Korean J Radiol.
2018 Feb;19(1):130-138. 10.3348/kjr.2018.19.1.130.
Assessing the Blood Supply Status of the Focal Ground-Glass Opacity in Lungs Using Spectral Computed Tomography
- Affiliations
-
- 1Department of Radiology, First Affiliated Hospital of Dalian Medical University, Liaoning 116011, China. zjy_lzy@163.com
- KMID: 2425118
- DOI: http://doi.org/10.3348/kjr.2018.19.1.130
Abstract
OBJECTIVE
To exploit material decomposition analysis in dual-energy spectral computed tomography (CT) to assess the blood supply status of the ground-glass opacity (GGO) in lungs.
MATERIALS AND METHODS
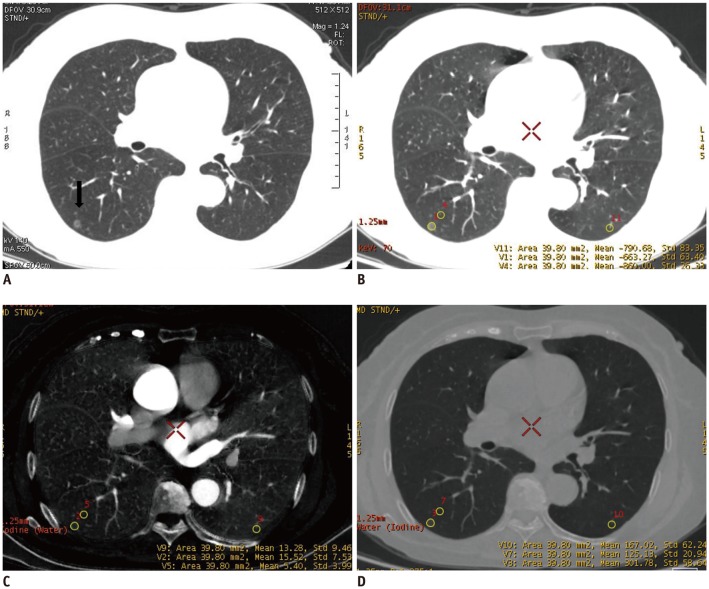
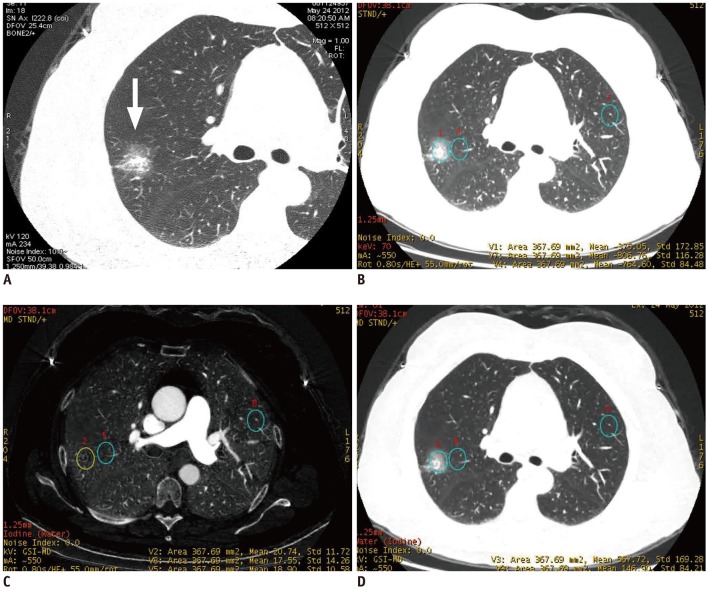
This retrospective study included 48 patients with lung adenocarcinoma, who underwent a contrast-enhanced dual-energy spectral CT scan before treatment (53 GGOs in total). The iodine concentration (IC) and water content (WC) of the GGO, the contralateral and ipsilateral normal lung tissues were measured in the arterial phase (AP) and their differences were analyzed. IC, normalized IC (NIC), and WC values were compared between the pure ground-glass opacity (pGGO) and the mixed ground-glass opacity (mGGO), and between the group of preinvasive lesions and the minimally invasive adenocarcinoma (MIA) and invasive adenocarcinoma (IA) groups.
RESULTS
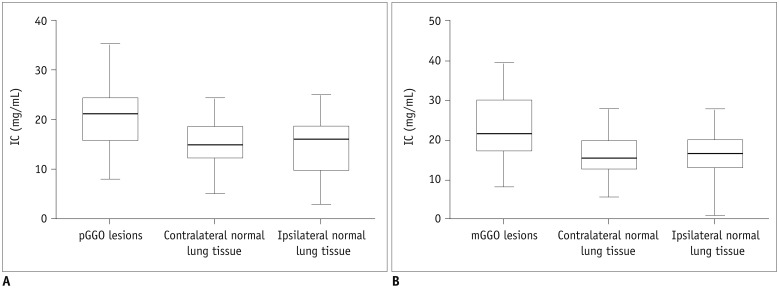
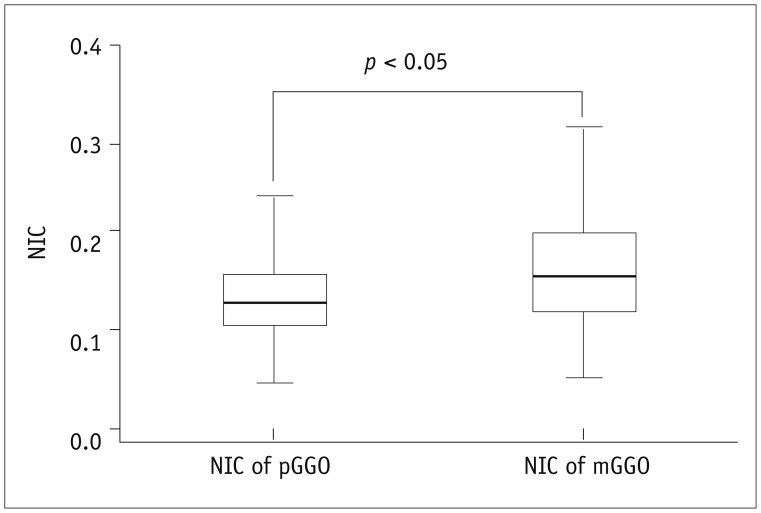
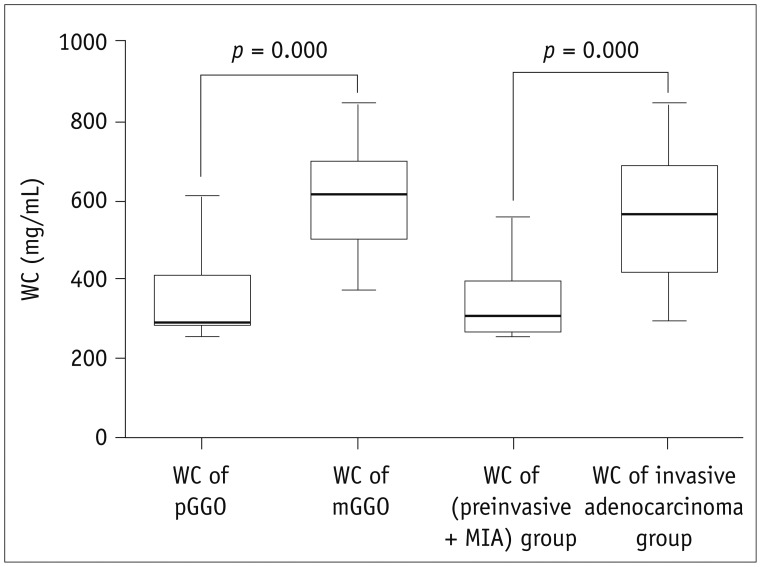
The values of pGGO (IC = 20.9 ± 6.2 mg/mL and WC = 345.1 ± 87.1 mg/mL) and mGGO (IC = 23.8 ± 8.3 mg/mL and WC = 606.8 ± 124.5 mg/mL) in the AP were significantly higher than those of the contralateral normal lung tissues (IC = 15.0 ± 4.9 mg/mL and WC = 156.4 ± 36.8 mg/mL; IC = 16.2 ± 5.7 mg/mL and WC = 169.4 ± 41.0 mg/mL) and ipsilateral normal lung tissues (IC = 15.1 ± 6.2 mg/mL and WC = 156.3 ± 38.8 mg/mL; IC = 15.9 ± 6.0 mg/mL and WC = 174.7 ± 39.2 mg/mL; all p < 0.001). After normalizing the data according to the values of the artery, pGGO (NIC = 0.1 and WC = 345.1 ± 87.1 mg/mL) and mGGO (NIC = 0.2 and WC = 606.8 ± 124.5 mg/mL) were statistically different (p = 0.049 and p < 0.001, respectively), but not for the IC value (p = 0.161). The WC values of the group with preinvasive lesions and MIA (345.4 ± 96.1 mg/mL) and IA (550.1 ± 158.2 mg/mL) were statistically different (p < 0.001).
CONCLUSION
Using dual-energy spectral CT and material decomposition analysis, the IC in GGO can be quantitatively measured which can be an indicator of the blood supply status in the GGO.
Keyword
MeSH Terms
Figure
Reference
-
1. Henschke CI, Yankelevitz DF, Mirtcheva R, McGuinness G, McCauley D, Miettinen OS. ELCAP Group. CT screening for lung cancer: frequency and significance of part-solid and nonsolid nodules. AJR Am J Roentgenol. 2002; 178:1053–1057. PMID: 11959700.2. Sudo N, Nambu A, Yamakawa T, Kawamoto M, Fujino S, Watanabe M, et al. Pulmonary focal fibrosis associated with microscopic arterio-venous fistula manifesting as focal ground-glass opacity on thin-section CT. BMC Pulm Med. 2013; 13:3. PMID: 23316757.
Article3. Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, et al. International association for the study of lung cancer/American thoracic society/European respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol. 2011; 6:244–285. PMID: 21252716.4. Eguchi T, Kondo R, Kawakami S, Matsushita M, Yoshizawa A, Hara D, et al. Computed tomography attenuation predicts the growth of pure ground-glass nodules. Lung Cancer. 2014; 84:242–247. PMID: 24681281.
Article5. Matsuguma H, Oki I, Nakahara R, Suzuki H, Kasai T, Kamiyama Y, et al. Comparison of three measurements on computed tomography for the prediction of less invasiveness in patients with clinical stage I non-small cell lung cancer. Ann Thorac Surg. 2013; 95:1878–1884. PMID: 23618519.
Article6. Maeyashiki T, Suzuki K, Hattori A, Matsunaga T, Takamochi K, Oh S. The size of consolidation on thin-section computed tomography is a better predictor of survival than the maximum tumour dimension in resectable lung cancer. Eur J Cardiothorac Surg. 2013; 43:915–918. PMID: 23024235.
Article7. Murakawa T, Konoeda C, Ito T, Inoue Y, Sano A, Nagayama K, et al. The ground glass opacity component can be eliminated from the T-factor assessment of lung adenocarcinoma. Eur J Cardiothorac Surg. 2013; 43:925–932. PMID: 23047267.
Article8. Lee SM, Park CM, Goo JM, Lee HJ, Wi JY, Kang CH. Invasive pulmonary adenocarcinomas versus preinvasive lesions appearing as ground-glass nodules: differentiation by using CT features. Radiology. 2013; 268:265–273. PMID: 23468575.
Article9. Eguchi T, Yoshizawa A, Kawakami S, Kumeda H, Umesaki T, Agatsuma H, et al. Tumor size and computed tomography attenuation of pulmonary pure ground-glass nodules are useful for predicting pathological invasiveness. PLoS One. 2014; 9:e97867. PMID: 24846292.
Article10. Hasegawa M, Sone S, Takashima S, Li F, Yang ZG, Maruyama Y, et al. Growth rate of small lung cancers detected on mass CT screening. Br J Radiol. 2000; 73:1252–1259. PMID: 11205667.
Article11. Chang B, Hwang JH, Choi YH, Chung MP, Kim H, Kwon OJ, et al. Natural history of pure ground-glass opacity lung nodules detected by low-dose CT scan. Chest. 2013; 143:172–178. PMID: 22797081.
Article12. Cho S, Yang H, Kim K, Jheon S. Pathology and prognosis of persistent stable pure ground-glass opacity nodules after surgical resection. Ann Thorac Surg. 2013; 96:1190–1195. PMID: 23968760.
Article13. Lee SW, Leem CS, Kim TJ, Lee KW, Chung JH, Jheon S, et al. The long-term course of ground-glass opacities detected on thin-section computed tomography. Respir Med. 2013; 107:904–910. PMID: 23514949.
Article14. Tsunezuka Y, Shimizu Y, Tanaka N, Takayanagi T, Kawano M. Positron emission tomography in relation to Noguchi's classification for diagnosis of peripheral non-small-cell lung cancer 2 cm or less in size. World J Surg. 2007; 31:314–317. PMID: 17219276.
Article15. Uehara H, Tsutani Y, Okumura S, Nakayama H, Adachi S, Yoshimura M, et al. Prognostic role of positron emission tomography and high-resolution computed tomography in clinical stage IA lung adenocarcinoma. Ann Thorac Surg. 2013; 96:1958–1965. PMID: 24021765.
Article16. Yuan X, Zhang J, Quan C, Cao J, Ao G, Tian Y, et al. Differentiation of malignant and benign pulmonary nodules with first-pass dual-input perfusion CT. Eur Radiol. 2013; 23:2469–2474. PMID: 23793548.
Article17. Shi J, Schmid-Bindert G, Fink C, Sudarski S, Apfaltrer P, Pilz LR, et al. Dynamic volume perfusion CT in patients with lung cancer: baseline perfusion characteristics of different histological subtypes. Eur J Radiol. 2013; 82:e894–e900. PMID: 24094644.
Article18. Johnson TR, Krauss B, Sedlmair M, Grasruck M, Bruder H, Morhard D, et al. Material differentiation by dual energy CT: initial experience. Eur Radiol. 2007; 17:1510–1517. PMID: 17151859.
Article19. Chae EJ, Song JW, Krauss B, Song KS, Lee CW, Lee HJ, et al. Dual-energy computed tomography characterization of solitary pulmonary nodules. J Thorac Imaging. 2010; 25:301–310. PMID: 21042068.
Article20. Chae EJ, Song JW, Seo JB, Krauss B, Jang YM, Song KS. Clinical utility of dual-energy CT in the evaluation of solitary pulmonary nodules: initial experience. Radiology. 2008; 249:671–681. PMID: 18796658.
Article21. Kawai T, Shibamoto Y, Hara M, Arakawa T, Nagai K, Ohashi K. Can dual-energy CT evaluate contrast enhancement of ground-glass attenuation? Phantom and preliminary clinical studies. Acad Radiol. 2011; 18:682–689. PMID: 21393031.22. Lee HJ, Goo JM, Lee CH, Yoo CG, Kim YT, Im JG. Nodular ground-glass opacities on thin-section CT: size change during follow-up and pathological results. Korean J Radiol. 2007; 8:22–31. PMID: 17277560.
Article23. Thieme SF, Johnson TR, Lee C, McWilliams J, Becker CR, Reiser MF, et al. Dual-energy CT for the assessment of contrast material distribution in the pulmonary parenchyma. AJR Am J Roentgenol. 2009; 193:144–149. PMID: 19542406.
Article24. Remy-Jardin M, Giraud F, Remy J, Copin MC, Gosselin B, Duhamel A. Importance of ground-glass attenuation in chronic diffuse infiltrative lung disease: pathologic-CT correlation. Radiology. 1993; 189:693–698. PMID: 8234692.
Article
- Full Text Links
- Actions
-
Cited
- CITED
-
- Close
- Share
- Similar articles
-
- Ground-Glass Opacity in Lung Metastasis from Adenocarcinoma of the Stomach: A Case Report
- Paraquat Induced Lung Injury: Long-term Follow-up of HRCT
- High-resolution CT findings of pleuropulmonary involvement in systemic lupus erythematosus
- The Clinical Approach to Nodular Ground Glass Opacity in the Lung
- Sequential Change of Fibrosing Alveolitis On High-Resolution CT: Correlation with Finding at Pulmonary Function Test and Bronchoalveolar Lavage