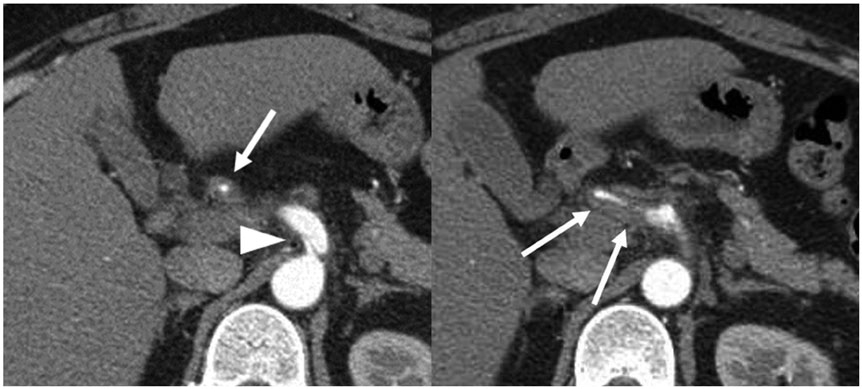
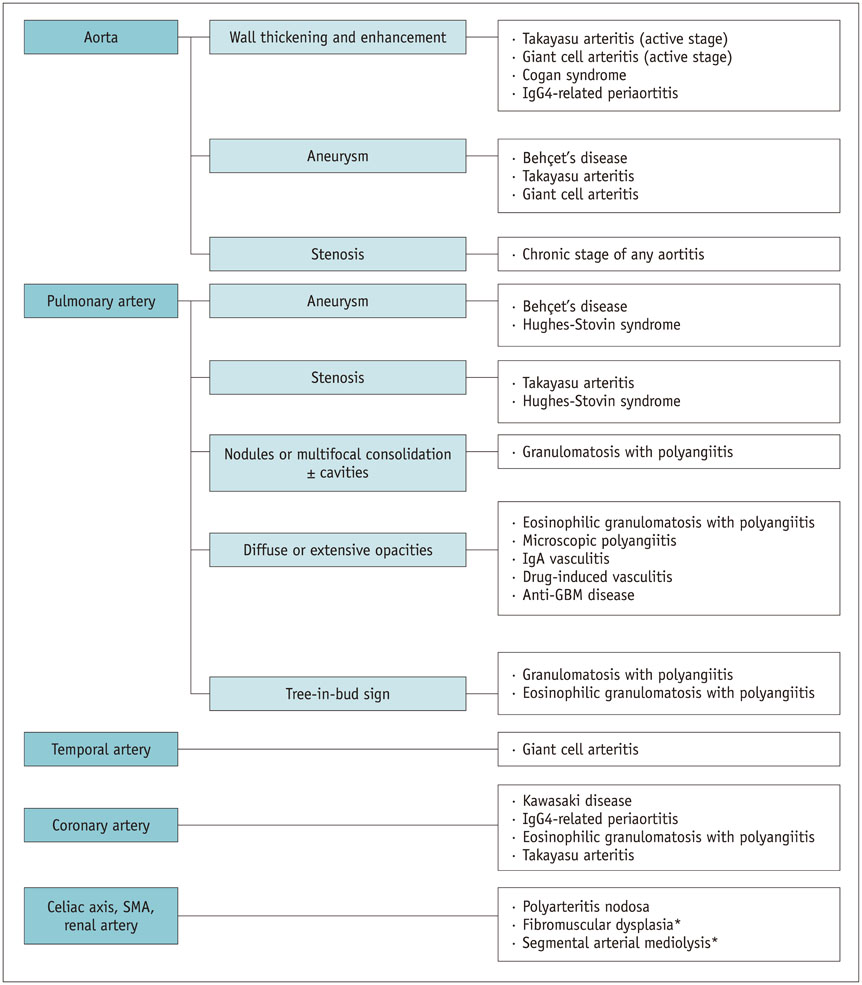
- Vasculitis, characterized by inflammation of vessel walls, is comprised of heterogeneous clinicopathological entities, and thus poses a diagnostic challenge. The most widely used approach for classifying vasculitides is based on the International Chapel Hill Consensus Conference (CHCC) nomenclature system. Based on the recently revised CHCC 2012, we propose computed tomography (CT) features of vasculitides and a differential diagnosis based on location and morphological characteristics. Finally, vasculitis mimics should be differentiated, because erroneous application of immunosuppressive drugs on vasculitis mimics may be ineffective, even deteriorating. This article presents the utility of CT in the diagnosis and differential diagnosis of vasculitides.
MeSH Terms
-
Behcet Syndrome/diagnosis/diagnostic imaging
Cogan Syndrome/diagnosis/diagnostic imaging
Consensus
Diagnosis, Differential
Giant Cell Arteritis/diagnosis/diagnostic imaging
Granulomatosis with Polyangiitis/diagnosis/diagnostic imaging
Humans
Mucocutaneous Lymph Node Syndrome/diagnosis/diagnostic imaging
Polyarteritis Nodosa/diagnosis/diagnostic imaging
Takayasu Arteritis/diagnosis/diagnostic imaging
Tomography, X-Ray Computed
Vasculitis/*diagnosis/diagnostic imaging
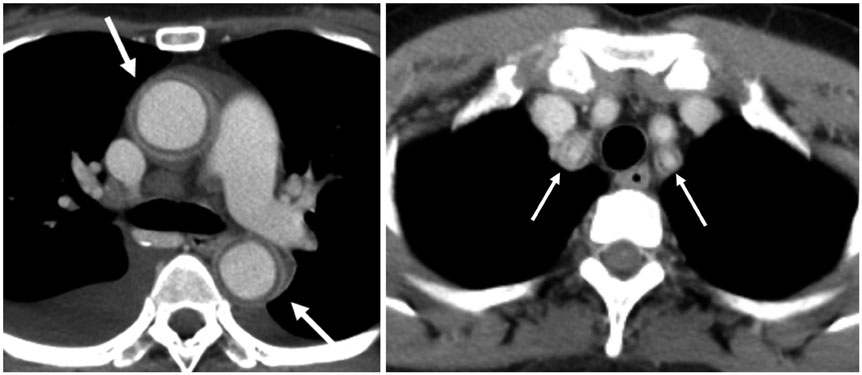
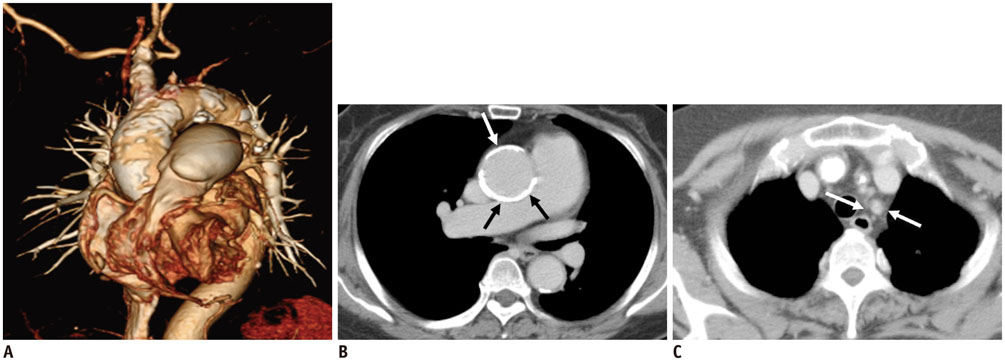
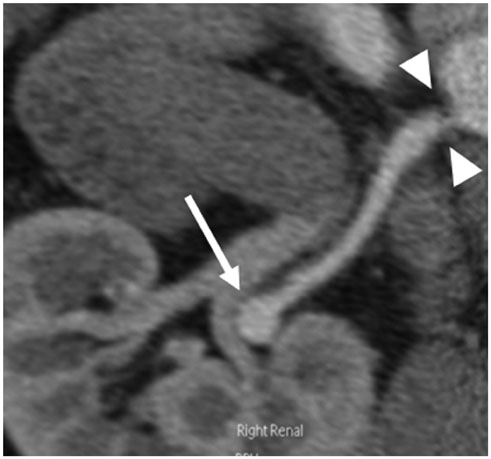
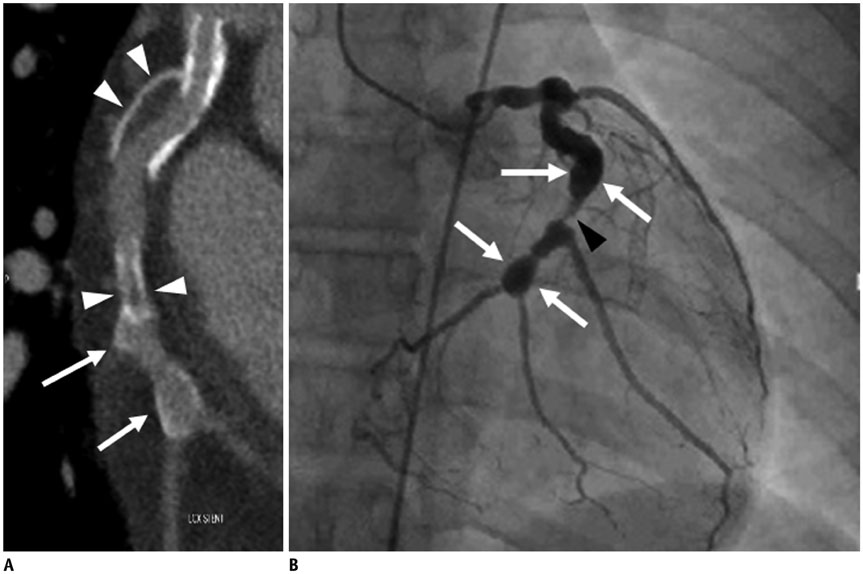
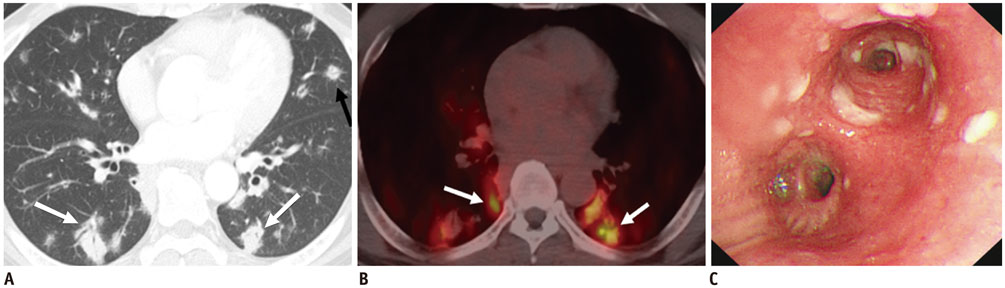
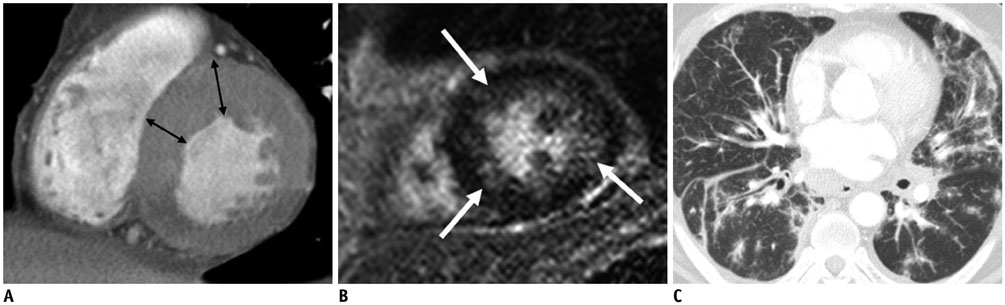
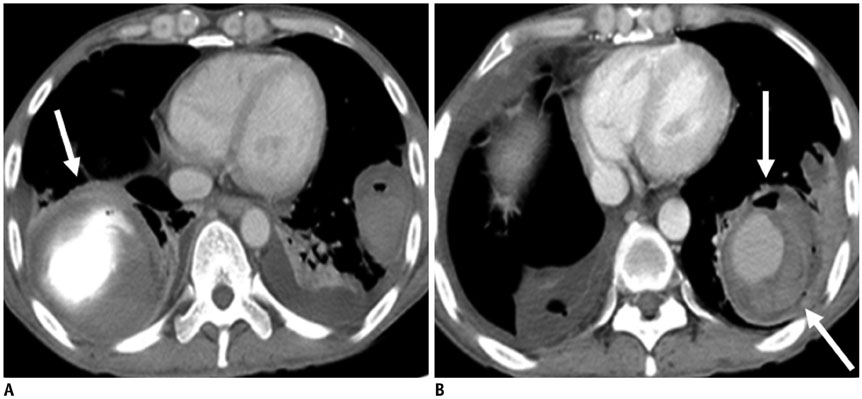
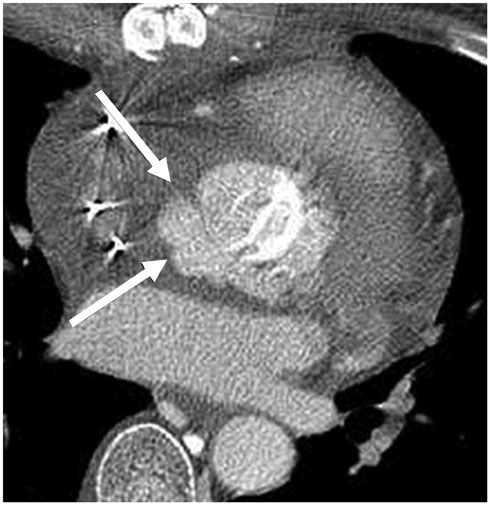
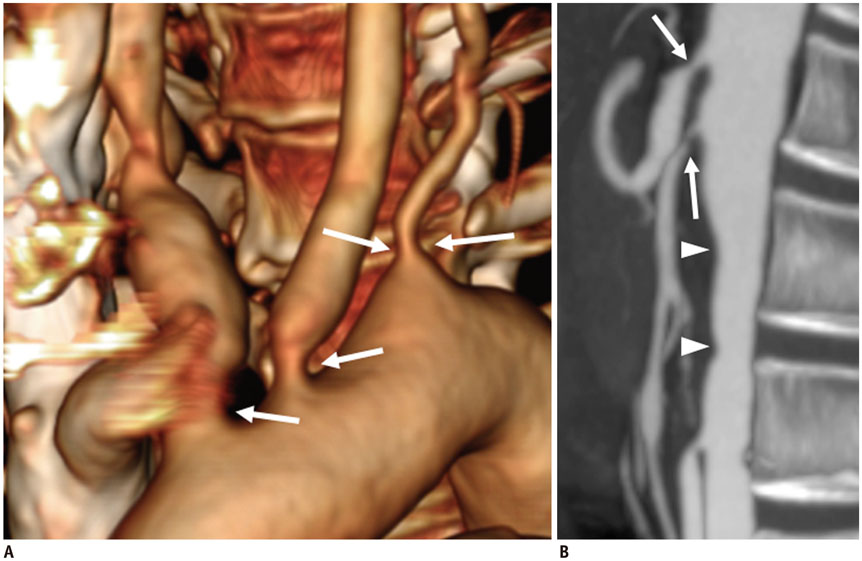
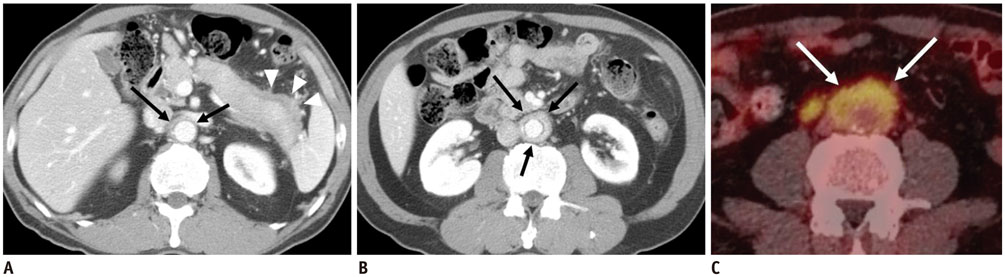
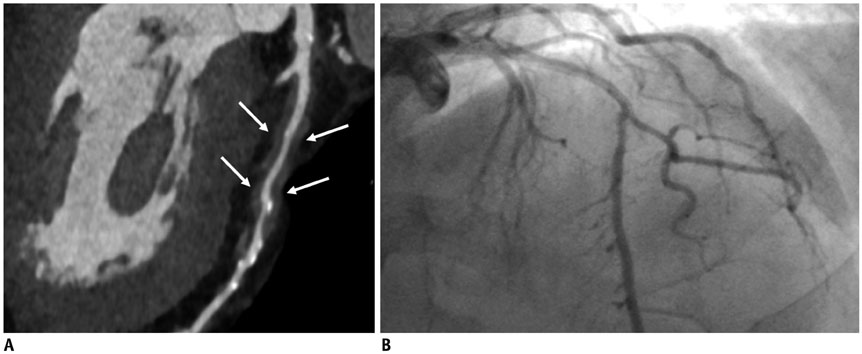
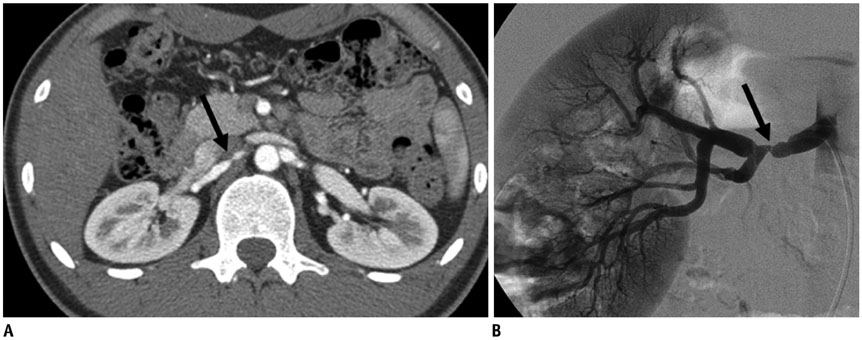
Figure
Reference
-
1. Restrepo CS, Ocazionez D, Suri R, Vargas D. Aortitis: imaging spectrum of the infectious and inflammatory conditions of the aorta. Radiographics. 2011; 31:435–451.2. Castañer E, Alguersuari A, Gallardo X, Andreu M, Pallardó Y, Mata JM, et al. When to suspect pulmonary vasculitis: radiologic and clinical clues. Radiographics. 2010; 30:33–53.3. Kim YJ, Yong HS, Kim SM, Kim JA, Yang DH, Hong YJ, et al. Korean guidelines for the appropriate use of cardiac CT. Korean J Radiol. 2015; 16:251–285.4. Jennette JC, Falk RJ, Bacon PA, Basu N, Cid MC, Ferrario F, et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 2013; 65:1–11.5. Jennette JC. Overview of the 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Clin Exp Nephrol. 2013; 17:603–606.6. Kallenberg CG. Key advances in the clinical approach to ANCA-associated vasculitis. Nat Rev Rheumatol. 2014; 10:484–493.7. Khanna G, Sargar K, Baszis KW. Pediatric vasculitis: recognizing multisystemic manifestations at body imaging. Radiographics. 2015; 35:849–865.8. de Souza AW, de Carvalho JF. Diagnostic and classification criteria of Takayasu arteritis. J Autoimmun. 2014; 48-49:79–83.9. Kang EJ, Kim SM, Choe YH, Lee GY, Lee KN, Kim DK. Takayasu arteritis: assessment of coronary arterial abnormalities with 128-section dual-source CT angiography of the coronary arteries and aorta. Radiology. 2014; 270:74–81.10. Gornik HL, Creager MA. Aortitis. Circulation. 2008; 117:3039–3051.11. Koide K. Takayasu arteritis in Japan. Heart Vessels Suppl. 1992; 7:48–54.12. Park YB, Hong SK, Choi KJ, Sohn DW, Oh BH, Lee MM, et al. Takayasu arteritis in Korea: clinical and angiographic features. Heart Vessels Suppl. 1992; 7:55–59.13. Lim AY, Lee GY, Jang SY, Gwag HB, Choi SH, Jeon ES, et al. Gender differences in clinical and angiographic findings of patients with Takayasu arteritis. Clin Exp Rheumatol. 2015; 33:2 Suppl 89. S-132–S-137.14. Wolfe F, Smythe HA, Yunus MB, Bennett RM, Bombardier C, Goldenberg DL, et al. The American College of Rheumatology 1990 Criteria for the Classification of Fibromyalgia. Report of the Multicenter Criteria Committee. Arthritis Rheum. 1990; 33:160–172.15. Hata A, Noda M, Moriwaki R, Numano F. Angiographic findings of Takayasu arteritis: new classification. Int J Cardiol. 1996; 54:S155–S163.16. Zhu FP, Luo S, Wang ZJ, Jin ZY, Zhang LJ, Lu GM. Takayasu arteritis: imaging spectrum at multidetector CT angiography. Br J Radiol. 2012; 85:e1282–e1292.17. Gotway MB, Araoz PA, Macedo TA, Stanson AW, Higgins CB, Ring EJ, et al. Imaging findings in Takayasu’s arteritis. AJR Am J Roentgenol. 2005; 184:1945–1950.18. Mason JC. Takayasu arteritis--advances in diagnosis and management. Nat Rev Rheumatol. 2010; 6:406–415.19. Park JH, Chung JW, Im JG, Kim SK, Park YB, Han MC. Takayasu arteritis: evaluation of mural changes in the aorta and pulmonary artery with CT angiography. Radiology. 1995; 196:89–93.20. Khandelwal N, Kalra N, Garg MK, Kang M, Lal A, Jain S, et al. Multidetector CT angiography in Takayasu arteritis. Eur J Radiol. 2011; 77:369–374.21. Yamato M, Lecky JW, Hiramatsu K, Kohda E. Takayasu arteritis: radiographic and angiographic findings in 59 patients. Radiology. 1986; 161:329–334.22. Calvo-Romero JM. Giant cell arteritis. Postgrad Med J. 2003; 79:511–515.23. Stanson AW, Friese JL, Johnson CM, McKusick MA, Breen JF, Sabater EA, et al. Polyarteritis nodosa: spectrum of angiographic findings. Radiographics. 2001; 21:151–159.24. Guillevin L, Lhote F, Cohen P, Sauvaget F, Jarrousse B, Lortholary O, et al. Polyarteritis nodosa related to hepatitis B virus. A prospective study with long-term observation of 41 patients. Medicine (Baltimore). 1995; 74:238–253.25. Díaz-Zamudio M, Bacilio-Pérez U, Herrera-Zarza MC, Meave-González A, Alexanderson-Rosas E, Zambrana-Balta GF, et al. Coronary artery aneurysms and ectasia: role of coronary CT angiography. Radiographics. 2009; 29:1939–1954.26. Chung CJ, Stein L. Kawasaki disease: a review. Radiology. 1998; 208:25–33.27. Jeon CH, Kim YK, Chun EJ, Kim JA, Yong HS, Doo KW, et al. Coronary artery vasculitis: assessment with cardiac multi-detector computed tomography. Int J Cardiovasc Imaging. 2015; 31:Suppl 1. 59–67.28. Suresh E. Diagnostic approach to patients with suspected vasculitis. Postgrad Med J. 2006; 82:483–488.29. Martinez F, Chung JH, Digumarthy SR, Kanne JP, Abbott GF, Shepard JA, et al. Common and uncommon manifestations of Wegener granulomatosis at chest CT: radiologic-pathologic correlation. Radiographics. 2012; 32:51–69.30. Almouhawis HA, Leao JC, Fedele S, Porter SR. Wegener’s granulomatosis: a review of clinical features and an update in diagnosis and treatment. J Oral Pathol Med. 2013; 42:507–516.31. Lohrmann C, Uhl M, Kotter E, Burger D, Ghanem N, Langer M. Pulmonary manifestations of Wegener granulomatosis: CT findings in 57 patients and a review of the literature. Eur J Radiol. 2005; 53:471–477.32. Ananthakrishnan L, Sharma N, Kanne JP. Wegener’s granulomatosis in the chest: high-resolution CT findings. AJR Am J Roentgenol. 2009; 192:676–682.33. Comarmond C, Pagnoux C, Khellaf M, Cordier JF, Hamidou M, Viallard JF, et al. Eosinophilic granulomatosis with polyangiitis (Churg-Strauss): clinical characteristics and long-term followup of the 383 patients enrolled in the French Vasculitis Study Group cohort. Arthritis Rheum. 2013; 65:270–281.34. Worthy SA, Müller NL, Hansell DM, Flower CD. Churg-Strauss syndrome: the spectrum of pulmonary CT findings in 17 patients. AJR Am J Roentgenol. 1998; 170:297–300.35. Knockaert DC. Cardiac involvement in systemic inflammatory diseases. Eur Heart J. 2007; 28:1797–1804.36. Yune S, Choi DC, Lee BJ, Lee JY, Jeon ES, Kim SM, et al. Detecting cardiac involvement with magnetic resonance in patients with active eosinophilic granulomatosis with polyangiitis. Int J Cardiovasc Imaging. 2016; 32:Suppl 1. 155–162.37. Chae EJ, Do KH, Seo JB, Park SH, Kang JW, Jang YM, et al. Radiologic and clinical findings of BehcXMLLink_XYZet disease: comprehensive review of multisystemic involvement. Radiographics. 2008; 28:e31.38. Owlia MB, Mehrpoor G. Behcet’s disease: new concepts in cardiovascular involvements and future direction for treatment. ISRN Pharmacol. 2012; 2012:760484.39. Chung MP, Yi CA, Lee HY, Han J, Lee KS. Imaging of pulmonary vasculitis. Radiology. 2010; 255:322–341.40. Marzban M, Mandegar MH, Karimi A, Abbasi K, Movahedi N, Navabi MA, et al. Cardiac and great vessel involvement in “Behcet’s disease”. J Card Surg. 2008; 23:765–768.41. Grasland A, Pouchot J, Hachulla E, Blétry O, Papo T, Vinceneux P. Study Group for Cogan's Syndrome. Typical and atypical Cogan’s syndrome: 32 cases and review of the literature. Rheumatology (Oxford). 2004; 43:1007–1015.42. Inoue D, Zen Y, Abo H, Gabata T, Demachi H, Yoshikawa J, et al. Immunoglobulin G4-related periaortitis and periarteritis: CT findings in 17 patients. Radiology. 2011; 261:625–633.43. Molloy ES, Langford CA. Vasculitis mimics. Curr Opin Rheumatol. 2008; 20:29–34.44. Filippone EJ, Foy A, Galanis T, Pokuah M, Newman E, Lallas CD, et al. Segmental arterial mediolysis: report of 2 cases and review of the literature. Am J Kidney Dis. 2011; 58:981–987.45. Baker-LePain JC, Stone DH, Mattis AN, Nakamura MC, Fye KH. Clinical diagnosis of segmental arterial mediolysis: differentiation from vasculitis and other mimics. Arthritis Care Res (Hoboken). 2010; 62:1655–1160.
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Classification of Antineutrophil Cytoplasmic Antibody-associated Vasculitis
- Update on the Classification Criteria for Vasculitis
- Comparison of the 2022 ACR/EULAR Classification Criteria for Antineutrophil Cytoplasmic Antibody-Associated Vasculitis with Previous Criteria
- Antineutrophil Cytoplasmic Antibody-Associated Vasculitis in Korea: A Narrative Review
- Pitfalls in the Interpretation of Chicago Classification for Esophageal Motility Disorders